Optimizing Bendamustine Intermediate Production for Commercial Scale and Purity
The pharmaceutical industry continuously seeks robust synthetic routes for oncology drugs, particularly for alkylating agents like bendamustine hydrochloride. A pivotal advancement in this domain is documented in patent CN102558069B, which details a highly efficient method for preparing critical intermediates such as [1-methyl-2-(4'-butyric acid ethyl)-5-N,N-bis(2'-chloroethyl)]-1H-benzimidazole. This technology addresses long-standing challenges in yield optimization and impurity control, offering a pathway that is significantly more viable for large-scale manufacturing than previous iterations. By leveraging specific borane-mediated reduction techniques, the process achieves purity levels exceeding 99.5% with single impurities maintained below 0.1%. For R&D Directors and Procurement Managers, understanding the nuances of this patented approach is essential for securing a reliable pharmaceutical intermediate supplier capable of meeting stringent regulatory standards while ensuring supply chain continuity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of bendamustine intermediates relied heavily on hazardous reagents that complicated both the reaction engineering and the downstream purification processes. Traditional routes often necessitated the use of thionyl chloride for substitution reactions, a reagent known to generate significant quantities of difficult-to-remove impurities that compromise the final quality of the active pharmaceutical ingredient. Furthermore, the reliance on ethylene oxide for alkylation steps imposed severe constraints on industrial equipment, requiring high-pressure reactors and specialized safety protocols to mitigate the risks associated with this carcinogenic gas. These factors collectively contributed to higher operational costs, extended lead times for high-purity pharmaceutical intermediates, and increased environmental burdens due to complex waste streams. Consequently, manufacturers faced substantial difficulties in scaling these processes without incurring prohibitive expenses or safety liabilities.
The Novel Approach
In stark contrast, the methodology outlined in the referenced patent introduces a transformative strategy that circumvents these traditional bottlenecks by utilizing chloroacetic acid and borane complexes in tetrahydrofuran. This innovative route consolidates multiple reaction steps into a more streamlined sequence, effectively reducing the overall processing time and minimizing the exposure of intermediates to conditions that foster degradation. By eliminating the need for ethylene oxide and thionyl chloride, the new approach drastically simplifies the reactor requirements and enhances the safety profile of the manufacturing facility. Moreover, the process demonstrates a remarkable ability to suppress side reactions, resulting in a crude product that requires less intensive purification to reach commercial grade specifications. This shift represents a significant leap forward in cost reduction in pharmaceutical intermediate manufacturing, aligning technical feasibility with economic efficiency.
Mechanistic Insights into Borane-Mediated Reduction and Alkylation
The core of this synthetic breakthrough lies in the precise manipulation of the reduction and alkylation mechanisms using borane-tetrahydrofuran complexes. Unlike conventional reducing agents that might lack selectivity or require harsh conditions, borane facilitates the smooth conversion of the carboxylic acid or ester functionalities while simultaneously enabling the N-alkylation of the benzimidazole ring. The reaction is conducted at controlled temperatures below 25°C, which is critical for preventing thermal decomposition and ensuring the stereochemical integrity of the molecule. This mild condition not only preserves the structural fidelity of the sensitive benzimidazole core but also allows for the use of standard glass-lined or stainless steel reactors without the need for exotic materials resistant to extreme corrosion. The mechanistic efficiency ensures that the molar ratio of alkylating agent to substrate can be optimized, reducing excess reagent waste and simplifying the workup procedure.
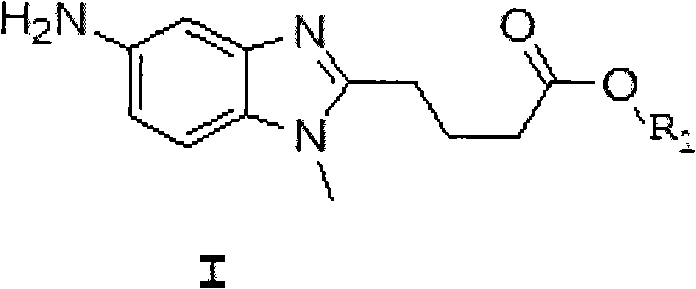
Furthermore, a crucial aspect of this mechanism involves the choice of the starting material form, specifically utilizing the free base of [1-methyl-2-(4'-butyric acid ethyl)-5-amino]-1H-benzimidazole rather than its hydrochloride salt. When the hydrochloride salt is used, a portion of the expensive reducing agent is consumed merely to neutralize the hydrochloric acid before the actual reduction can occur, leading to inefficient reagent usage and lower overall yields. By employing the free base, the process ensures that every mole of the reducing agent contributes directly to the desired chemical transformation, thereby maximizing atom economy. This subtle yet impactful modification underscores the depth of process optimization achieved in this patent, providing a clear advantage for commercial scale-up of complex pharmaceutical intermediates where margin improvements are vital.
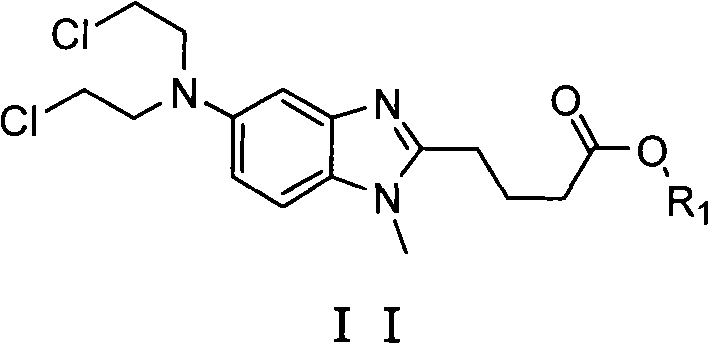
Impurity control is another pillar of this mechanistic design, achieved through a carefully orchestrated precipitation and recrystallization protocol. The reaction mixture is adjusted to a neutral pH using sodium carbonate, which induces the precipitation of the crude product while leaving soluble impurities in the supernatant. Subsequent purification involves dissolving the crude solid in a mixture of methanol and water, a solvent system chosen for its ability to differentially solubilize the target compound versus residual byproducts. Cooling this solution in an ice bath promotes the formation of well-defined crystals, effectively excluding trace impurities from the crystal lattice. This physical separation method is far more robust than chromatographic techniques for large-scale production, offering a scalable solution that consistently delivers purity levels above 99.6%. Such rigorous control over the impurity profile is indispensable for meeting the stringent specifications required by global regulatory bodies for oncology drug substances.
How to Synthesize Bendamustine Key Intermediate Efficiently
Implementing this synthesis route requires strict adherence to the defined operational parameters to ensure reproducibility and safety at scale. The process begins with the dissolution of the amino-benzimidazole starting material and chloroacetic acid in an organic solvent, followed by the controlled addition of the borane reducing agent while maintaining the internal temperature below 25°C to prevent exothermic runaway. Once the reaction is complete, the mixture is concentrated and treated with a carbonate solution to precipitate the crude intermediate, which is then isolated via filtration. The final purification step utilizes a methanol-water recrystallization to achieve the target purity, avoiding the use of more hazardous or expensive solvents like methyl tert-butyl ether. Detailed standardized synthesis steps see the guide below.
- Dissolve the amino-benzimidazole starting material and chloroacetic acid in an organic solvent like tetrahydrofuran, then add a borane reducing agent below 25°C.
- Adjust the reaction mixture to neutral pH using a carbonate solution to precipitate the crude product, followed by filtration and washing.
- Purify the crude solid by dissolving in a methanol-water mixture, cooling to induce crystallization, and drying to obtain the final high-purity intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis route translates into tangible strategic benefits that extend beyond mere technical metrics. The elimination of hazardous reagents like ethylene oxide reduces the regulatory burden and insurance costs associated with storing and handling dangerous chemicals, thereby lowering the overall overhead of the manufacturing operation. Additionally, the simplified purification process using methanol and water significantly cuts down on solvent procurement costs and waste disposal fees, contributing to substantial cost savings over the lifecycle of the product. The robustness of the reaction conditions also implies a lower risk of batch failures, ensuring a more predictable and reliable supply of critical materials for downstream API production. This stability is crucial for maintaining uninterrupted drug supply chains in the competitive oncology market.
- Cost Reduction in Manufacturing: The process achieves significant economic efficiency by optimizing reagent consumption and eliminating the need for expensive purification solvents. By using the free base starting material, the consumption of reducing agents is minimized, as no excess is required to neutralize acid salts. Furthermore, the replacement of methyl tert-butyl ether with a methanol-water system for recrystallization drastically reduces solvent costs and simplifies solvent recovery operations. These cumulative efficiencies result in a lower cost of goods sold, allowing for more competitive pricing strategies without compromising margin integrity.
- Enhanced Supply Chain Reliability: The avoidance of ethylene oxide removes a major bottleneck related to specialized equipment and safety regulations, making the production process more accessible to a wider range of qualified manufacturers. This flexibility enhances the resilience of the supply chain by reducing dependency on facilities with niche high-pressure capabilities. Moreover, the high yield and consistency of the reaction minimize the need for reprocessing or discarding off-spec batches, ensuring that delivery schedules are met with greater certainty. This reliability is paramount for partners seeking a reliable pharmaceutical intermediate supplier who can guarantee continuity of supply.
- Scalability and Environmental Compliance: The mild reaction conditions and aqueous workup procedures make this process inherently easier to scale from pilot plant to commercial production volumes without encountering significant engineering hurdles. The reduction in hazardous waste generation aligns with increasingly strict environmental regulations, reducing the liability and cost associated with waste treatment. This green chemistry approach not only improves the corporate sustainability profile but also future-proofs the manufacturing process against tightening environmental legislation, ensuring long-term operational viability.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of bendamustine intermediates using this advanced methodology. These insights are derived directly from the patent data and practical implementation experiences, aiming to clarify the advantages of this route over legacy methods. Understanding these details helps stakeholders make informed decisions about process adoption and supplier selection. Comprehensive answers based on patent specifics follow below.
Q: Why is the free base starting material preferred over the hydrochloride salt?
A: Using the free base form of the amino-benzimidazole eliminates the need for excess reducing agents to neutralize hydrochloric acid, thereby improving overall yield and reducing reagent consumption significantly.
Q: What are the safety advantages of this synthesis route?
A: This method avoids the use of highly hazardous ethylene oxide and corrosive thionyl chloride, reducing equipment requirements and enhancing operational safety for industrial scale-up.
Q: How does the purification process impact cost?
A: The recrystallization utilizes a methanol-water system instead of expensive methyl tert-butyl ether, lowering solvent costs and simplifying waste treatment protocols.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bendamustine Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development and commercialization of life-saving oncology therapies. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to verify that every batch meets the highest international standards. Our expertise in handling complex chemistries, such as the borane-mediated reductions described in CN102558069B, positions us as a strategic partner capable of navigating the intricacies of modern pharmaceutical manufacturing.
We invite you to engage with our technical procurement team to discuss how we can support your specific project requirements and optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our manufacturing capabilities can reduce your overall production costs while enhancing quality. We encourage potential partners to contact us for specific COA data and route feasibility assessments to validate our capacity to meet your demanding specifications. Let us collaborate to drive efficiency and innovation in your drug development pipeline.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
