Industrial Scale Synthesis of 5-Mercapto Tetrazole Acetic Acid for Advanced Pharmaceutical Applications
The pharmaceutical industry continuously demands more efficient and safer pathways for producing critical beta-lactam intermediates, particularly those required for cephalosporin antibiotics like Ceforanide. A significant technological breakthrough in this domain is documented in patent CN107805225B, which details a novel preparation method for 5-mercapto tetrazole acetic acid and its corresponding sodium salts. This innovation addresses long-standing challenges regarding toxicity and yield that have plagued previous manufacturing standards. By shifting away from hazardous alkylating agents and embracing a phase-transfer catalytic system, this technology offers a robust foundation for reliable pharmaceutical intermediate supplier networks seeking to optimize their supply chains. The strategic implementation of this chemistry not only enhances process safety but also aligns with modern environmental compliance standards required by top-tier global drug manufacturers.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 5-mercapto tetrazole acetic acid has relied heavily on the methodology outlined in US4782012, which necessitates the use of glycine as a starting material followed by methylation. 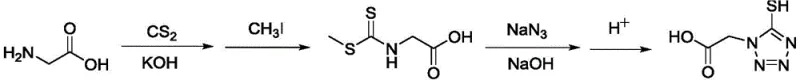 The critical bottleneck in this traditional pathway is the obligatory use of methyl iodide, a reagent known for its high toxicity, carcinogenic potential, and stringent storage requirements such as protection from light. From a procurement and operational standpoint, handling methyl iodide introduces significant overhead costs related to specialized containment, personal protective equipment, and hazardous waste disposal. Furthermore, the conventional route often suffers from inconsistent reaction yields and complex purification steps required to remove residual heavy metals and toxic byproducts. These factors collectively create a fragile supply chain vulnerable to regulatory scrutiny and production delays, making it increasingly unsuitable for the rigorous demands of modern high-purity API intermediate manufacturing.
The critical bottleneck in this traditional pathway is the obligatory use of methyl iodide, a reagent known for its high toxicity, carcinogenic potential, and stringent storage requirements such as protection from light. From a procurement and operational standpoint, handling methyl iodide introduces significant overhead costs related to specialized containment, personal protective equipment, and hazardous waste disposal. Furthermore, the conventional route often suffers from inconsistent reaction yields and complex purification steps required to remove residual heavy metals and toxic byproducts. These factors collectively create a fragile supply chain vulnerable to regulatory scrutiny and production delays, making it increasingly unsuitable for the rigorous demands of modern high-purity API intermediate manufacturing.
The Novel Approach
In stark contrast, the innovative route presented in CN107805225B utilizes isothiocyanato ethyl acetate reacting directly with sodium azide in the presence of a phase transfer catalyst. 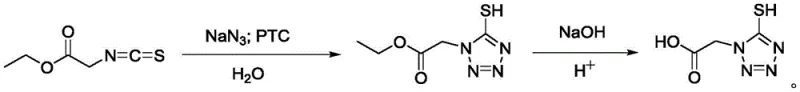 This fundamental shift in synthetic strategy eliminates the need for toxic methylating agents entirely, replacing them with safer, more commercially available raw materials. The process operates effectively in an aqueous environment facilitated by benzyltrimethyl ammonium chloride, which significantly reduces the reliance on volatile organic solvents during the reaction phase. By streamlining the molecular construction of the tetrazole ring, this approach not only mitigates safety risks but also simplifies the downstream processing workflow. For procurement managers, this translates into cost reduction in pharmaceutical intermediate manufacturing by lowering raw material acquisition costs and minimizing the financial burden associated with hazardous waste management and regulatory compliance.
This fundamental shift in synthetic strategy eliminates the need for toxic methylating agents entirely, replacing them with safer, more commercially available raw materials. The process operates effectively in an aqueous environment facilitated by benzyltrimethyl ammonium chloride, which significantly reduces the reliance on volatile organic solvents during the reaction phase. By streamlining the molecular construction of the tetrazole ring, this approach not only mitigates safety risks but also simplifies the downstream processing workflow. For procurement managers, this translates into cost reduction in pharmaceutical intermediate manufacturing by lowering raw material acquisition costs and minimizing the financial burden associated with hazardous waste management and regulatory compliance.
Mechanistic Insights into Phase Transfer Catalyzed Tetrazole Cyclization
The core of this technological advancement lies in the efficient nucleophilic substitution and subsequent cyclization mechanisms driven by phase transfer catalysis (PTC). In this system, the azide ion, generated from sodium azide in the aqueous phase, is transported into the organic interface by the quaternary ammonium salt catalyst. This facilitates a rapid attack on the electrophilic carbon of the isothiocyanate group, initiating the formation of the tetrazole heterocycle. The precise control of reaction temperature between 70-80°C ensures optimal kinetic energy for cyclization while preventing the decomposition of the sensitive azide species. This mechanistic precision is crucial for R&D directors focused on purity and杂质谱 (impurity profiles), as it minimizes the formation of side products that typically arise from uncontrolled exothermic reactions in older methodologies. The result is a cleaner reaction matrix that requires less aggressive purification, thereby preserving the integrity of the final molecular structure.
Furthermore, the subsequent conversion to sodium salts demonstrates exceptional control over stoichiometry and crystallization dynamics. Whether producing the mono-sodium or disodium salt, the process allows for precise pH adjustment and temperature modulation to dictate the final salt form. 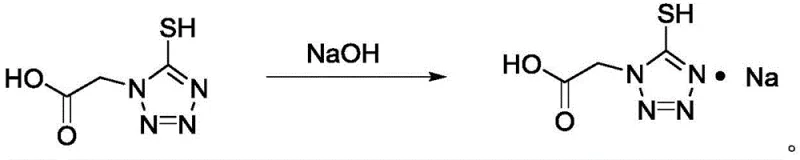 The ability to achieve single-step yields of up to 90% for these salt forms indicates a highly selective reaction environment where competing hydrolysis or degradation pathways are effectively suppressed. For quality assurance teams, this high selectivity means that the impurity profile is predictable and manageable, reducing the need for extensive chromatographic separations. The use of activated carbon treatment in the final purification step further ensures that any trace colored impurities or organic residuals are adsorbed, delivering a product that meets the stringent visual and chemical specifications required for parenteral antibiotic production.
The ability to achieve single-step yields of up to 90% for these salt forms indicates a highly selective reaction environment where competing hydrolysis or degradation pathways are effectively suppressed. For quality assurance teams, this high selectivity means that the impurity profile is predictable and manageable, reducing the need for extensive chromatographic separations. The use of activated carbon treatment in the final purification step further ensures that any trace colored impurities or organic residuals are adsorbed, delivering a product that meets the stringent visual and chemical specifications required for parenteral antibiotic production.
How to Synthesize 5-Mercapto Tetrazole Acetic Acid Efficiently
Implementing this synthesis route requires careful attention to the addition rates of reagents and the maintenance of an inert nitrogen atmosphere to ensure safety and reproducibility. The process begins with the preparation of the aqueous catalytic system, followed by the controlled dropwise addition of the isothiocyanate substrate to manage exotherms. Detailed standard operating procedures regarding temperature ramps and pH adjustments are critical for maximizing the 80% overall yield reported in the patent data. The following guide outlines the standardized synthesis steps optimized for commercial scale-up of complex pharmaceutical intermediates, ensuring that technical teams can replicate the high-efficiency results observed in the laboratory setting.
- Reaction Setup: Combine sodium azide, benzyltrimethyl ammonium chloride, and water in a reactor, heating to 50-70°C under nitrogen protection.
- Cyclization: Slowly add isothiocyanato ethyl acetate, maintain temperature at 70-80°C for 2-8 hours to form the tetrazole ring structure.
- Isolation and Purification: Adjust pH, extract with ethyl acetate, remove solvent, and recrystallize using activated carbon to obtain high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain heads and procurement managers, the adoption of this patented methodology offers profound strategic advantages beyond mere chemical efficiency. The elimination of methyl iodide removes a significant logistical bottleneck, as this substance often faces strict transportation regulations and availability fluctuations in the global chemical market. By substituting it with isothiocyanato ethyl acetate and sodium azide, manufacturers can secure a more stable and resilient supply of raw materials. This stability is essential for reducing lead time for high-purity pharmaceutical intermediates, ensuring that production schedules for downstream antibiotics like Ceforanide remain uninterrupted. Additionally, the simplified workup procedure involving ethyl acetate extraction and vacuum drying reduces the consumption of energy and solvents, contributing to a leaner manufacturing cost structure.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the removal of expensive and hazardous reagents. Without the need for methyl iodide, the direct material costs are significantly lowered, and the associated costs for specialized storage and safety monitoring are drastically reduced. Furthermore, the high yield of up to 80% for the acid and 90% for the salts means that less raw material is wasted per unit of output, enhancing the overall atom economy of the plant. This efficiency allows for substantial cost savings that can be passed down the supply chain or reinvested into further process optimization initiatives.
- Enhanced Supply Chain Reliability: Relying on commodity chemicals like sodium azide and water-soluble catalysts improves the robustness of the supply chain against market volatility. Unlike specialized alkylating agents that may have limited suppliers, the inputs for this new route are widely available from multiple global sources. This diversification of the supplier base mitigates the risk of single-source dependency and ensures continuous production capability even during regional disruptions. For supply chain planners, this reliability translates into more accurate forecasting and the ability to maintain optimal inventory levels without the fear of sudden raw material shortages.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, utilizing standard reactor configurations and straightforward separation techniques that are easily transferred from pilot to production scale. The reduced generation of hazardous waste, particularly the absence of iodide-containing byproducts, simplifies effluent treatment and lowers environmental compliance costs. This alignment with green chemistry principles not only satisfies regulatory requirements but also enhances the corporate sustainability profile of the manufacturer. Consequently, the commercial scale-up of complex pharmaceutical intermediates becomes a smoother, less risky endeavor that aligns with the long-term environmental goals of multinational corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the patent specifications and practical considerations for industrial application. Understanding these details is vital for stakeholders evaluating the feasibility of integrating this intermediate into their existing production portfolios. The answers provided reflect the consensus on safety, yield, and operational parameters established by the intellectual property documentation.
Q: How does this new synthesis route improve safety compared to traditional methods?
A: The novel process completely eliminates the use of methyl iodide, a toxic and light-sensitive reagent required in conventional US4782012 routes, significantly reducing occupational health hazards and storage costs.
Q: What is the expected yield improvement for industrial production?
A: According to patent data, the overall yield for the 5-mercaptotetrazole synthesis route reaches up to 80%, with single-step yields for sodium salts achieving up to 90%, offering substantial efficiency gains.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the method utilizes water as a primary medium with phase transfer catalysis, simplifying workup procedures and waste treatment, making it highly feasible for ton-scale production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Mercapto Tetrazole Acetic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the successful formulation of life-saving antibiotics. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory concept to industrial reality is seamless and efficient. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of 5-mercapto tetrazole acetic acid meets the exacting standards of the global pharmaceutical industry. Our infrastructure is designed to support the complex chemical transformations required by modern patents while adhering to the highest safety and environmental protocols.
We invite you to collaborate with us to leverage this advanced synthesis technology for your specific project needs. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements and logistical constraints. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. By partnering with us, you secure a supply chain partner dedicated to innovation, reliability, and mutual growth in the competitive landscape of fine chemical manufacturing.
