Scalable Synthesis of High-Purity Tetraethyl Ranelate for Commercial API Production
Scalable Synthesis of High-Purity Tetraethyl Ranelate for Commercial API Production
The pharmaceutical industry's demand for high-quality osteoporosis treatments has driven intense scrutiny on the supply chain of Strontium Ranelate precursors. Patent CN103319454B introduces a groundbreaking preparation method for tetraethyl ranelate, a critical intermediate that directly influences the purity and efficacy of the final active pharmaceutical ingredient. This technology addresses long-standing bottlenecks in heterocyclic synthesis by utilizing ammonium sulfide as a novel solubility promoter, transforming a traditionally sluggish and waste-intensive process into a streamlined, high-yield operation. For R&D directors and procurement strategists, this patent represents a pivotal shift towards more sustainable and cost-effective manufacturing of complex thiophene derivatives. By enabling reaction times to be foreshortened drastically while achieving purity levels exceeding 99.6%, this methodology sets a new benchmark for reliable pharmaceutical intermediates supplier capabilities in the global market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of tetraethyl ranelate has been plagued by inefficient protocols that compromise both economic viability and environmental safety. Early literature, such as Chem Ber (1966), documented the use of morpholine as an acid-binding agent, a method that resulted in a meager reaction yield of only 42.0% after more than 3 hours of processing. The toxicity of morpholine presents severe occupational health risks, making it increasingly untenable for modern GMP facilities. Subsequent attempts to replace morpholine with diethylamine in aqueous systems, as seen in Journal of Chemical Technology and Biotechnology (1990), improved yields to 80.0% but introduced massive volumes of alkaline wastewater due to the need for extensive washing to neutralize the amine. Furthermore, the use of water as a solvent creates a biphasic system where organic reactants are poorly soluble, leading to uneven reaction rates and the encapsulation of inorganic salts within the product crystal lattice, which drastically reduces quality.
The Novel Approach
The methodology disclosed in CN103319454B fundamentally reengineers the reaction environment to overcome these solubility and kinetic barriers. By introducing ammonium sulfide as a solubility promoter in an ethanol medium, the process ensures that sulfur is fully dissolved, creating a true homogeneous phase that facilitates rapid and uniform cyclization. This innovation reduces the reaction time for Intermediate I formation from the conventional 3 to 5 hours down to merely 1.0 hour, representing a dramatic increase in throughput capacity. Additionally, the subsequent alkylation step utilizes inexpensive iodide salts (potassium iodide or sodium iodide) instead of costly and environmentally hazardous quaternary ammonium salts like Adogen 464. This switch not only lowers raw material costs but also simplifies the purification process, as the iodide catalysts are easily removed during filtration, eliminating the need for complex extraction procedures that generate significant organic waste.
Mechanistic Insights into Ammonium Sulfide-Promoted Cyclization
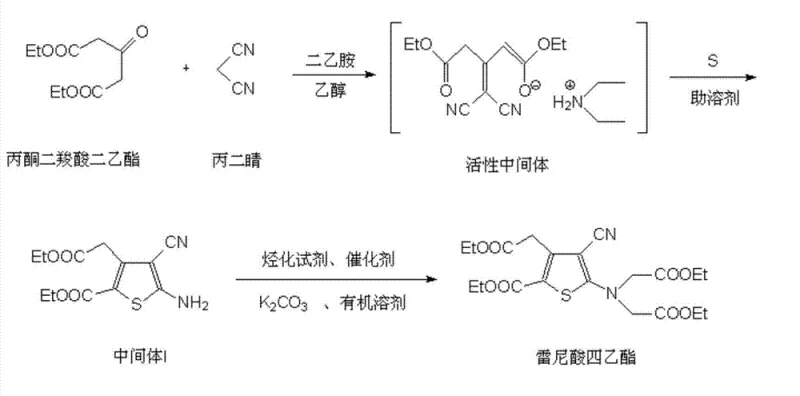
The core chemical breakthrough lies in the manipulation of sulfur solubility within the alcoholic solvent system. In traditional ethanol-based syntheses, elemental sulfur has limited solubility, which restricts the rate of the cyclization reaction with the active intermediate formed from acetonedicarboxylic acid diethyl ester and malononitrile. The addition of ammonium sulfide acts as a powerful solubility promoter, likely forming polysulfide species that are highly soluble in ethanol. This ensures that the sulfurating agent is molecularly dispersed throughout the reaction matrix, allowing for immediate and efficient attack on the active methylene groups of the intermediate. Consequently, the activation energy for the ring-closing step is effectively lowered, driving the equilibrium towards the desired thiophene product with exceptional speed and selectivity. This homogeneous mechanism prevents the formation of localized hot spots or concentration gradients that typically lead to side reactions and polymeric byproducts.
Following the formation of the thiophene ring, the purity of the final tetraethyl ranelate is governed by the efficiency of the N-alkylation step. The patent specifies the use of potassium iodide or sodium iodide as catalysts, which function through a halogen exchange mechanism. The iodide ion, being a superior nucleophile and leaving group compared to chloride or bromide, reacts rapidly with the ethyl haloacetate alkylating agent to generate iodoacetyl ethyl acetate in situ. This iodo-species is significantly more electrophilic, facilitating a swift nucleophilic attack by the primary amine nitrogen of Intermediate I. This catalytic cycle minimizes the residence time of the reactants at elevated temperatures, thereby suppressing thermal degradation and the formation of dialkylated impurities. The result is a product profile with single impurity levels controlled below 0.1%, meeting the stringent specifications required for downstream conversion into Strontium Ranelate without the need for repeated recrystallization.
How to Synthesize Tetraethyl Ranelate Efficiently
The operational protocol for this synthesis is designed for seamless integration into existing multipurpose reactor setups, requiring minimal modification to standard equipment. The process begins with the condensation of acetonedicarboxylic acid diethyl ester and malononitrile in ethanol, followed by the critical addition of ammonium sulfide and sulfur for the cyclization step. Detailed control of temperature and addition rates is essential to maintain the homogeneous phase and maximize yield. Once Intermediate I is isolated, the subsequent alkylation in acetone with iodide catalysis proceeds rapidly under mild reflux conditions. For a comprehensive breakdown of the specific mass ratios, temperature profiles, and workup procedures, please refer to the standardized synthesis guide below.
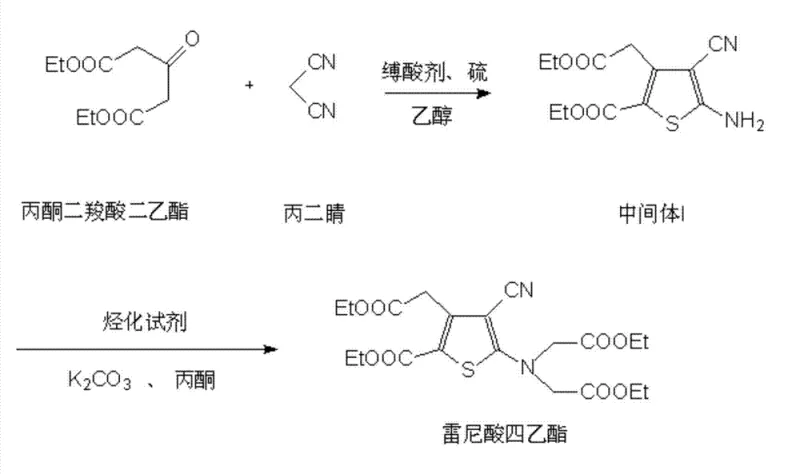
- React acetonedicarboxylic acid diethyl ester with malononitrile in ethanol using diethylamine as an acid-binding agent to form an active intermediate.
- Add ammonium sulfide as a solubility promoter and sulfur to the mixture, then reflux to cyclize into 5-amino-4-cyano-3-(2-ethoxyl-2-carboxymethyl)-thiophene-2-ethyl formate (Intermediate I).
- Alkylate Intermediate I with ethyl bromoacetate in acetone using potassium carbonate and a catalytic amount of potassium iodide or sodium iodide to yield tetraethyl ranelate.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, this patented process offers substantial advantages in terms of cost structure and supply chain resilience. The elimination of expensive phase-transfer catalysts and toxic amines directly reduces the bill of materials, while the drastic reduction in reaction time increases asset utilization rates, allowing manufacturers to produce significantly more volume within the same timeframe. The simplified workup procedure, which avoids extensive aqueous washing and complex extractions, leads to a marked decrease in solvent consumption and waste disposal costs. For procurement managers, this translates into a more stable pricing model for high-purity pharmaceutical intermediates, insulated from the volatility associated with specialized reagent markets. Furthermore, the robustness of the chemistry ensures consistent batch-to-batch quality, reducing the risk of production delays caused by out-of-specification materials.
- Cost Reduction in Manufacturing: The replacement of costly quaternary ammonium catalysts and toxic morpholine with commodity chemicals like ammonium sulfide and potassium iodide drives down raw material expenses significantly. The shortened reaction cycles reduce energy consumption for heating and stirring, while the high yield minimizes the loss of valuable starting materials. Additionally, the avoidance of complex purification steps lowers labor and utility costs associated with waste treatment and solvent recovery, resulting in a leaner and more profitable manufacturing process for cost reduction in API manufacturing.
- Enhanced Supply Chain Reliability: The reagents utilized in this novel pathway, such as ethanol, acetone, and simple inorganic salts, are globally available commodities with stable supply chains, unlike specialized ionic liquids or crown ethers. This ubiquity ensures that production schedules are not disrupted by shortages of niche catalysts. The high purity of the intermediate (>99.6%) reduces the need for reprocessing or sourcing alternative suppliers to meet quality standards, thereby securing a continuous flow of materials for downstream Strontium Ranelate production and reducing lead time for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: The homogeneous nature of the reaction and the absence of biphasic wastewater generation make this process inherently scalable from pilot plant to commercial tonnage. The reduction in alkaline wastewater and organic solvent waste aligns with increasingly strict environmental regulations, mitigating the risk of regulatory fines or shutdowns. This environmental efficiency supports the commercial scale-up of complex pharmaceutical intermediates, ensuring long-term operational continuity and enhancing the corporate sustainability profile of the manufacturing facility.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, offering clarity on the practical benefits of the ammonium sulfide promotion and iodide catalysis. Understanding these nuances is critical for technical teams evaluating the feasibility of technology transfer and for procurement specialists assessing the total cost of ownership.
Q: What is the key innovation in this tetraethyl ranelate synthesis method?
A: The key innovation is the use of ammonium sulfide as a solubility promoter for sulfur in ethanol, which creates a homogeneous reaction phase, significantly reducing reaction time from 3-5 hours to 1.0 hour and improving yield.
Q: Why are potassium iodide or sodium iodide preferred as catalysts?
A: Potassium iodide and sodium iodide act as efficient nucleophilic catalysts that accelerate the alkylation step. They are inexpensive, easily removed during filtration, and help achieve product purity greater than 99.6% with single impurities below 0.1%.
Q: How does this method improve environmental compliance compared to prior art?
A: This method avoids the use of toxic morpholine and eliminates the need for biphasic water-organic systems that generate large volumes of alkaline wastewater. The homogeneous ethanol system allows for simpler filtration and reduced solvent waste.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tetraethyl Ranelate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the quality of the final API is inextricably linked to the purity of its precursors. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN103319454B are fully realized in industrial practice. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including the detection of trace impurities at the 0.1% level. We are committed to delivering high-purity pharmaceutical intermediates that meet the exacting standards of global regulatory bodies, providing our partners with a secure foundation for their drug development pipelines.
We invite forward-thinking pharmaceutical companies to collaborate with us to leverage this advanced synthesis technology for their Strontium Ranelate projects. Our technical team is prepared to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this optimized route can enhance your margin structure. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can accelerate your path to market with reliable, high-quality chemical solutions.
