Advanced Synthesis of 6,7-Methylene Steroids for High-Purity Pharmaceutical Intermediates
The pharmaceutical industry constantly seeks robust and cost-effective pathways for synthesizing complex steroidal intermediates, particularly those serving as precursors for advanced hormonal therapies. Patent CN101343306B introduces a significant technological advancement in the synthesis of 6,7-methylene steroid compounds, which are critical intermediates in the production of novel progestogens such as Drospirenone. This patent discloses a refined cyclopropanation method that replaces the traditionally expensive diiodomethane with the more economically viable dibromomethane, utilizing a zinc-copper (Zn-Cu) couple catalytic system initiated by iodine. By optimizing the reagent stoichiometry and reaction conditions, this process achieves high conversion rates while substantially lowering the unit consumption of methylating agents. For R&D directors and procurement specialists, this represents a pivotal shift towards more sustainable and budget-friendly manufacturing protocols without compromising the structural integrity or purity of the final steroidal scaffold.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the introduction of a cyclopropyl group at the 6,7-position of steroid skeletons has relied heavily on the Simmons-Smith reaction using diiodomethane as the methylene source. As illustrated in the prior art referenced by US4435327, the conventional pathway involves reacting a 6,7-olefinic steroid with methylene iodide under Zn-Cu catalysis to yield the corresponding methylene derivative. However, this traditional approach suffers from severe economic and operational drawbacks that hinder its viability for large-scale industrial application. The primary limitation is the exorbitant cost of diiodomethane, which is not only expensive to purchase but also required in excessive quantities; reports indicate a unit consumption reaching as high as 16 kg per kg of substrate. Furthermore, the handling of large volumes of iodine-containing reagents complicates waste management and increases the environmental burden, creating significant bottlenecks for supply chain managers aiming to optimize production costs and minimize hazardous waste disposal fees.
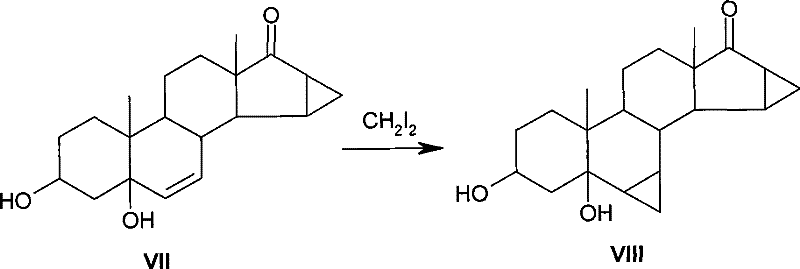
The Novel Approach
In stark contrast to the legacy methods, the novel approach detailed in CN101343306B leverages dibromomethane as a superior methylating agent, fundamentally altering the cost structure of the synthesis. This innovative method maintains the core utility of the Zn-Cu reagent but optimizes the reaction environment by introducing iodine as a specific initiator to activate the less reactive bromine species effectively. The process operates within a moderate temperature range of 65 to 75 degrees Celsius and utilizes glycol dimethyl ether as a high-boiling solvent to ensure stable reaction kinetics. By switching to dibromomethane, the unit consumption is dramatically reduced to a range of 7 to 10 kg per kg of substrate, representing a massive improvement in atom economy and reagent efficiency. This strategic substitution not only lowers the direct material costs but also simplifies the downstream purification processes, as the byproduct profile is more manageable compared to the iodine-heavy waste streams of the conventional route.
Mechanistic Insights into Zn-Cu Catalyzed Cyclopropanation
The core of this synthetic breakthrough lies in the precise generation and reactivity of the organozinc carbenoid species. In this mechanism, the Zn-Cu couple serves as the active metal surface that facilitates the oxidative insertion into the carbon-halogen bond of dibromomethane. The presence of the iodine initiator is crucial; it likely forms a more reactive zinc-iodide species in situ or activates the zinc surface, thereby lowering the activation energy required to generate the bromomethylzinc carbenoid intermediate. This carbenoid species then undergoes a concerted addition across the electron-rich 6,7-double bond of the steroid nucleus. The stereoelectronic properties of the rigid steroid framework direct the approach of the carbenoid, ensuring high diastereoselectivity during the formation of the cyclopropane ring. Understanding this mechanistic nuance is vital for R&D teams, as it highlights the importance of maintaining strict anhydrous conditions and controlling the addition rate of the dibromomethane to prevent non-selective side reactions or polymerization of the carbenoid species.
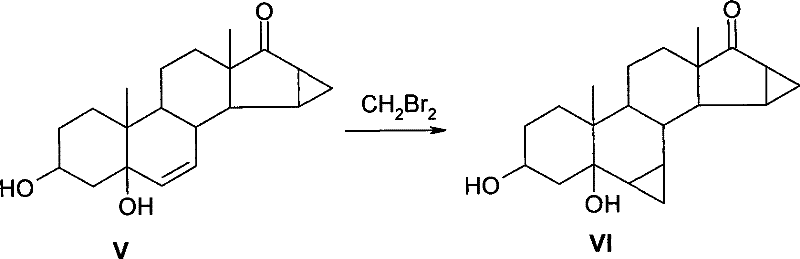
Impurity control in this reaction is governed by the stability of the Zn-Cu reagent and the precise stoichiometric balance of the reactants. The patent specifies a molar ratio of the steroid substrate to dibromomethane between 1:9 and 1:14, with a preferred range of 1:11 to 1:13, which ensures that there is sufficient methylene source to drive the reaction to completion without overwhelming the system with unreacted halides that could lead to poly-halogenated byproducts. The use of glycol dimethyl ether as a solvent is also mechanistically significant, as its coordinating ability helps stabilize the organozinc intermediates, preventing their premature decomposition. Furthermore, the workup procedure involving filtration through diatomite and extraction with ethyl acetate is designed to effectively remove zinc salts and residual copper species, resulting in a crude product with high purity levels, often exceeding 85 percent as detected by HPLC. This rigorous control over reaction parameters ensures that the final API intermediate meets the stringent quality standards required for pharmaceutical registration.
How to Synthesize 6,7-Methylene Steroids Efficiently
The synthesis of these valuable steroidal intermediates requires careful attention to the preparation of the catalyst and the control of reaction exotherms. The standardized protocol begins with the activation of the zinc powder using copper acetate in glacial acetic acid to form the Zn-Cu couple, followed by thorough washing and drying to ensure maximum reactivity. Once the catalyst is prepared, the steroid substrate is dissolved in the solvent, and the reaction is initiated under an inert atmosphere to prevent oxidation of the sensitive organometallic species. The detailed standardized synthesis steps are provided in the guide below.
- Preparation of Zn-Cu Reagent: Neutralize verdigris in glacial acetic acid, add zinc powder, stir until colorless, wash with acetic acid and ether, and dry under argon.
- Reaction Setup: Dissolve the 6,7-olefinic steroid substrate in glycol dimethyl ether, add the prepared Zn-Cu reagent and iodine initiator under inert gas protection.
- Cyclopropanation and Workup: Heat to 40-70°C, drip dibromomethane slowly, maintain temperature for 1-3 hours, then filter, extract, and purify the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this dibromomethane-based synthesis route offers compelling advantages that directly impact the bottom line and operational resilience. The most immediate benefit is the substantial reduction in raw material expenditure, driven by the replacement of high-cost diiodomethane with the significantly cheaper dibromomethane. This switch does not merely offer a marginal saving; it fundamentally restructures the variable cost of goods sold (COGS) for this key intermediate, allowing for more competitive pricing strategies in the global market. Additionally, the reduced unit consumption of the methylating agent means that logistics and storage requirements for hazardous chemicals are minimized, simplifying inventory management and reducing the risk associated with storing large quantities of volatile reagents.
- Cost Reduction in Manufacturing: The elimination of expensive diiodomethane and the reduction in reagent volume per kilogram of product lead to drastic cost savings in the manufacturing process. By lowering the dependency on precious iodine resources, the production facility can achieve a more stable cost structure that is less susceptible to fluctuations in the global halogen market. This efficiency extends to the waste treatment phase, where the lower load of heavy halogen waste reduces the complexity and expense of effluent treatment, further enhancing the overall economic viability of the process for large-scale commercial operations.
- Enhanced Supply Chain Reliability: Dibromomethane is a widely available commodity chemical with a robust global supply chain, unlike diiodomethane which can be subject to supply constraints and longer lead times. Utilizing a more common reagent ensures that production schedules are not disrupted by raw material shortages, providing a higher degree of certainty for delivery commitments to downstream API manufacturers. The simplicity of the reaction setup, which does not require exotic catalysts or extreme conditions, also means that the process can be easily transferred between different manufacturing sites or scaled up rapidly to meet surges in demand without significant re-engineering of the production line.
- Scalability and Environmental Compliance: The process is inherently scalable due to its use of standard organic solvents and manageable reaction temperatures, making it ideal for transition from pilot plant to multi-ton commercial production. From an environmental perspective, the reduction in iodine usage aligns with green chemistry principles by minimizing the generation of persistent halogenated organic pollutants. This compliance with stricter environmental regulations reduces the regulatory burden on the manufacturer and mitigates the risk of production shutdowns due to environmental non-compliance, ensuring a continuous and sustainable supply of high-purity pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing clarity on the feasibility and benefits of the technology for potential partners and licensees.
Q: Why is dibromomethane preferred over diiodomethane in this steroid synthesis?
A: Dibromomethane is significantly cheaper than diiodomethane and requires lower unit consumption (7-10 kg/kg vs 16 kg/kg), drastically reducing raw material costs for industrial scale-up.
Q: What is the role of the Zn-Cu couple in this reaction?
A: The Zn-Cu couple acts as the active metal species that generates the carbenoid intermediate from dibromomethane, facilitating the cyclopropanation of the 6,7-double bond efficiently.
Q: Can this method be scaled for commercial production of Drospirenone intermediates?
A: Yes, the method uses simple operations, common solvents like glycol dimethyl ether, and avoids expensive reagents, making it highly suitable for large-scale commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 6,7-Methylene Steroid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient synthetic routes in the competitive landscape of pharmaceutical intermediates. Our team of expert chemists has thoroughly analyzed the technology disclosed in CN101343306B and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We are committed to delivering high-purity 6,7-methylene steroids that meet stringent purity specifications, utilizing our rigorous QC labs to ensure every batch conforms to the highest international standards. Our state-of-the-art facilities are equipped to handle the specific requirements of Zn-Cu mediated reactions, ensuring safety and consistency at every stage of the manufacturing process.
We invite global pharmaceutical partners to collaborate with us to leverage this cost-effective technology for your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized synthesis of 6,7-methylene steroids can enhance your project's economic potential and accelerate your time to market.
