Optimizing Vinpocetine Production: A Technical Analysis of High-Yield Semi-Synthetic Routes for Commercial Scale-up
Optimizing Vinpocetine Production: A Technical Analysis of High-Yield Semi-Synthetic Routes for Commercial Scale-up
The pharmaceutical landscape for cerebrovascular therapeutics continues to demand robust, scalable, and environmentally compliant manufacturing processes for key active ingredients. Patent CN102702191A presents a refined semi-synthetic methodology for the production of Vinpocetine, a critical derivative of the Apocynaceae periwinkle alkaloid vincamine. This technical disclosure addresses longstanding challenges in the industry by optimizing the dehydration and structural modification steps required to convert vincamine into the bioactive ethyl apovincaminate. The strategic implementation of methanesulfonyl chloride activation followed by base-catalyzed elimination represents a significant departure from earlier, more hazardous protocols. By leveraging this specific pathway, manufacturers can achieve superior control over impurity profiles while maintaining high throughput efficiency. 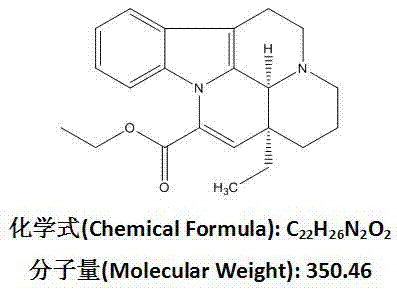
For R&D directors and procurement specialists evaluating potential partners, understanding the nuances of this synthetic route is paramount. The process outlined in the patent not only simplifies the operational workflow but also aligns with modern green chemistry principles by reducing solvent toxicity and energy consumption. As a reliable vinpocetine supplier, analyzing such proprietary methods allows us to benchmark our own production capabilities against the state-of-the-art, ensuring that we deliver materials that meet the stringent quality expectations of global regulatory bodies. The following analysis dissects the mechanistic advantages and commercial implications of this specific synthetic strategy.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Vinpocetine has been plagued by significant technical and environmental hurdles that impact both cost and supply chain stability. Early methodologies, such as those described in the Mondelo patent, relied heavily on the use of acetonitrile as a primary solvent and 2-fluoro-1,3,5-trinitrobenzene as a catalyst. While these methods could achieve reasonable yields, the reliance on acetonitrile introduces severe toxicity concerns and complicates waste management protocols, thereby increasing the overall cost reduction in pharmaceutical manufacturing efforts. Furthermore, the catalysts employed in these traditional routes are often rare or expensive, creating a bottleneck for large-scale production and introducing volatility into the raw material supply chain. Other approaches utilizing Titanium tetraethoxide for transesterification have shown promise but frequently suffer from low total recovery rates and complex workup procedures that hinder commercial viability.
The Novel Approach
In stark contrast to these legacy methods, the novel approach detailed in CN102702191A utilizes a streamlined two-step sequence that prioritizes reagent availability and operational simplicity. The core innovation lies in the initial activation of the vincamine hydroxyl group using methanesulfonyl chloride (MsCl) in the presence of triethylamine within a toluene medium. This mesylation step proceeds under mild conditions, transitioning from an ice-water bath to room temperature, which minimizes thermal degradation of the sensitive alkaloid scaffold. 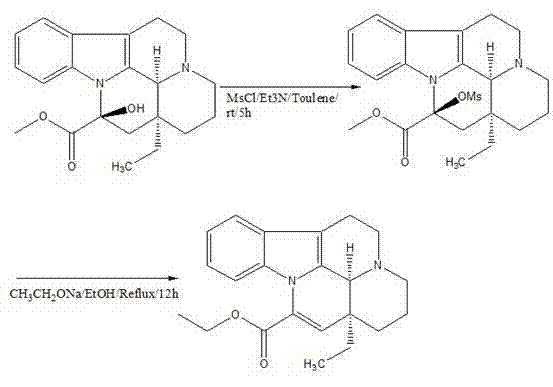
Following the formation of the mesylate intermediate, the subsequent elimination reaction employs sodium ethoxide in absolute ethanol, a reagent system that is both cost-effective and widely available in bulk quantities. This substitution of expensive transition metal catalysts with common organic bases drastically simplifies the downstream processing requirements. The result is a process that not only boasts a high total recovery rate of approximately 71% but also achieves a product purity of 99% without the need for energy-intensive recrystallization steps. This efficiency makes the route exceptionally attractive for the commercial scale-up of complex alkaloids, offering a clear pathway to consistent, high-quality output.
Mechanistic Insights into MsCl-Mediated Activation and Elimination
From a mechanistic perspective, the success of this synthesis hinges on the precise activation of the C-14 hydroxyl group of vincamine. In the first stage, the reaction between vincamine and methanesulfonyl chloride, facilitated by triethylamine as a proton scavenger, generates a mesylate ester. This transformation is critical because the hydroxyl group itself is a poor leaving group; converting it into a mesylate creates an excellent leaving group that primes the molecule for the subsequent elimination. The use of toluene as the solvent in this step is particularly advantageous due to its ability to dissolve the organic substrates effectively while allowing for easy removal via distillation post-reaction. 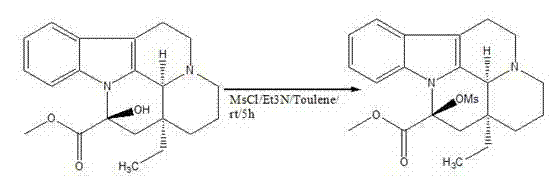
The second stage involves an E2-type elimination mechanism driven by the strong base, sodium ethoxide. The ethoxide ion abstracts a proton from the adjacent carbon, facilitating the expulsion of the mesylate group and the concurrent formation of the characteristic exocyclic double bond found in Vinpocetine. This concerted mechanism is highly stereoselective and minimizes the formation of regioisomers or other structural impurities that often plague acid-catalyzed dehydration methods. By controlling the reaction temperature at 80°C and managing the stoichiometry of the base, the process ensures that the elimination proceeds to completion without inducing unwanted side reactions on the indole nitrogen or the ester moiety. This level of mechanistic control is essential for maintaining the high-purity vinpocetine standards required for clinical applications.
How to Synthesize Vinpocetine Efficiently
The practical execution of this synthesis requires careful attention to reaction parameters, particularly temperature control during the exothermic mesylation phase and the maintenance of anhydrous conditions during the elimination step. The protocol dictates a slow addition of reagents to manage heat generation and ensure uniform mixing, which is vital for preventing localized hot spots that could degrade the product. Operators must also be diligent in the pH adjustment phases during workup, utilizing saturated potassium carbonate and hydrochloric acid to selectively precipitate the product while leaving impurities in the aqueous phase. For a detailed breakdown of the specific operational parameters, reagent ratios, and safety precautions necessary for replicating this high-yield process, please refer to the standardized synthesis guide below.
- Activate the hydroxyl group of Vincamine using Methanesulfonyl chloride (MsCl) and Triethylamine in Toluene to form the mesylate intermediate.
- Perform an elimination reaction by treating the intermediate with Sodium Ethoxide in absolute ethanol under reflux conditions.
- Isolate the final product through acid-base extraction and filtration, achieving high purity without recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the shift towards this specific synthetic route offers tangible benefits that extend beyond mere chemical yield. The elimination of rare earth or transition metal catalysts removes a significant variable from the sourcing equation, reducing exposure to price volatility in the metals market. Furthermore, the use of commodity chemicals like toluene, ethanol, and triethylamine ensures that raw material availability remains robust even during periods of global supply chain disruption. This reliability is crucial for maintaining continuous production schedules and meeting the rigorous delivery timelines expected by downstream pharmaceutical clients.
- Cost Reduction in Manufacturing: The replacement of expensive catalysts such as Titanium tetraethoxide with inexpensive bases like sodium ethoxide results in a substantial decrease in direct material costs. Additionally, the simplified workup procedure, which avoids complex chromatographic purification or multiple recrystallizations, significantly lowers utility consumption and labor hours per batch. The ability to recover solvents like toluene and ethanol further enhances the economic efficiency of the process, driving down the overall cost of goods sold without compromising on quality standards.
- Enhanced Supply Chain Reliability: By relying on reagents that are produced on a massive industrial scale globally, the risk of supply interruption is minimized. Unlike specialized catalysts that may have limited suppliers and long lead times, the chemicals required for this MsCl-mediated route are readily accessible from multiple vendors. This diversification of the supply base provides a strategic buffer against geopolitical or logistical shocks, ensuring that production of this critical cerebrovascular agent can continue uninterrupted.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, utilizing standard reactor configurations and avoiding hazardous reagents that require specialized containment. The reduction in toxic solvent usage, specifically the move away from acetonitrile, aligns with increasingly strict environmental regulations regarding volatile organic compound (VOC) emissions and waste disposal. This compliance reduces the regulatory burden on manufacturing sites and facilitates smoother audits, making the facility a more attractive partner for multinational corporations with strict sustainability mandates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Vinpocetine using this optimized semi-synthetic pathway. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a transparent view of the technology's capabilities. Understanding these details helps stakeholders make informed decisions regarding vendor qualification and process adoption.
Q: Why is the MsCl/Et3N system preferred over traditional acid catalysis for Vinpocetine synthesis?
A: The MsCl/Et3N system offers milder reaction conditions and avoids the use of toxic solvents like acetonitrile or expensive catalysts like Titanium tetraethoxide, leading to better environmental compliance and lower raw material costs.
Q: How does this process ensure high purity without recrystallization?
A: The specific control of reaction temperature and the use of a selective elimination step minimize side reactions and impurity formation, allowing the product to reach 99% purity directly after filtration and drying.
Q: Is this synthetic route suitable for large-scale industrial production?
A: Yes, the process utilizes common industrial solvents like Toluene and Ethanol and involves simple unit operations such as filtration and distillation, making it highly scalable and robust for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Vinpocetine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the theoretical advantages of a patent must be translated into consistent, commercial reality. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the efficiencies described in CN102702191A are fully realized in our manufacturing plants. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to verify that every batch of Vinpocetine meets or exceeds the 99% purity benchmark without the need for additional processing. Our commitment to quality assurance guarantees that the material you receive is perfectly suited for formulation into final dosage forms.
We invite potential partners to engage with our technical procurement team to discuss how our optimized manufacturing capabilities can support your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits of our production methods compared to traditional sources. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to validate our capacity to serve as a long-term, strategic partner in your supply chain.
