Optimizing Bendamustine Intermediate Production: A Technical Breakthrough in Reductive Alkylation
Optimizing Bendamustine Intermediate Production: A Technical Breakthrough in Reductive Alkylation
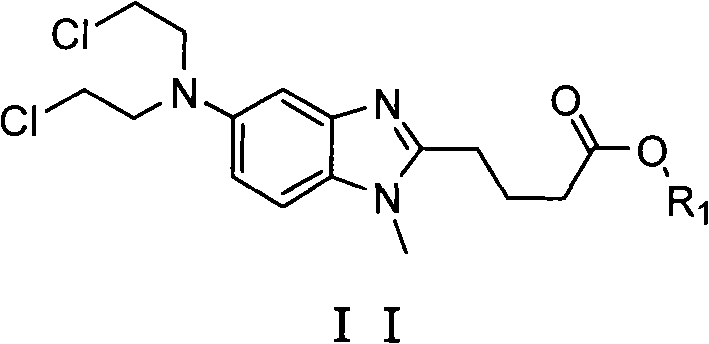
The pharmaceutical industry continuously seeks robust synthetic routes for oncology API intermediates that balance high purity with operational safety. Patent CN102558069A introduces a significant advancement in the preparation of key bendamustine intermediates, specifically targeting the synthesis of [1-methyl-2-(4'-butyric acid ester)-5-N,N-bis(2'-chloroethyl)]-1H-benzimidazole derivatives. This technology addresses critical bottlenecks found in legacy manufacturing processes by shifting from hazardous gaseous reagents to controlled liquid-phase reductive alkylation. For R&D directors and procurement specialists, this patent represents a viable pathway to secure a reliable pharmaceutical intermediates supplier capable of delivering material with purity exceeding 99.5% while mitigating the risks associated with traditional nitrogen mustard synthesis. The core innovation lies in the strategic selection of starting materials and the optimization of the reduction-alkylation sequence, ensuring that the final product meets stringent regulatory specifications for downstream API synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of nitrogen mustard-containing intermediates like those required for bendamustine has relied heavily on the use of ethylene oxide and thionyl chloride. These conventional pathways, while chemically feasible, present severe operational challenges for large-scale manufacturing. The use of ethylene oxide requires specialized high-pressure equipment and rigorous safety protocols due to its explosive and carcinogenic nature, creating significant barriers to entry for many contract manufacturers. Furthermore, processes utilizing thionyl chloride for chlorination steps often generate substantial quantities of acidic byproducts and sulfur-containing waste, complicating effluent treatment and increasing environmental compliance costs. Prior art methods, such as those described in reference documents within the patent, frequently suffer from lower yields, typically hovering around 65% to 70%, and struggle to control single impurities below the critical 0.1% threshold without extensive and costly chromatographic purification steps.
The Novel Approach
The methodology disclosed in CN102558069A fundamentally reengineers the synthetic route by employing a reductive alkylation strategy using chloroacetic acid derivatives and borane complexes. Instead of relying on the dangerous addition of ethylene oxide to an amine, this novel approach facilitates the formation of the bis-chloroethyl moiety through a safer, solution-phase reaction. By utilizing the free base form of the amino-benzimidazole starting material rather than its hydrochloride salt, the process eliminates the stoichiometric waste associated with neutralizing hydrochloric acid. This shift not only streamlines the reaction stoichiometry but also enhances the overall yield, pushing performance metrics to over 74% and potentially reaching 82% under optimized conditions. The result is a cleaner reaction profile that inherently supports cost reduction in pharmaceutical intermediates manufacturing by reducing reagent consumption and simplifying the work-up procedure.
Mechanistic Insights into Borane-Mediated Reductive Alkylation
The chemical elegance of this process lies in the mechanism of converting the primary amine of the starting material into the tertiary bis-chloroethyl amine of the target intermediate. The reaction likely proceeds through an initial amidation between the aromatic amine and chloroacetic acid, followed by a powerful reduction mediated by borane species such as borane-tetrahydrofuran or borane-dimethyl sulfide. Borane is uniquely suited for this transformation as it selectively reduces amide and carboxylic acid functionalities to amines and alcohols under mild conditions, typically below 25°C. This low-temperature operation is crucial for maintaining the stability of the benzimidazole core and preventing the decomposition of the sensitive chloroethyl groups. The use of the free base starting material ensures that the full equivalent of the reducing agent is available for the desired chemical transformation rather than being consumed by proton neutralization, thereby maximizing atom economy and reaction efficiency.
Impurity control is another critical aspect where this mechanism excels. Traditional methods often produce complex mixtures of mono-alkylated species and polymeric byproducts due to the uncontrolled reactivity of ethylene oxide. In contrast, the stepwise nature of the amide formation and subsequent reduction allows for tighter kinetic control over the alkylation process. The patent data indicates that single impurities are consistently maintained below 0.1%, a specification that is vital for meeting the rigorous quality standards of global oncology drug regulators. This high level of purity is further reinforced by the recrystallization protocol, which leverages the differential solubility of the product in methanol-water mixtures to exclude trace organic contaminants, ensuring that the high-purity pharmaceutical intermediates delivered to the next synthesis stage are of exceptional quality.
How to Synthesize Bendamustine Intermediate Efficiently
Implementing this synthesis route requires precise control over reaction parameters to replicate the high yields and purity reported in the patent literature. The process is designed to be scalable, moving seamlessly from laboratory glassware to industrial reactors with minimal modification. Operators must pay close attention to the temperature profile during the addition of the reducing agent, as exothermic events can compromise product integrity if not managed correctly. The following guide outlines the standardized operational framework derived from the patent examples, providing a clear roadmap for technical teams aiming to adopt this superior manufacturing protocol. Detailed standardized synthesis steps are provided in the section below to ensure reproducibility and safety compliance.
- Dissolve the amino-benzimidazole starting material and chloroacetic acid derivative in an organic solvent like THF, then add a borane reducing agent below 25°C.
- Quench the reaction mixture to neutrality using a carbonate solution, filter the crude solid precipitate, and wash thoroughly with water.
- Purify the crude product by dissolving in a methanol-water mixture, stirring in ice water to induce crystallization, and drying under reduced pressure.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible benefits that extend beyond mere chemical yield. The elimination of hazardous gases like ethylene oxide drastically reduces the capital expenditure required for specialized containment systems, making the production of these intermediates more accessible to a broader range of qualified suppliers. This diversification of the supply base enhances supply chain reliability, reducing the risk of disruptions caused by facility-specific safety shutdowns. Furthermore, the switch to common solvents like tetrahydrofuran and methanol, coupled with the avoidance of expensive purification solvents like methyl tert-butyl ether, leads to substantial cost savings in raw material procurement. These efficiencies translate directly into a more competitive pricing structure for the final API, allowing pharmaceutical companies to optimize their cost of goods sold without compromising on quality.
- Cost Reduction in Manufacturing: The process achieves significant cost optimization by improving reagent efficiency. By using the free base starting material, the consumption of expensive reducing agents like borane and alkylating agents like chloroacetic acid is minimized, as no excess is needed to counteract acid salts. Additionally, the simplified purification process using methanol and water eliminates the need for costly solvent exchanges and complex distillation steps associated with ether-based systems. This streamlined workflow reduces energy consumption and labor hours, driving down the overall manufacturing cost per kilogram of the intermediate.
- Enhanced Supply Chain Reliability: Sourcing intermediates produced via this method mitigates risks associated with hazardous material logistics. Since the process avoids the transport and storage of pressurized ethylene oxide, suppliers face fewer regulatory hurdles and transportation delays. The robustness of the reaction, which tolerates standard industrial equipment rather than requiring bespoke high-pressure vessels, means that more manufacturing sites are capable of producing this material. This increased capacity availability ensures a stable and continuous supply of high-purity pharmaceutical intermediates, safeguarding production schedules for downstream API manufacturers.
- Scalability and Environmental Compliance: The environmental footprint of this synthesis is markedly lower than traditional methods. The absence of sulfur-containing reagents like thionyl chloride simplifies waste treatment, reducing the load on effluent processing plants and lowering disposal costs. The use of aqueous methanol for crystallization is inherently greener and easier to recover than halogenated or ether solvents. These factors make the process highly scalable and compliant with increasingly strict environmental regulations, ensuring long-term viability for commercial scale-up of complex pharmaceutical intermediates without the threat of regulatory shutdowns.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. Understanding these details is essential for technical teams evaluating the feasibility of integrating this intermediate into their supply chain. The answers are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering a transparent view of the technology's capabilities and limitations.
Q: Why is the free base starting material preferred over the hydrochloride salt?
A: Using the free base eliminates the need for excess reducing agents and bases to neutralize hydrochloric acid, significantly improving atom economy and reducing waste generation compared to salt-based protocols.
Q: What are the safety advantages of this method over traditional ethylene oxide routes?
A: This protocol avoids the use of highly hazardous ethylene oxide gas and thionyl chloride, replacing them with safer liquid reagents like chloroacetic acid and borane complexes, thereby lowering equipment safety requirements.
Q: How does the purification process impact overall production costs?
A: The method utilizes a cost-effective methanol-water system for recrystallization instead of expensive solvents like methyl tert-butyl ether (MTBE), simplifying solvent recovery and reducing raw material expenses.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bendamustine Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development of life-saving oncology therapies. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot scale to full manufacturing is seamless and efficient. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to verify that every batch of bendamustine intermediate meets the >99.5% purity benchmark established by leading patent technologies. Our infrastructure is designed to handle complex reductive alkylation chemistries safely, providing our partners with a secure and dependable source for their critical raw materials.
We invite procurement leaders and R&D directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements. By leveraging our optimized manufacturing protocols, we can help you reduce lead time for high-purity pharmaceutical intermediates while achieving significant economic benefits. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can strengthen your supply chain resilience and drive your project forward.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
