Advanced Semi-Synthetic Route for Vinpocetine: High-Yield Manufacturing for Global Pharma Supply Chains
Advanced Semi-Synthetic Route for Vinpocetine: High-Yield Manufacturing for Global Pharma Supply Chains
The pharmaceutical landscape for cerebrovascular therapeutics demands robust, scalable, and environmentally compliant manufacturing processes for key active ingredients. Patent CN102702191A introduces a refined semi-synthetic methodology for the production of Vinpocetine, a critical derivative of the natural alkaloid Vincamine. This technology represents a significant leap forward in process chemistry, addressing the longstanding challenges of toxicity and low yield associated with earlier synthetic routes. By leveraging a straightforward two-step sequence involving mesylation followed by base-catalyzed elimination, this method delivers high-purity Vinpocetine suitable for stringent regulatory markets. For R&D directors and procurement strategists, understanding the nuances of this pathway is essential for securing a reliable pharmaceutical intermediates supplier capable of meeting global demand without compromising on quality or environmental standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of Vinpocetine has been plagued by significant technical and environmental hurdles that complicate supply chain stability. Early methodologies, such as the Mondelo patent, relied heavily on acetonitrile as a solvent and utilized 2-fluoro-1,3,5-trinitrobenzene as a catalyst. While these methods offered reasonable yields, the extreme toxicity of acetonitrile poses severe occupational health risks and creates substantial waste disposal liabilities, driving up the operational costs for any fine chemical manufacturer. Furthermore, the reliance on rare and specialized catalysts introduces fragility into the supply chain, making production vulnerable to raw material shortages. Other approaches utilizing titanium tetraethoxide for transesterification have suffered from inherently low total recovery rates, necessitating complex purification steps that erode profit margins and increase energy consumption. These legacy processes are increasingly untenable in a modern regulatory environment that prioritizes green chemistry and cost efficiency.
The Novel Approach
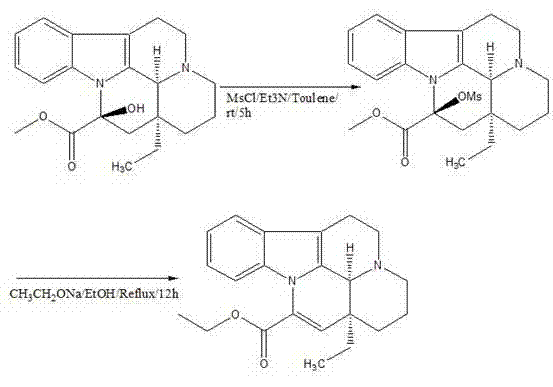
In stark contrast, the methodology disclosed in CN102702191A offers a streamlined, two-step semi-synthetic route that elegantly bypasses these historical bottlenecks. The process initiates with the activation of the hydroxyl group on the Vincamine scaffold using methanesulfonyl chloride (MsCl) in the presence of triethylamine, followed by a clean elimination reaction using sodium ethoxide. This strategic shift eliminates the need for toxic nitrile solvents, replacing them with safer, industry-standard solvents like toluene and ethanol. The result is a process that not only simplifies the operational workflow but also drastically improves the mass balance of the reaction. As illustrated in the reaction scheme below, the transformation is direct and high-yielding, providing a clear pathway for cost reduction in pharmaceutical intermediates manufacturing by minimizing waste and maximizing throughput.
Mechanistic Insights into Mesylation-Elimination Sequence
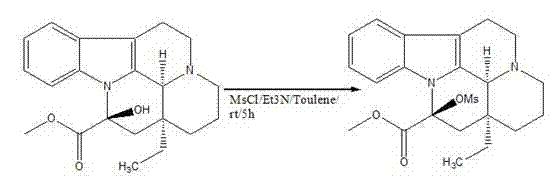
The core of this technological advancement lies in the precise chemical manipulation of the Vincamine molecule to facilitate the formation of the exocyclic double bond characteristic of Vinpocetine. The first stage involves the conversion of the secondary alcohol at the C-14 position into a mesylate ester. This is achieved by treating Vincamine with methanesulfonyl chloride and triethylamine in toluene. The triethylamine acts as a proton scavenger, neutralizing the hydrochloric acid byproduct and driving the equilibrium toward the formation of the activated intermediate. This activation is crucial because the hydroxyl group itself is a poor leaving group; converting it to a mesylate creates an excellent leaving group that primes the molecule for the subsequent elimination step. The reaction is conducted under mild conditions, starting in an ice-water bath and warming to room temperature, which minimizes thermal degradation of the sensitive indole alkaloid skeleton.
Following the isolation of the mesylate intermediate, the second stage employs a classic E2 elimination mechanism to generate the desired alkene. By introducing sodium ethoxide in absolute ethanol at elevated temperatures (80°C), the base abstracts a proton from the adjacent carbon, triggering the simultaneous departure of the mesylate group and the formation of the double bond. A critical aspect of this mechanism is the control of impurities through pH regulation. The patent specifies adjusting the pH to 12 using saturated potassium carbonate or water during the workup. This alkaline environment ensures that acidic impurities remain ionized and soluble in the aqueous phase, while the neutral Vinpocetine product precipitates out as a solid. This intrinsic purification mechanism is key to achieving the reported 99% purity without the need for energy-intensive recrystallization or chromatographic separation, thereby enhancing the commercial scale-up of complex pharmaceutical intermediates.
How to Synthesize Vinpocetine Efficiently
Implementing this synthesis requires strict adherence to the stoichiometric ratios and temperature profiles outlined in the patent to ensure optimal conversion and safety. The process begins with the dissolution of Vincamine in toluene, followed by the controlled addition of reagents to manage the exothermic nature of the mesylation. Once the intermediate is secured, the elimination step demands anhydrous conditions to prevent the hydrolysis of the sodium ethoxide. The detailed standardized operating procedures, including specific molar equivalents and safety protocols for handling methanesulfonyl chloride, are critical for reproducibility. For technical teams looking to adopt this route, the following guide outlines the essential operational parameters derived directly from the patent examples.
- Activate the hydroxyl group of Vincamine by reacting with methanesulfonyl chloride (MsCl) and triethylamine in toluene at 0-25°C to form the mesylate intermediate.
- Isolate the intermediate by pH adjustment to 12 using potassium carbonate, followed by filtration and vacuum drying.
- Perform elimination by reacting the intermediate with sodium ethoxide in absolute ethanol at 80°C for 12 hours, followed by acid-base workup to obtain pure Vinpocetine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this synthesis method offers tangible strategic benefits beyond mere chemical elegance. The primary advantage is the substantial simplification of the raw material portfolio. By eliminating the need for exotic catalysts like fluorinated nitrobenzenes or titanium alkoxides, the process relies on commodity chemicals that are readily available from multiple global suppliers. This diversification of the supply base significantly mitigates the risk of production stoppages due to single-source dependency. Furthermore, the use of toluene and ethanol—solvents with well-established recovery and recycling infrastructures—allows for a more circular manufacturing model. This reduces the volume of hazardous waste requiring disposal, directly translating to lower environmental compliance costs and a smaller carbon footprint for the final product.
- Cost Reduction in Manufacturing: The economic viability of this route is driven by its high atom economy and simplified downstream processing. The patent reports a remarkable 95% yield for the intermediate step and a total recovery of 71% for the final product, figures that are exceptionally high for alkaloid derivatization. Because the product precipitates directly upon pH adjustment, the need for expensive column chromatography or multiple recrystallizations is obviated. This reduction in unit operations lowers both labor costs and energy consumption, as there is no need for prolonged solvent evaporation cycles or complex purification trains. The elimination of toxic solvents also reduces the capital expenditure required for specialized containment and ventilation systems, offering a clear path to cost reduction in pharmaceutical intermediates manufacturing.
- Enhanced Supply Chain Reliability: The robustness of this chemical route ensures consistent output quality, which is vital for maintaining long-term contracts with downstream API manufacturers. The process tolerances are wide enough to accommodate standard industrial equipment, reducing the likelihood of batch failures due to minor fluctuations in temperature or mixing efficiency. Moreover, the starting material, Vincamine, is a naturally occurring alkaloid with a stable supply chain derived from plant extraction. By coupling a stable natural feedstock with a reliable synthetic sequence, manufacturers can offer reducing lead time for high-purity pharmaceutical intermediates, ensuring that customers receive their materials on schedule without the delays often associated with troubleshooting finicky synthetic routes.
- Scalability and Environmental Compliance: From a scale-up perspective, the reaction conditions are inherently safe and manageable. The exothermic mesylation is controlled by slow addition and cooling, while the elimination step proceeds at a moderate 80°C, well within the capabilities of standard glass-lined or stainless steel reactors. The absence of cryogenic conditions or high-pressure hydrogenation steps simplifies the engineering requirements for plant expansion. Additionally, the waste stream is significantly cleaner; the primary byproducts are triethylamine hydrochloride and sodium methanesulfonate, which are far easier to treat than the heavy metal or nitro-aromatic wastes generated by alternative methods. This alignment with green chemistry principles facilitates smoother regulatory approvals and supports the sustainability goals of modern pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Vinpocetine synthesis technology. These answers are derived directly from the experimental data and claims presented in patent CN102702191A, providing a factual basis for decision-making. Understanding these details is crucial for evaluating the feasibility of integrating this route into existing production lines or for qualifying new suppliers.
Q: What are the critical advantages of this Vinpocetine synthesis method over traditional routes?
A: This method avoids the use of toxic solvents like acetonitrile and rare catalysts such as 2-fluoro-1,3,5-trinitrobenzene found in older patents. It utilizes common reagents (MsCl, NaOEt) and achieves a high total yield of 71% with 99% purity without recrystallization.
Q: How does this process ensure high purity suitable for pharmaceutical applications?
A: The process employs a precise pH control strategy during workup. By regulating the pH to 12 with saturated potassium carbonate or water, impurities remain in solution while the product precipitates as a solid, ensuring the final product meets EP7.0 standards.
Q: Is this synthesis route scalable for industrial production?
A: Yes, the route is highly scalable. It uses standard industrial solvents like toluene and ethanol and operates at mild temperatures (ice bath to 80°C). The simple filtration steps and lack of complex chromatographic purification make it ideal for large-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Vinpocetine Supplier
The synthesis method described in CN102702191A exemplifies the type of process innovation that drives value in the modern pharmaceutical supply chain. At NINGBO INNO PHARMCHEM, we specialize in translating such patented laboratory methodologies into robust, commercial-scale manufacturing processes. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the high yields and purity levels demonstrated in the lab are faithfully reproduced in the plant. We operate with stringent purity specifications and maintain rigorous QC labs equipped to verify that every batch of Vinpocetine meets the demanding standards of the global market, including EP and USP monographs.
We invite procurement leaders and R&D directors to engage with us to explore how this optimized synthesis route can benefit your specific supply chain requirements. By partnering with us, you gain access to a Customized Cost-Saving Analysis that quantifies the potential efficiencies of switching to this greener, high-yield method. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments tailored to your project timelines and volume needs.
