Advanced Sildenafil Intermediate Synthesis: Overcoming Traditional Limitations for Commercial Scale-up
Advanced Sildenafil Intermediate Synthesis: Overcoming Traditional Limitations for Commercial Scale-up
The pharmaceutical industry continuously seeks robust, scalable, and cost-effective pathways for the production of high-volume Active Pharmaceutical Ingredients (APIs) such as Sildenafil Citrate. A pivotal advancement in this domain is detailed in patent CN112608317A, which discloses a novel preparation method that fundamentally restructures the synthetic logic of the Sildenafil side chain and its subsequent condensation. Unlike conventional routes that rely on hazardous heavy metal reductions or expensive coupling agents, this innovation leverages a Zinc-Ammonium Chloride reduction system and Thionyl Chloride activation to achieve superior process efficiency. For R&D directors and supply chain managers, this represents a critical opportunity to optimize the manufacturing of this high-demand erectile dysfunction treatment. By transitioning away from Palladium-Carbon hydrogenation and Carbonyldiimidazole (CDI) coupling, the process mitigates significant safety risks and reduces the burden of impurity control, particularly regarding genetic toxicants. This report analyzes the technical merits of this patented methodology, demonstrating how it serves as a reliable foundation for a reliable sildenafil intermediate supplier aiming to enhance market competitiveness through technological superiority.
The Limitations of Conventional Methods vs. The Novel Approach
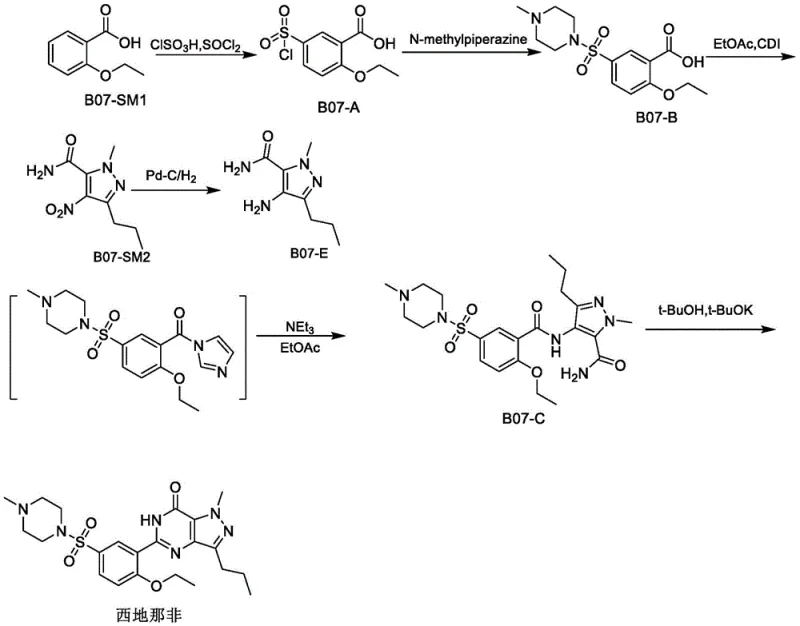
The Limitations of Conventional Methods
Historically, the industrial synthesis of Sildenafil has been plagued by several critical bottlenecks that impact both cost and safety profiles. Prior art, such as that disclosed in US5250534A, utilizes stannous chloride for the reduction of the nitro group on the pyrazole ring. This approach introduces severe downstream processing challenges, including the inevitable introduction of tin heavy metals which require complex and costly removal steps to meet stringent pharmaceutical specifications. Furthermore, the workup often involves massive volumes of dichloromethane, leading to emulsification issues during extraction and significant environmental waste. Alternatively, methods described in literature like Org. Process Res. Dev. 2000 employ Palladium-Carbon catalytic hydrogenation. While effective chemically, this necessitates specialized high-pressure equipment, posing serious safety hazards in a production environment. Moreover, Pd-C reduction often generates unreduced intermediates and impurities with warning structures that are difficult to purge. Perhaps most critically, the condensation step using CDI as a coupling agent is notoriously sluggish, requiring reaction times as long as 70 hours, which drastically inflates production cycles and equipment occupancy costs.
The Novel Approach
The methodology presented in patent CN112608317A offers a transformative solution to these entrenched problems by re-engineering the reduction and condensation steps. Instead of relying on precious metal catalysts or toxic tin reagents, the new process employs a Zinc powder and Ammonium Chloride system in an aqueous ethanol medium. This shift not only eliminates the need for high-pressure hydrogenation vessels but also simplifies the post-reaction workup to a straightforward filtration and salification, avoiding the emulsification nightmares associated with dichloromethane extractions. In the condensation phase, the process replaces the slow-acting CDI with Thionyl Chloride activation. This modification accelerates the reaction kinetics dramatically, reducing the condensation timeframe from days to merely a few hours. By integrating these changes, the novel approach delivers a streamlined workflow that minimizes solvent consumption, avoids the introduction of difficult-to-remove imidazole impurities, and significantly shortens the overall production period, making it highly attractive for cost reduction in API manufacturing.
Mechanistic Insights into Zinc-Mediated Reduction and Acid Chloride Activation
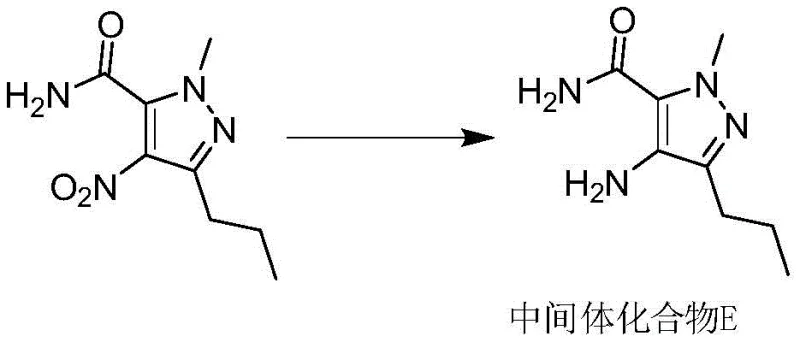
The core chemical innovation lies in the mechanistic efficiency of the Zinc-Ammonium Chloride reduction system. In this pathway, 1-methyl-4-nitro-3-propyl-1H-pyrazole-5-carboxamide undergoes a multi-electron transfer reduction where Zinc acts as the electron donor in the presence of ammonium ions. This system effectively converts the nitro group to the corresponding amine (Intermediate E) without the over-reduction or side reactions often seen with heterogeneous catalysis. The presence of ammonium chloride buffers the reaction medium, facilitating the protonation steps necessary for the reduction while maintaining a pH environment that prevents the degradation of the sensitive pyrazole ring. Following isolation as an acid salt, this intermediate is ready for immediate coupling. The subsequent activation of the benzoic acid derivative (Intermediate B) utilizes Thionyl Chloride to generate a highly reactive acid chloride species in situ. This electrophile reacts rapidly with the nucleophilic amine of Intermediate E in the presence of an acid-binding agent like triethylamine or pyridine. This mechanism bypasses the formation of the stable acyl-imidazole intermediate required in CDI couplings, thereby removing the kinetic barrier that previously extended reaction times to 70 hours. The result is a clean, high-yielding amide bond formation that proceeds to completion within approximately 2.5 hours total process time.
From an impurity control perspective, this mechanistic shift is profound. The avoidance of Palladium-Carbon eliminates the risk of residual heavy metal contamination and the formation of de-halogenated or hydrogenolysis byproducts that are common in catalytic hydrogenation. Furthermore, by eschewing CDI, the process inherently prevents the formation of imidazole-related impurities, which are often classified as genotoxic and require rigorous and expensive analytical monitoring and purification. The Zinc reduction system produces inorganic zinc salts as byproducts, which are easily removed via aqueous washing and filtration, leaving the organic product with high purity. This inherent cleanliness of the reaction profile ensures that the final Sildenafil product meets stringent quality standards with minimal downstream purification effort. For a reliable sildenafil intermediate supplier, this level of impurity control is paramount, as it directly correlates to the regulatory success and batch consistency of the final drug substance.
How to Synthesize Sildenafil Efficiently
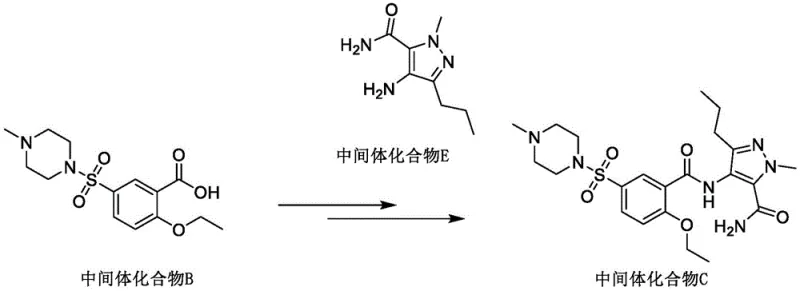
The implementation of this patented route requires precise control over reaction parameters to maximize yield and purity, particularly during the exothermic activation and reduction phases. The process is divided into three distinct operational stages: the preparation of the amine side chain via reduction, the activation and condensation to form the open-chain precursor, and the final cyclization. Operators must pay close attention to temperature control during the addition of Zinc powder to manage hydrogen evolution and heat generation. Similarly, the activation of the benzoic acid with Thionyl Chloride must be conducted under anhydrous conditions to prevent hydrolysis of the acid chloride. The following guide outlines the standardized operational framework derived from the patent examples, providing a clear roadmap for technical teams looking to adopt this superior methodology.
- Reduce 1-methyl-4-nitro-3-propyl-1H-pyrazole-5-carboxamide using zinc powder and ammonium chloride in aqueous ethanol to obtain Intermediate E.
- Activate Intermediate B (benzoic acid derivative) with thionyl chloride in an aprotic solvent, then condense with Intermediate E salt using an acid-binding agent to form Intermediate C.
- Perform ring-closing cyclization on Intermediate C in a lower alkyl alcohol solvent under alkaline conditions to yield the final Sildenafil product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of the synthesis route described in CN112608317A translates into tangible strategic benefits that extend beyond mere chemical elegance. The primary advantage lies in the drastic simplification of the supply chain for raw materials and the reduction of dependency on specialized infrastructure. By eliminating the need for Palladium on Carbon and high-pressure hydrogenation reactors, manufacturers can operate within standard glass-lined or stainless steel vessels, significantly lowering capital expenditure (CAPEX) and maintenance costs. Furthermore, the substitution of expensive coupling agents like CDI with commodity chemicals like Thionyl Chloride and Zinc powder results in substantial cost savings in direct material expenses. The reduction in reaction time from 70 hours to roughly 2.5 hours for the condensation step dramatically increases plant throughput, allowing for more batches to be produced within the same timeframe, effectively expanding capacity without physical expansion. This efficiency gain is crucial for meeting global demand fluctuations without incurring overtime labor costs or delaying shipments.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the elimination of high-cost reagents and the simplification of waste treatment. Traditional methods utilizing CDI involve expensive reagents that contribute significantly to the Cost of Goods Sold (COGS). By switching to Thionyl Chloride, which is a bulk commodity chemical, the direct material cost is significantly reduced. Additionally, the removal of Tin or Palladium catalysts eliminates the need for expensive scavenger resins or complex extraction protocols designed to lower heavy metal residues to ppm levels. The simplified workup, which relies on filtration rather than complex liquid-liquid extractions prone to emulsification, reduces solvent consumption and labor hours. These factors combine to create a leaner manufacturing process that offers a competitive pricing structure for the final intermediate, enhancing margin potential for the manufacturer.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of widely available, non-specialized raw materials. Zinc powder, ammonium chloride, and thionyl chloride are produced globally in massive quantities, ensuring that supply disruptions are unlikely compared to specialized catalysts which may have limited suppliers or long lead times. The robustness of the chemistry also means that the process is less sensitive to minor variations in raw material quality, reducing the rate of batch failures. The shortened production cycle time means that inventory turnover is faster, allowing the supply chain to respond more agilely to market demands. This reliability is essential for maintaining continuous production schedules and ensuring that downstream API manufacturers receive their materials on time, preventing bottlenecks in the broader pharmaceutical supply network.
- Scalability and Environmental Compliance: From an environmental and scaling perspective, this route is exceptionally well-suited for commercial scale-up. The absence of high-pressure hydrogenation removes a major safety barrier to scaling, as the process can be run safely in standard reactors at atmospheric pressure. The waste stream is significantly cleaner, consisting primarily of inorganic zinc salts and aqueous washes, which are easier and cheaper to treat than organic waste streams laden with heavy metals or imidazole derivatives. This aligns with increasingly strict environmental regulations regarding heavy metal discharge and solvent emissions. The ability to scale this process from kilogram to multi-ton scales without encountering the engineering challenges associated with high-pressure gas handling makes it an ideal candidate for large-volume commercial production, ensuring long-term viability and regulatory compliance.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel Sildenafil synthesis route. These insights are derived directly from the comparative data and experimental examples provided in the patent documentation, offering clarity on how this method outperforms legacy technologies. Understanding these nuances is vital for technical teams evaluating the feasibility of technology transfer and for procurement professionals assessing the long-term value proposition of this supply source.
Q: How does the Zinc-Ammonium Chloride reduction system improve safety compared to traditional methods?
A: Traditional methods often utilize stannous chloride (generating heavy metal waste) or Palladium-Carbon hydrogenation (requiring high-pressure equipment). The Zinc-Ammonium Chloride system operates at atmospheric pressure without introducing toxic heavy metals like tin, significantly simplifying post-treatment and reducing environmental hazards.
Q: What are the advantages of using Thionyl Chloride activation over Carbonyldiimidazole (CDI)?
A: Using CDI typically requires prolonged reaction times (up to 70 hours) and introduces difficult-to-remove imidazole impurities. Thionyl Chloride activation is rapid (approx. 2 hours) and avoids the formation of genotoxic imidazole-related impurities, ensuring higher purity and shorter production cycles.
Q: Is this synthesis route suitable for large-scale commercial production?
A: Yes, the route is specifically designed for industrial scalability. It eliminates the need for specialized high-pressure hydrogenation reactors and complex extraction processes prone to emulsification, relying instead on standard filtration and crystallization unit operations common in fine chemical manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sildenafil Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced synthetic routes like the one described in CN112608317A requires a partner with deep technical expertise and proven manufacturing capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in practice. Our facilities are equipped with the necessary infrastructure to handle the specific unit operations required by this process, including robust filtration systems and standard reactor trains that eliminate the need for specialized high-pressure equipment. We maintain stringent purity specifications and operate rigorous QC labs to monitor for the specific impurities discussed, such as imidazole derivatives and heavy metals, guaranteeing that every batch of Sildenafil intermediate meets the highest global pharmacopoeia standards.
We invite pharmaceutical companies and generic drug manufacturers to collaborate with us to leverage this cost-effective and scalable technology. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this Zinc/Thionyl Chloride route for your specific volume requirements. We encourage you to contact us to request specific COA data from our pilot batches and to discuss route feasibility assessments tailored to your supply chain needs. Let us help you secure a stable, high-quality, and economically optimized supply of Sildenafil intermediates for your global markets.
