Optimizing Drospirenone Intermediate Production via Novel 6,7-Methylene Steroid Synthesis
The pharmaceutical industry constantly seeks methods to optimize the synthesis of complex steroid intermediates, particularly for high-value active pharmaceutical ingredients (APIs) like Drospirenone. Patent CN101343306B discloses a groundbreaking method for synthesizing 6,7-methylene steroid compounds, which serve as critical precursors in the production of novel progestogens. This technology addresses a long-standing economic bottleneck in steroid chemistry by replacing expensive iodine-based methylating agents with more cost-effective bromine-based alternatives. By utilizing dibromomethane in conjunction with a zinc-copper couple catalyst, the process achieves high efficiency while significantly reducing raw material consumption. For global procurement teams and R&D directors, this represents a pivotal shift towards more sustainable and economically viable manufacturing routes for complex hormonal intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the introduction of a cyclopropyl group at the 6,7-position of steroid skeletons has relied heavily on the Simmons-Smith reaction variant using diiodomethane (CH2I2). As described in prior art such as US4435327, this conventional approach involves reacting a steroid with a 6,7-double bond structure with diiodomethane under the catalysis of a Zn-Cu reagent. While chemically effective, this method suffers from severe economic drawbacks that hinder its industrial scalability. The primary limitation is the exorbitant cost of diiodomethane, which is a specialized reagent with limited global supply compared to simpler halomethanes. Furthermore, the stoichiometric requirements are inefficient; reports indicate that the unit consumption of diiodomethane in these reactions can reach as high as 16 kg per kg of product. This massive material throughput not only drives up the direct cost of goods sold (COGS) but also creates significant waste management challenges due to the heavy iodine content in the effluent.
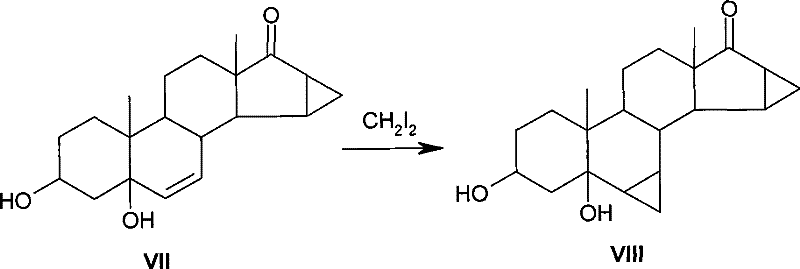
The Novel Approach
The innovative method disclosed in CN101343306B fundamentally alters the economic equation by substituting diiodomethane with dibromomethane (CH2Br2). This seemingly simple substitution requires precise optimization of reaction conditions to overcome the lower reactivity of the bromine analog compared to its iodine counterpart. The new process operates in a solvent system, preferably glycol dimethyl ether, using iodine merely as an initiator rather than the primary source of the methylene group. By shifting the bulk methylating agent to dibromomethane, the unit consumption is drastically reduced to a range of 7 to 10 kg/kg. This reduction is not merely incremental; it represents a structural change in the cost basis of the synthesis. Dibromomethane is a commodity chemical with a much lower price point and higher availability, ensuring that the supply chain for this critical intermediate remains robust and less susceptible to the volatility often seen with specialty iodine reagents.
Mechanistic Insights into Zn-Cu Catalyzed Cyclopropanation
The core of this synthesis lies in the generation of a reactive carbenoid species capable of inserting a methylene unit across the electron-rich 6,7-double bond of the steroid nucleus. The reaction utilizes a zinc-copper (Zn-Cu) couple, which is prepared by treating zinc powder with a copper salt, typically copper acetate, in glacial acetic acid. This activation step creates a highly reactive metal surface that facilitates the oxidative addition of the carbon-halogen bond in dibromomethane. Unlike the traditional Simmons-Smith reaction which relies heavily on the weak C-I bond, this modified protocol leverages the initiator (iodine) to jump-start the formation of the organozinc intermediate. Once formed, the iodomethylzinc species or the corresponding bromomethylzinc carbenoid attacks the alkene, forming the cyclopropane ring with high stereoselectivity. The presence of the 15-beta, 16-beta-methylene group in the starting material adds steric complexity, yet the Zn-Cu system demonstrates sufficient activity to navigate this crowded molecular environment effectively.
Controlling impurities in this cyclopropanation is critical for downstream pharmaceutical applications, particularly when the product serves as an intermediate for Drospirenone. The reaction conditions specified in the patent, such as maintaining a temperature between 65°C and 75°C, are optimized to balance reaction rate with selectivity. Excessive heat could lead to non-specific radical reactions or decomposition of the sensitive steroid backbone, while insufficient heat might result in incomplete conversion and residual starting material. The use of glycol dimethyl ether as a solvent is also mechanistically significant; it acts as a Lewis base that stabilizes the organozinc carbenoid species, preventing premature decomposition and ensuring a steady concentration of the active reagent throughout the reaction course. This stabilization is key to achieving the reported purities of over 85-90% directly from the crude reaction mixture, minimizing the need for extensive and yield-eroding purification steps later in the process.
How to Synthesize 6,7-Methylene Steroids Efficiently
The synthesis of these valuable intermediates requires strict adherence to the optimized parameters regarding reagent ratios and temperature control to ensure reproducibility. The process begins with the careful preparation of the Zn-Cu couple, followed by the suspension of the steroid substrate in the ether solvent. The addition of the dibromomethane must be controlled carefully to manage the exotherm and gas evolution associated with the reaction initiation. For a detailed breakdown of the specific operational steps, reagent quantities, and workup procedures required to implement this technology in a pilot or production setting, please refer to the standardized synthesis guide below.
- Prepare the Zn-Cu reagent by treating zinc powder with copper acetate in glacial acetic acid, followed by thorough washing with acetic acid and ether.
- Suspend the steroid substrate containing a 6,7-double bond in glycol dimethyl ether with the prepared Zn-Cu reagent and a catalytic amount of iodine.
- Add dibromomethane dropwise while heating to 65-75°C, maintaining the temperature until the cyclopropanation is complete, then isolate the product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition from diiodomethane to dibromomethane offers tangible strategic benefits beyond simple chemistry. The primary advantage is the substantial reduction in raw material costs, driven by the replacement of a high-cost specialty reagent with a widely available commodity chemical. This shift insulates the manufacturing process from the price volatility often associated with iodine markets. Additionally, the lower unit consumption means that less material needs to be purchased, stored, and handled per batch, optimizing warehouse utilization and reducing logistics overhead. The simplified waste profile, characterized by the absence of massive amounts of iodine byproducts, further translates to lower environmental compliance costs and easier disposal protocols, contributing to a greener and more sustainable supply chain operation.
- Cost Reduction in Manufacturing: The most immediate impact of this technology is the drastic lowering of the variable cost per kilogram of the intermediate. By utilizing dibromomethane, which is significantly cheaper than diiodomethane, and reducing the overall mass of reagent required by nearly half, the direct material cost is optimized. This efficiency allows manufacturers to maintain healthy margins even in competitive generic drug markets. Furthermore, the elimination of expensive iodine-based reagents reduces the burden on downstream purification processes, as there is less heavy metal contamination to remove, thereby saving on adsorbents and filtration media costs.
- Enhanced Supply Chain Reliability: Relying on dibromomethane enhances supply security because it is a standard industrial chemical produced in large volumes globally, unlike diiodomethane which may have fewer suppliers and longer lead times. This abundance ensures that production schedules are not disrupted by raw material shortages. The robustness of the Zn-Cu reagent preparation, which uses common zinc powder and copper salts, further diversifies the supply base. Companies adopting this method can negotiate better terms with vendors due to the commoditized nature of the inputs, ensuring a steady flow of materials essential for continuous manufacturing campaigns.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, utilizing standard reactor configurations and common solvents like glycol dimethyl ether. The reduced load of halogenated waste, specifically iodine, simplifies the environmental treatment of effluent streams. This aligns with increasingly stringent global environmental regulations regarding heavy metal discharge. The ability to run the reaction at moderate temperatures (65-75°C) without requiring cryogenic conditions or extreme pressures makes the technology energy-efficient and safer to operate on a multi-ton scale, facilitating a smoother transition from laboratory discovery to commercial production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing clarity on the feasibility and advantages of the dibromomethane route for stakeholders evaluating this technology for their supply chains.
Q: Why is dibromomethane preferred over diiodomethane in this synthesis?
A: Dibromomethane is significantly cheaper and more readily available than diiodomethane. The patented process reduces unit consumption from 16 kg/kg to 7-10 kg/kg, drastically lowering raw material costs.
Q: What is the role of the Zn-Cu couple in this reaction?
A: The Zn-Cu couple acts as the reducing agent that activates the dibromomethane, generating the reactive carbenoid species necessary to insert the methylene group across the 6,7-double bond of the steroid skeleton.
Q: Can this process be scaled for industrial production of Drospirenone intermediates?
A: Yes, the process is designed for industrial suitability. It uses common solvents like glycol dimethyl ether and avoids the extreme costs associated with iodine-based reagents, making large-scale manufacturing economically viable.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 6,7-Methylene Steroid Supplier
NINGBO INNO PHARMCHEM stands at the forefront of implementing advanced synthetic methodologies like the one described in CN101343306B to deliver high-value steroid intermediates. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this cost-saving chemistry are fully realized in practice. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 6,7-methylene steroid meets the exacting standards required for API synthesis. Our commitment to process optimization allows us to offer competitive pricing without compromising on the quality or consistency of the final product.
We invite pharmaceutical partners to collaborate with us to leverage this efficient synthesis route for their Drospirenone projects. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage potential clients to contact us directly to obtain specific COA data and route feasibility assessments, ensuring that your supply chain is built on the most robust and economical chemical foundations available today.
