Advanced Green Synthesis of 5-Mercapto Tetrazole Acetic Acid for Industrial Pharmaceutical Applications
The pharmaceutical industry is constantly seeking robust, scalable, and environmentally benign pathways for the production of critical heterocyclic intermediates, particularly those serving as key building blocks for cephalosporin antibiotics. Patent CN107805225B introduces a transformative preparation method for 5-mercapto tetrazole acetic acid and its sodium salts, addressing long-standing safety and efficiency bottlenecks in the synthesis of this vital ceforanide intermediate. By shifting away from hazardous alkylating agents and embracing a phase-transfer catalyzed aqueous system, this technology offers a compelling value proposition for manufacturers aiming to optimize their supply chains while adhering to stricter environmental regulations. The core innovation lies in the strategic replacement of toxic methyl iodide with a safer cycloaddition protocol using sodium azide and isothiocyanato ethyl acetate, mediated by benzyltrimethyl ammonium chloride. This report provides a deep technical and commercial analysis of this patented route, highlighting its potential to redefine cost structures and operational safety standards in the fine chemical sector.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
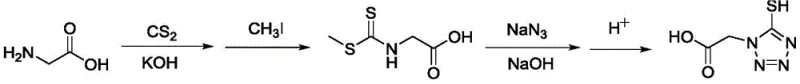
Historically, the industrial synthesis of 5-mercapto tetrazole acetic acid has been dominated by the methodology outlined in US4782012, a route that, while effective, carries significant operational burdens and safety liabilities. The conventional process relies on the initial reaction of glycine with carbon disulfide and potassium hydroxide, followed by a critical methylation step using methyl iodide to form the S-methyl intermediate. Methyl iodide is a notorious reagent in chemical manufacturing; it is highly toxic, volatile, and extremely sensitive to light, necessitating specialized storage facilities and rigorous containment protocols to prevent worker exposure and environmental release. Furthermore, the use of such potent alkylating agents often leads to complex impurity profiles that are difficult to purge, requiring extensive downstream purification which erodes overall yield. The reliance on organic solvents and hazardous reagents in this legacy route creates a high barrier to entry for safe scale-up, increasing both capital expenditure for safety infrastructure and operational expenditure for waste disposal and regulatory compliance.
The Novel Approach
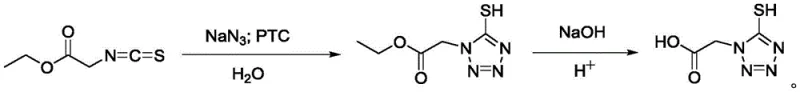
In stark contrast to the legacy methodology, the novel approach disclosed in CN107805225B leverages a direct [2+3] cycloaddition strategy that fundamentally redesigns the molecular construction of the tetrazole ring. Instead of building the ring and then methylating it, this process reacts isothiocyanato ethyl acetate directly with sodium azide in the presence of a phase transfer catalyst within an aqueous medium. This shift eliminates the need for methyl iodide entirely, removing a major toxicological hazard from the production floor. The reaction proceeds under mild thermal conditions, typically between 50°C and 80°C, which significantly reduces energy consumption compared to high-temperature organic refluxes. Moreover, the use of water as the primary reaction medium not only lowers solvent costs drastically but also enhances the thermal safety profile of the reactor, mitigating the risk of exothermic runaways. The resulting process is cleaner, safer, and inherently more scalable, offering a streamlined pathway from raw materials to the high-purity acid and its corresponding sodium salts required for antibiotic synthesis.
Mechanistic Insights into Phase Transfer Catalyzed Cycloaddition
The success of this novel synthesis hinges on the efficient operation of the phase transfer catalyst (PTC), specifically benzyltrimethyl ammonium chloride, which acts as a molecular shuttle between the aqueous and organic phases. In this system, the inorganic sodium azide resides primarily in the aqueous phase, while the organic substrate, isothiocyanato ethyl acetate, is largely immiscible with water. The quaternary ammonium cation of the PTC exchanges its chloride anion for the azide anion at the interface, forming a lipophilic ion pair that can dissolve into the organic phase or the interfacial region. Once in proximity to the isothiocyanate group, the azide ion undergoes a nucleophilic attack on the electrophilic carbon of the isothiocyanate, initiating the cyclization cascade that forms the tetrazole ring. This mechanism ensures that the reaction kinetics are not limited by the poor solubility of the inorganic salt in the organic substrate, allowing for high conversion rates even at moderate temperatures. The careful control of pH and temperature during the subsequent hydrolysis step ensures that the ethyl ester group is cleaved efficiently to yield the free carboxylic acid without degrading the sensitive tetrazole-thiol moiety.
From an impurity control perspective, this mechanistic pathway offers distinct advantages over the traditional alkylation route. By avoiding the use of methyl iodide, the process eliminates the formation of S-methylation byproducts and N-methylation impurities that are common in the legacy route and difficult to separate. The aqueous environment also facilitates the removal of inorganic salts and polar byproducts through simple washing or crystallization steps. The patent data indicates that the crude product obtained after extraction and solvent removal is of sufficient quality to undergo a straightforward recrystallization or activated carbon treatment to achieve high purity. This simplified purification train is critical for pharmaceutical intermediates, where strict limits on genotoxic impurities and heavy metals must be met. The robustness of the PTC system ensures consistent batch-to-batch reproducibility, a key factor for regulatory approval and commercial reliability.
How to Synthesize 5-Mercapto Tetrazole Acetic Acid Efficiently
The practical implementation of this synthesis involves a carefully sequenced addition of reagents to maintain reaction control and maximize yield. The process begins with the preparation of the catalytic system, followed by the controlled addition of the isothiocyanate substrate to manage exotherms. Detailed standardized operating procedures regarding stoichiometry, agitation speeds, and specific workup parameters are essential for transferring this laboratory-scale success to pilot and commercial plants. The following guide outlines the critical operational phases derived from the patent examples to ensure optimal outcomes.
- Charge a reactor with sodium azide, benzyltrimethyl ammonium chloride (PTC), and water, then heat to 50-70°C under nitrogen.
- Slowly add isothiocyanato ethyl acetate dropwise, maintain temperature at 70-80°C for 2-8 hours to complete the cycloaddition.
- Adjust pH to 10-12 with NaOH, heat again to hydrolyze the ester, then acidify with HCl to precipitate the crude product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented technology translates into tangible strategic benefits that extend beyond simple unit price reductions. The elimination of methyl iodide, a reagent subject to stringent regulatory controls and fluctuating market availability, de-risks the supply chain significantly. By switching to commodity chemicals like sodium azide and isothiocyanato ethyl acetate, manufacturers can secure more stable long-term contracts and reduce dependency on specialty chemical suppliers who may face production disruptions. Furthermore, the shift to an aqueous-based process dramatically lowers the volume of hazardous organic waste generated, leading to substantial cost savings in waste treatment and disposal fees. These operational efficiencies compound over time, creating a more resilient and cost-competitive manufacturing footprint for the final antibiotic product.
- Cost Reduction in Manufacturing: The economic argument for this new route is compelling, driven primarily by the removal of expensive and hazardous reagents. Methyl iodide is not only costly to purchase but also incurs high hidden costs related to specialized storage, handling equipment, and personal protective equipment (PPE) requirements. By replacing this with a catalytic amount of benzyltrimethyl ammonium chloride and inexpensive water as the solvent, the direct material costs are significantly reduced. Additionally, the high yields reported (up to 80% for the acid and 90% for the salts) mean that less raw material is wasted per kilogram of product, further enhancing the overall cost efficiency. The simplified workup procedure, which avoids complex distillation or chromatography steps, also reduces utility consumption and labor hours, contributing to a leaner cost structure.
- Enhanced Supply Chain Reliability: Supply chain continuity is paramount in the pharmaceutical industry, and this process enhances reliability by utilizing widely available starting materials. Sodium azide and isothiocyanato ethyl acetate are produced by multiple global suppliers, reducing the risk of single-source bottlenecks. In contrast, methyl iodide supply can be constrained by environmental regulations in certain regions, leading to volatility in lead times. The robustness of the aqueous reaction system also means that the process is less sensitive to minor variations in raw material quality or environmental conditions, ensuring consistent production schedules. This stability allows supply chain planners to forecast demand more accurately and maintain lower safety stock levels, freeing up working capital.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new challenges, but the aqueous nature of this synthesis makes it inherently scalable. Water has a high heat capacity, which aids in temperature control during exothermic reactions, a critical safety feature when moving from liters to cubic meters. From an environmental standpoint, the process aligns perfectly with green chemistry principles by minimizing the use of volatile organic compounds (VOCs). This reduces the facility's carbon footprint and simplifies compliance with increasingly strict environmental protection laws. The ability to produce high-purity products with minimal environmental impact positions the manufacturer as a preferred partner for multinational corporations with aggressive sustainability goals.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this new synthesis route is essential for stakeholders evaluating its integration into existing production lines. The following questions address common concerns regarding safety, yield, and scalability, drawing directly from the experimental data and technical disclosures within the patent documentation. These insights are intended to facilitate informed decision-making for R&D and operations teams.
Q: Why is the new synthesis route for 5-mercapto tetrazole acetic acid considered safer than traditional methods?
A: The traditional route described in US4782012 relies heavily on methyl iodide, a highly toxic, volatile, and light-sensitive alkylating agent that poses severe occupational health risks and requires complex containment systems. The novel process disclosed in CN107805225B completely eliminates the use of methyl iodide by utilizing a phase-transfer catalyzed cycloaddition between sodium azide and isothiocyanato ethyl acetate in an aqueous medium, significantly reducing toxicity hazards and simplifying waste treatment protocols.
Q: What are the yield improvements observed in the patented aqueous synthesis method?
A: The patented methodology demonstrates a substantial improvement in overall process efficiency, achieving a total yield of up to 80% for the 5-mercapto tetrazole acetic acid synthesis. Furthermore, the subsequent conversion to mono-sodium and disodium salts exhibits single-step yields reaching up to 90%, which represents a significant optimization over older methodologies that often suffered from lower conversion rates and difficult purification steps due to side reactions.
Q: How does the use of Phase Transfer Catalysts impact the scalability of this reaction?
A: The utilization of benzyltrimethyl ammonium chloride as a phase transfer catalyst allows the reaction to proceed efficiently in a biphasic or aqueous system, facilitating the interaction between the inorganic azide ions and the organic isothiocyanate substrate. This mechanism not only accelerates the reaction rate under mild thermal conditions (50-80°C) but also ensures excellent heat transfer characteristics inherent to aqueous systems, making the process highly amenable to large-scale commercial manufacturing without the thermal runaway risks associated with organic solvent-based alkylations.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Mercapto Tetrazole Acetic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to greener, more efficient synthetic routes is not just a regulatory necessity but a strategic imperative for maintaining competitiveness in the global pharmaceutical market. Our team of expert chemists has thoroughly analyzed the technological breakthroughs presented in CN107805225B and is fully prepared to implement this advanced PTC-catalyzed methodology at commercial scale. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the high yields and purity specifications demonstrated in the patent are faithfully reproduced in our manufacturing facilities. Our rigorous QC labs are equipped to monitor critical quality attributes, guaranteeing that every batch of 5-mercapto tetrazole acetic acid meets the stringent requirements necessary for ceforanide synthesis.
We invite procurement leaders and technical directors to engage with us to explore how this optimized synthesis route can drive value for your organization. By leveraging our technical expertise and manufacturing capacity, we can offer a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. We encourage you to contact our technical procurement team today to request specific COA data, route feasibility assessments, and competitive quotations. Let us partner with you to secure a sustainable, cost-effective, and reliable supply of this critical pharmaceutical intermediate.
