Advanced Catalytic Strategies for Commercial Scale-Up of High-Purity Strontium Ranelate
Introduction to Advanced Strontium Ranelate Manufacturing
The global demand for effective osteoporosis treatments continues to drive the need for high-quality active pharmaceutical ingredients (APIs), with Strontium Ranelate standing out as a critical therapeutic agent. The preparation method disclosed in patent CN103319455B represents a significant technological leap in the synthesis of this compound, addressing long-standing challenges related to purity, yield, and environmental impact. Traditional manufacturing routes have often struggled with low conversion rates, toxic reagents, and difficult purification processes that compromise the final drug substance's quality. This innovative approach introduces a refined catalytic system and a novel purification strategy that ensures the production of Strontium Ranelate with purity levels exceeding 99.6% and single impurities controlled below 0.05%. For pharmaceutical manufacturers and procurement specialists, understanding the nuances of this patented technology is essential for securing a reliable supply chain of high-purity pharmaceutical intermediates. The method not only enhances the chemical integrity of the final product but also streamlines the production workflow, offering substantial advantages in terms of operational efficiency and regulatory compliance for bulk drug manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
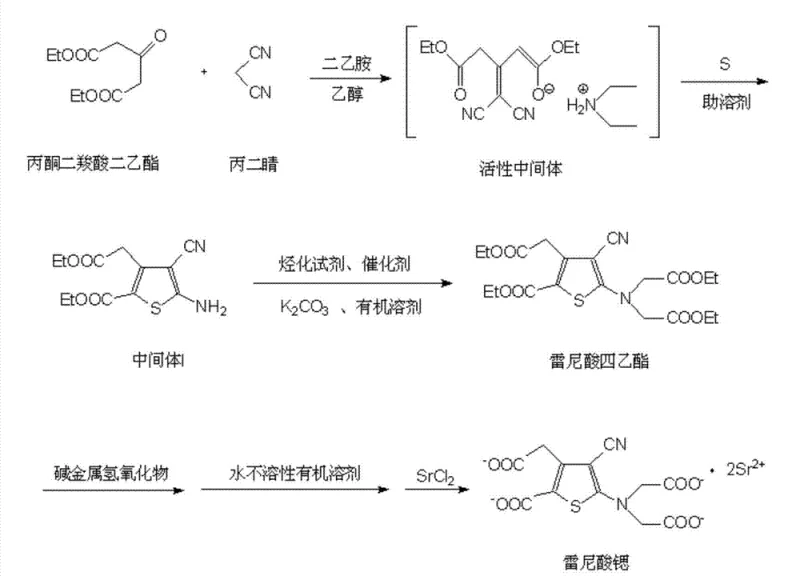
Historical methods for synthesizing Strontium Ranelate and its key precursor, tetraethyl ranelate, have been plagued by significant inefficiencies and environmental hazards. Early protocols often relied on morpholine as an acid-binding agent, which, while functional, posed severe toxicity risks and resulted in reaction yields as low as 42.0%, necessitating extensive downstream processing. Subsequent improvements utilizing diethylamine managed to increase yields to around 80.0%, but this came at the cost of generating massive volumes of alkaline wastewater due to the need for extensive washing to neutralize the amine. Furthermore, the alkylation step to form tetraethyl ranelate traditionally employed expensive and environmentally persistent catalysts such as C8-C10 type quaternary ammonium salts, crown ethers, or ionic liquids. These catalysts not only increased raw material costs but also introduced steric hindrance that limited catalytic efficiency, often requiring reaction times exceeding 15 hours. Additionally, purification of the intermediate sodium salt typically involved heating and concentrating organic solvents, a process prone to inducing thermal degradation and the formation of decarboxylation impurities, ultimately capping the purity of the final API at unacceptable levels for modern pharmacopeial standards.
The Novel Approach
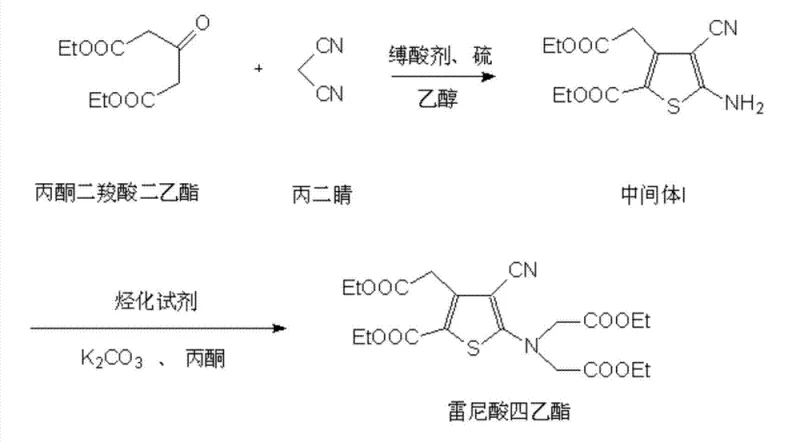
The methodology outlined in CN103319455B fundamentally reengineers the synthesis pathway to overcome these bottlenecks through strategic chemical modifications. In the initial cyclization step, the introduction of ammonium sulfide as a solubility promoter transforms the reaction dynamics by ensuring sulfur remains dissolved in the ethanol medium, facilitating a homogeneous reaction environment. This simple yet profound change reduces the reaction time for Intermediate I formation from several hours to just 1.0 hour while simultaneously boosting purity to over 99.6%. For the subsequent alkylation, the process replaces costly phase transfer catalysts with inexpensive potassium iodide or sodium iodide. These iodide salts act as potent nucleophilic catalysts, accelerating the substitution reaction to completion within 1.5 to 2.0 hours and achieving superior purity profiles without the ecological burden of quaternary ammonium residues. Finally, the purification stage utilizes a water-insoluble organic solvent, specifically methylene dichloride, to wash the aqueous sodium salt solution. This technique effectively extracts organic impurities without causing the emulsification common with other solvents, thereby eliminating the need for energy-intensive solvent concentration steps that typically degrade product quality.
Mechanistic Insights into Iodide-Catalyzed Alkylation and Solubility Promotion
The core innovation of this synthesis lies in the mechanistic role of the additives used to promote reaction kinetics and phase homogeneity. In the formation of Intermediate I, sulfur typically exhibits poor solubility in ethanol, leading to heterogeneous reaction conditions that slow down the cyclization of acetonedicarboxylic acid diethyl ester and malononitrile. The addition of ammonium sulfide leverages its high solubility in ethanol to act as a carrier, effectively increasing the local concentration of reactive sulfur species in the liquid phase. This homogenization ensures that the reactants interact more frequently and efficiently, driving the equilibrium towards the desired thiophene ring formation. The result is a drastic reduction in side reactions and a cleaner crude product that requires minimal recrystallization, directly impacting the overall mass balance of the process. This mechanism highlights the importance of phase management in heterocyclic synthesis, where solubility promoters can serve as a cost-effective alternative to changing bulk solvents or increasing reaction temperatures.
In the alkylation step, the choice of potassium iodide or sodium iodide as a catalyst is driven by the unique nucleophilic properties of the iodide ion. Unlike bulky quaternary ammonium salts which rely on phase transfer mechanisms that can be sterically hindered, iodide ions are small, highly nucleophilic, and excellent leaving groups. During the reaction with ethyl bromoacetate, the iodide ion displaces the bromide to form an intermediate iodoacetate species in situ, which is significantly more reactive towards the nucleophilic attack by the amino group of Intermediate I. This "in situ activation" lowers the activation energy of the substitution reaction, allowing it to proceed rapidly at moderate reflux temperatures. Furthermore, because the iodide catalyst is soluble in the alcoholic recrystallization solvent, it is easily removed during the filtration of the final tetraethyl ranelate, ensuring that no heavy metal or toxic organic residues remain in the product. This mechanistic elegance ensures that the process is not only faster but also inherently cleaner, aligning with the stringent impurity control requirements of modern API manufacturing.
How to Synthesize High-Purity Strontium Ranelate Efficiently
The implementation of this patented process requires precise control over reaction parameters to maximize the benefits of the novel catalytic system. The synthesis is divided into three distinct stages: the formation of the thiophene intermediate, the alkylation to the tetraethyl ester, and the final hydrolysis and salt formation. Each step has been optimized to minimize waste and maximize throughput, making it highly suitable for industrial scale-up. Operators must pay close attention to the stoichiometry of the solubility promoter and the temperature controls during hydrolysis to prevent the formation of thermally labile decarboxylation impurities. The following guide outlines the standardized operational framework derived from the patent examples, providing a roadmap for technical teams to replicate these high-yield results in a production environment.
- Cyclization of acetonedicarboxylic acid diethyl ester with malononitrile and sulfur using ammonium sulfide as a solubility promoter to form Intermediate I.
- Alkylation of Intermediate I with ethyl bromoacetate using potassium iodide or sodium iodide as a catalyst to generate tetraethyl ranelate.
- Hydrolysis of tetraethyl ranelate followed by washing with water-insoluble organic solvents and strontium salt formation to obtain the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthesis route offers compelling economic and logistical benefits that extend beyond simple yield improvements. The elimination of expensive and specialized catalysts such as crown ethers, ionic liquids, and long-chain quaternary ammonium salts significantly reduces the raw material cost base. These traditional catalysts not only carry a high price tag but also introduce complexity into the supply chain due to their specialized nature and potential regulatory restrictions regarding environmental toxicity. By substituting them with commodity chemicals like potassium iodide and ammonium sulfide, the process insulates the manufacturing cost from the volatility of niche chemical markets. Furthermore, the drastic reduction in reaction times—from over 15 hours to under 2 hours for the alkylation step—translates directly into increased reactor turnover rates. This enhanced throughput allows manufacturers to meet large volume demands with existing infrastructure, effectively expanding capacity without capital expenditure on new equipment.
- Cost Reduction in Manufacturing: The shift away from proprietary phase transfer catalysts to inexpensive inorganic salts drastically lowers the variable cost per kilogram of the API. Additionally, the simplified purification process, which avoids energy-intensive solvent concentration and distillation steps, reduces utility consumption and waste disposal costs. The high purity of the intermediate I means fewer resources are spent on recycling off-spec material, further optimizing the overall production budget.
- Enhanced Supply Chain Reliability: By relying on widely available commodity reagents rather than specialized catalysts, the risk of supply disruption is minimized. The robustness of the reaction conditions, particularly the tolerance for standard solvents like ethanol and acetone, ensures that production can continue even if specific grades of reagents are temporarily unavailable. The shortened cycle times also provide greater flexibility in scheduling, allowing for quicker response to urgent orders from downstream pharmaceutical partners.
- Scalability and Environmental Compliance: The process generates significantly less alkaline wastewater compared to diethylamine-based methods, reducing the burden on effluent treatment plants. The avoidance of toxic morpholine and persistent quaternary ammonium compounds simplifies environmental compliance and reduces the risk of regulatory penalties. The use of methylene dichloride for washing, while requiring careful handling, prevents emulsification and allows for efficient solvent recovery, supporting a more sustainable and circular manufacturing model.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this high-purity synthesis route. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, offering clarity on how this method outperforms legacy technologies. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this process for commercial production.
Q: How does the use of ammonium sulfide improve the synthesis of Intermediate I?
A: Ammonium sulfide acts as a solubility promoter, significantly increasing the solubility of sulfur in ethanol. This creates a homogeneous reaction phase, reducing reaction time from 3-5 hours to approximately 1 hour and improving yield and purity by preventing the encapsulation of inorganic salts.
Q: Why are potassium iodide or sodium iodide preferred over quaternary ammonium salts for alkylation?
A: Iodide ions act as strong nucleophiles and leaving groups, accelerating the substitution reaction significantly compared to bulky quaternary ammonium salts. This reduces reaction time, eliminates the environmental risks associated with long-chain quaternary salts, and achieves purity levels exceeding 99.6% without expensive catalysts.
Q: What is the advantage of using methylene dichloride for washing during hydrolysis?
A: Using water-insoluble organic solvents like methylene dichloride effectively removes organic impurities from the aqueous sodium salt solution without causing emulsification. This avoids the need for heating and concentrating organic solvents, thereby reducing thermal degradation of the product and minimizing waste generation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Strontium Ranelate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the intricate balance of catalysis and purification described in this patent is maintained at every batch size. We are committed to delivering products that meet stringent purity specifications, leveraging our rigorous QC labs to verify that every shipment of Strontium Ranelate adheres to the highest pharmacopeial standards. Our facility is equipped to handle the specific solvent systems and temperature controls required for this advanced synthesis, guaranteeing consistency and reliability for your supply chain.
We invite you to collaborate with us to optimize your sourcing strategy for this critical osteoporosis medication. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how our implementation of this patented route can reduce your total cost of ownership. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to chemical excellence can support your long-term business goals.
