Advanced Manufacturing of Rosuvastatin Calcium Side Chain Intermediates via Green Organometallic Routes
Introduction to Next-Generation Rosuvastatin Synthesis
The global pharmaceutical landscape continues to rely heavily on statins for managing hypercholesterolemia, with Rosuvastatin Calcium standing as a cornerstone therapy due to its potent HMG-CoA reductase inhibitory activity. As depicted in the molecular structure below, the integrity of the side chain, particularly the (3R,5S)-dihydroxy heptenoic acid moiety, is critical for biological efficacy. However, the manufacturing of this complex molecule has historically been plagued by environmentally hazardous reagents and convoluted synthetic pathways. Patent CN109574830B introduces a transformative approach to synthesizing the key intermediate D-7, shifting the paradigm from toxic cyanide-based chemistry to a greener, organometallic coupling strategy. This innovation not only addresses the urgent need for sustainable pharmaceutical intermediates manufacturing but also offers a robust solution for cost reduction in API manufacturing by streamlining the production workflow.
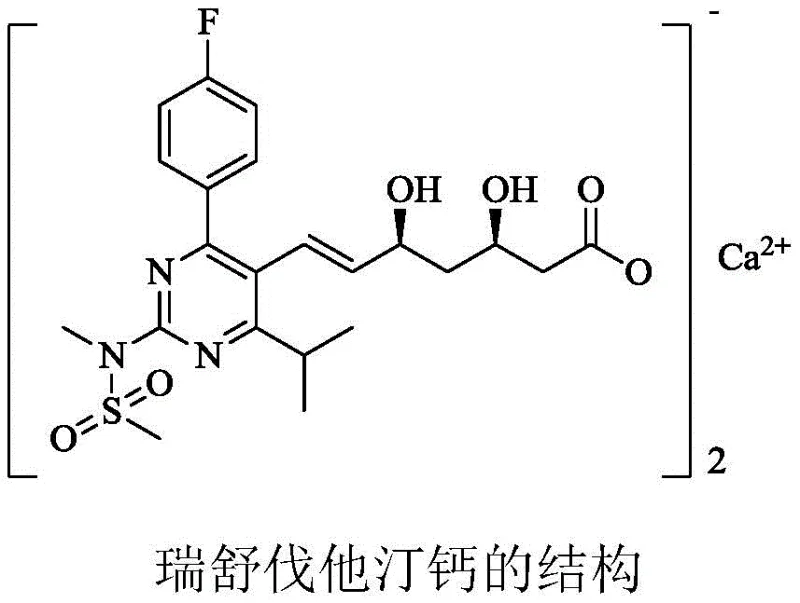
This technical insight report analyzes the proprietary methodology disclosed in the patent, highlighting how the novel intermediate, tert-butyl 6-acetoxy-3,5-dioxohexanoate (Compound I), serves as a pivotal junction for high-yield production. By leveraging advanced organometallic techniques and biocatalysis, this route effectively bypasses the limitations of classical Wittig and Blaise reactions. For procurement and supply chain leaders, understanding this shift is vital, as it represents a move towards reliable Rosuvastatin Calcium intermediate supplier partnerships that prioritize both regulatory compliance and operational efficiency. The following sections will dissect the mechanistic advantages and commercial implications of adopting this superior synthetic pathway.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of the Rosuvastatin side chain has relied heavily on methods that pose significant environmental, health, and safety (EHS) challenges. As illustrated in the prior art schemes, traditional routes often utilize the Blaise reaction involving zinc and highly toxic sodium cyanide, or the Asricon route which employs hazardous bromine and requires harsh reaction conditions. These conventional processes are characterized by lengthy synthetic sequences, often exceeding eight chemical steps, which inherently accumulate impurities and reduce overall material throughput. Furthermore, the reliance on strong bases like potassium bis(trimethylsilyl)amide in Wittig variations necessitates stringent moisture control and increases raw material costs substantially.
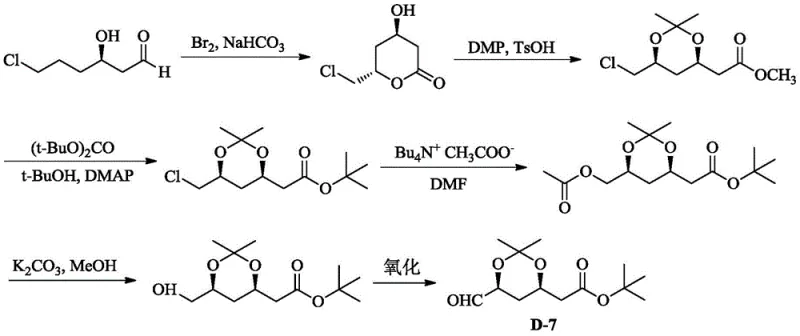
Beyond the safety hazards, the chemical inefficiencies of these legacy methods are profound. The conversion of chlorine atoms to acetoxyl groups in older protocols frequently demands temperatures above 100°C for extended periods. This thermal stress not only consumes excessive energy but also leads to product degradation, manifested as dark-colored crude materials that require extensive and costly purification efforts. The generation of nitrogenous wastewater from organic amine salts and the handling of cyanide waste create a massive burden on waste treatment facilities, driving up the operational expenditure for manufacturers. Consequently, the commercial scale-up of complex pharmaceutical intermediates via these routes is increasingly untenable in a regulatory environment that demands greener chemistry and lower carbon footprints.
The Novel Approach
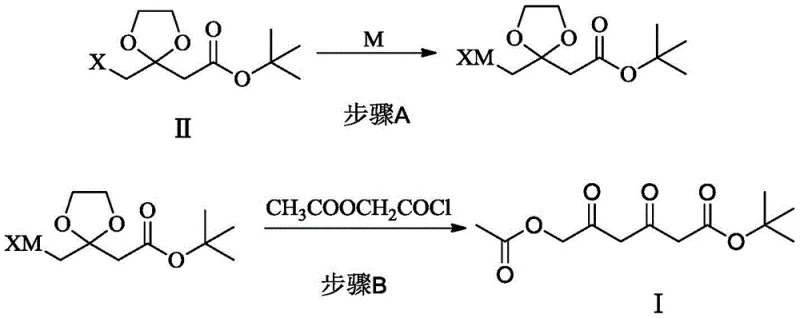
In stark contrast, the novel approach detailed in patent CN109574830B utilizes a concise, six-step sequence that fundamentally redesigns the construction of the carbon skeleton. The core innovation lies in the formation of Compound I through the coupling of a protected halogenated ester with acetoxyacetyl chloride. This strategy eliminates the need for sodium cyanide entirely, replacing it with safer organometallic reagents derived from magnesium or zinc. The reaction conditions are remarkably mild, typically proceeding at temperatures between -10°C and 60°C, which preserves the integrity of the sensitive functional groups and ensures a pale, high-quality product appearance without the need for aggressive decolorization steps.
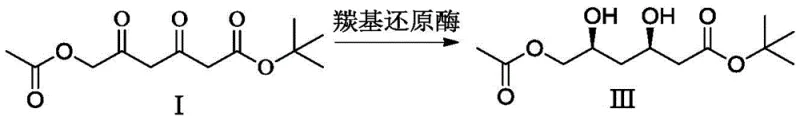
Moreover, this new methodology integrates seamlessly with modern biocatalytic technologies. The resulting diketo intermediate (Compound I) is an ideal substrate for stereoselective reduction using dicarbonyl reductases. This enzymatic step not only establishes the critical (3R,5S) stereochemistry with high precision but also operates under aqueous, neutral pH conditions, further reducing the solvent load and hazardous waste generation. By shortening the route and improving the total yield to over 82%, this approach offers a compelling value proposition for high-purity Rosuvastatin Calcium production, aligning perfectly with the industry's shift towards continuous processing and sustainable manufacturing practices.
Mechanistic Insights into Organometallic Coupling and Biocatalysis
The heart of this improved synthesis lies in the efficient generation of the carbon-carbon bonds via organometallic chemistry. The process begins with the activation of a halogenated acetal ester (Compound II) using magnesium or zinc metal in a refluxing ether-based solvent such as tetrahydrofuran (THF). This step generates a highly reactive Grignard or organozinc species in situ. Unlike traditional lithiation methods which can be pyrophoric and difficult to handle on a large scale, the magnesium and zinc reagents offer a balance of reactivity and stability that is conducive to safe industrial operations. The subsequent addition of this organometallic reagent to acetoxyacetyl chloride is carefully controlled, with the reagent added dropwise to the acid chloride to minimize side reactions such as enolization or self-condensation.
Following the formation of the diketo Compound I, the establishment of stereochemistry is achieved through a highly selective reduction process. The patent highlights the superiority of using dicarbonyl reductases over chemical reducing agents like sodium borohydride for this specific transformation. While chemical reduction can achieve the desired diol structure, it often requires chiral auxiliaries or low temperatures to maintain stereoselectivity, and may produce mixtures of diastereomers that complicate downstream purification. In contrast, the enzymatic reduction utilizes the inherent chirality of the protein active site to simultaneously reduce both carbonyl groups with exceptional specificity. This biocatalytic step, as shown in the reaction scheme below, proceeds at ambient temperatures (around 25°C) and neutral pH, exemplifying the principles of green chemistry while delivering the precise stereochemical configuration required for the final API.
How to Synthesize Rosuvastatin Calcium Intermediate Efficiently
The implementation of this synthesis route requires careful attention to reaction parameters, particularly during the organometallic formation and the quenching steps. The process is designed to be robust, tolerating slight variations in stoichiometry while maintaining high yields. For R&D teams looking to replicate or license this technology, the key lies in the preparation of the acetoxyacetyl chloride and the controlled addition of the metal reagent. The patent data suggests that maintaining the molar ratio of the organometallic precursor to the metal between 1:1 and 1:1.5 optimizes the conversion while minimizing unreacted starting materials. Detailed standard operating procedures for this specific transformation are critical for ensuring batch consistency.
- Formation of Organometallic Reagent: React a halogenated acetal ester (Compound II) with Magnesium or Zinc metal in a refluxing ether-based solvent like THF to generate the corresponding Grignard or Organozinc reagent.
- Coupling Reaction: Add the generated organometallic reagent dropwise to acetoxyacetyl chloride at low temperatures (-10 to 0°C) to form the diketo intermediate Compound I with high yield.
- Stereoselective Reduction: Convert Compound I to the diol Compound III using a dicarbonyl reductase enzyme or chiral borane reducing agents to establish the critical (3R,5S) stereochemistry required for the final API.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis route translates into tangible strategic advantages beyond mere technical elegance. The primary benefit is the drastic simplification of the supply chain risk profile. By eliminating sodium cyanide and bromine from the bill of materials, manufacturers remove the need for specialized hazardous material handling, storage, and disposal protocols. This reduction in EHS liability directly correlates to lower insurance premiums and reduced regulatory scrutiny, allowing for smoother operations across different geographical jurisdictions. Furthermore, the use of commodity chemicals like glycolic acid derivatives and common solvents ensures that raw material sourcing remains stable and immune to the volatility often seen with specialty reagents.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the significant shortening of the synthetic route. Reducing the step count from eight to six chemical reactions inherently lowers the consumption of solvents, reagents, and labor hours per kilogram of output. Additionally, the avoidance of high-temperature reactions reduces energy consumption for heating and cooling, while the improved product color and purity minimize the need for expensive chromatographic purification or recrystallization steps. The elimination of toxic cyanide also removes the substantial costs associated with specialized wastewater treatment and hazardous waste disposal, leading to substantial overall cost savings.
- Enhanced Supply Chain Reliability: The robustness of the organometallic coupling reaction ensures high reproducibility, which is essential for maintaining consistent supply to downstream API manufacturers. The mild reaction conditions reduce the likelihood of runaway reactions or equipment failure, thereby minimizing unplanned downtime. Moreover, the higher total yield (82.0% vs 70.9% in prior art) means that less raw material is required to produce the same amount of finished intermediate, effectively increasing the capacity of existing manufacturing assets without the need for capital-intensive expansion.
- Scalability and Environmental Compliance: As global regulations on pharmaceutical emissions tighten, the ability to demonstrate a 'green' synthesis pathway is a competitive differentiator. This process generates significantly less three-waste (waste water, waste gas, waste residue), particularly nitrogenous waste, simplifying compliance with environmental protection laws. The scalability is further enhanced by the use of standard reactor materials, as the mild conditions do not require exotic alloys to resist corrosion from harsh acids or bases, making technology transfer to contract manufacturing organizations (CMOs) faster and more cost-effective.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on the practical benefits of switching to this newer methodology.
Q: How does this new synthesis route improve EHS compliance compared to traditional methods?
A: The novel process completely eliminates the use of highly toxic sodium cyanide (NaCN) and avoids the use of hazardous bromine reagents found in conventional Blaise or Asricon routes. By utilizing mild organometallic coupling and enzymatic reduction, the process significantly lowers occupational health risks and reduces the cost and complexity of wastewater treatment associated with nitrogenous and heavy metal waste.
Q: What are the yield advantages of the organometallic coupling method for D-7 synthesis?
A: According to patent data, the total yield for synthesizing the key intermediate D-7 via this new route reaches approximately 82.0%, compared to roughly 70.9% for existing industrialized technologies. This improvement is driven by the avoidance of high-temperature, long-duration reactions that typically degrade product quality and color, resulting in a more efficient material throughput.
Q: Is this synthesis route suitable for large-scale commercial production?
A: Yes, the process is specifically designed for industrial scalability. It utilizes readily available raw materials such as glycolic acid derivatives and common solvents like THF. The reaction conditions are mild (often below 60°C), avoiding the extreme thermal stress that complicates reactor engineering in older methods, thereby ensuring consistent batch-to-batch quality and reliable supply continuity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Rosuvastatin Calcium Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to greener, more efficient synthetic routes is not just a regulatory requirement but a strategic imperative for maintaining competitiveness in the global pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in a GMP-compliant manufacturing environment. We operate stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of Rosuvastatin intermediate meets the highest international standards, providing our partners with the confidence needed for their own regulatory filings.
We invite forward-thinking pharmaceutical companies to collaborate with us to leverage this advanced technology for their supply chains. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis that quantifies the potential efficiencies for your specific volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your production of high-value statin intermediates is both economically viable and environmentally sustainable for the long term.
