Advanced Purification of Rosuvastatin Calcium for High-Purity Pharmaceutical Manufacturing
Advanced Purification of Rosuvastatin Calcium for High-Purity Pharmaceutical Manufacturing
The pharmaceutical industry continuously demands higher standards for Active Pharmaceutical Ingredients (APIs), particularly for cardiovascular medications like statins where purity directly correlates with patient safety and efficacy. A significant breakthrough in this domain is documented in patent CN102070537B, which details a novel refining method for Rosuvastatin Calcium. This technology addresses the critical limitations of traditional synthesis routes that often struggle with residual heavy metals, organic impurities, and inconsistent polymorphic forms. By leveraging a sophisticated combination of strong-acid ion exchange and chromatographic separation, this method ensures the production of Rosuvastatin Calcium with exceptional purity levels, often exceeding 99.5%. For global procurement teams and R&D directors, understanding this purification pathway is essential for securing a reliable supply chain of high-quality lipid-lowering agents.
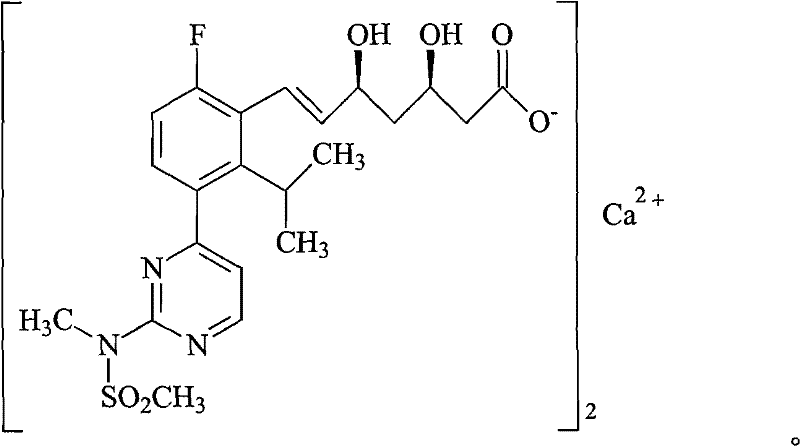
As illustrated in the structural diagram above, Rosuvastatin Calcium is a complex molecule featuring a fluorophenyl group, a pyrimidine ring, and a heptanoic acid side chain with specific stereochemistry at the 3R and 5S positions. The presence of the calcium ion (Ca2+) coordinated with two molecules of the acid form necessitates precise control during purification to maintain stoichiometric balance and crystal integrity. The patented process described in CN102070537B offers a robust solution to the challenges posed by this complex structure, moving away from simple precipitation methods towards a more controlled ion-exchange mechanism that significantly enhances the overall quality profile of the final API intermediate.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional methods for obtaining Rosuvastatin Calcium often rely heavily on direct synthesis followed by multiple recrystallization steps to achieve acceptable purity. However, these conventional approaches suffer from several inherent drawbacks that impact both cost and quality. Firstly, recrystallization is frequently inefficient at removing structurally similar impurities or trace heavy metals introduced during the catalytic synthesis stages. Secondly, the repeated use of organic solvents in recrystallization cycles increases environmental burdens and operational costs, creating significant waste disposal challenges for manufacturers. Furthermore, conventional methods often result in variable yields, with substantial product loss occurring during the filtration and washing stages, thereby driving up the cost per kilogram of the final active ingredient. These inefficiencies make it difficult for suppliers to guarantee consistent batch-to-batch quality, a critical requirement for regulatory compliance in major markets like the US and Europe.
The Novel Approach
In contrast, the novel approach outlined in the patent utilizes a targeted ion-exchange strategy that fundamentally alters the purification landscape. Instead of relying solely on solubility differences, this method exploits the ionic nature of the Rosuvastatin molecule. The process begins by converting the calcium salt into its free acid form using a strong-acid ion exchange resin, which effectively traps the active molecule while allowing many neutral organic impurities to pass through or be adsorbed onto the resin matrix. Subsequently, the pure acid form is eluted and converted back into the calcium salt using specific calcium sources like calcium hydroxide or calcium bicarbonate. This chemical transformation is highly selective and minimizes the introduction of new contaminants. Finally, a polishing step using chromatographic separation ensures that any remaining trace impurities are removed, resulting in a product that meets the most stringent pharmacopeial standards for potency and purity.
Mechanistic Insights into Ion-Exchange and Chromatographic Purification
The core of this technological advancement lies in the specific interaction between the Rosuvastatin molecule and the strong-acid cationic exchange resin, typically a styrene-divinylbenzene copolymer sulfonated to provide high acidity. When an aqueous solution of crude Rosuvastatin Calcium is passed through the resin bed, a proton exchange occurs where hydrogen ions from the resin displace the calcium ions bound to the Rosuvastatin acid moieties. This conversion to the free acid form is crucial because the acid exhibits different adsorption characteristics compared to the salt, allowing the resin to act not just as an exchanger but also as an adsorbent for non-electrolytic impurities such as residual solvents, unreacted starting materials, and colored by-products. The macroporous structure of resins like the D001 type provides a large surface area that enhances this adsorption capacity, effectively decolorizing the solution and removing heavy metal contaminants that might otherwise persist through standard filtration.
Following the adsorption phase, the elution step is meticulously controlled to ensure high recovery and purity. By using an aqueous solution of calcium hydroxide or calcium bicarbonate, the Rosuvastatin acid is neutralized in situ, reforming the calcium salt which is then soluble in the eluent. The use of calcium bicarbonate is particularly ingenious as the reaction generates carbon dioxide gas, which escapes the system, driving the reaction equilibrium towards completion without leaving behind extraneous anions that could contaminate the product. The flow rate of the eluent is a critical parameter, typically maintained between 0.5 to 3.0 ml/min, to ensure sufficient contact time for complete conversion. The final chromatographic separation on silica gel further refines the product, utilizing a mobile phase of methanol and calcium chloride water to separate the target molecule from any closely related analogues, ensuring the final dried product possesses the requisite physicochemical properties for tablet formulation.
How to Synthesize Rosuvastatin Calcium Efficiently
The implementation of this purification protocol requires precise adherence to the three-stage process defined in the patent to maximize yield and purity. Operators must carefully manage the pH levels during the ion exchange and elution phases, as deviations can lead to incomplete conversion or product degradation. The following guide outlines the standardized operational procedure derived from the patent examples, serving as a foundational reference for process engineers looking to adopt this technology for commercial production. Detailed parameters regarding resin loading, eluent concentration, and column dimensions are critical for scaling this from laboratory benchtop to pilot plant operations.
- Adsorb Rosuvastatin Calcium onto a strong-acid ion exchange resin to convert it into Rosuvastatin acid form while removing impurities.
- Elute the resin using an aqueous solution of calcium hydroxide or calcium bicarbonate to regenerate the calcium salt form.
- Perform final separation and purification using a silica gel chromatographic column followed by vacuum drying.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this ion-exchange purification technology translates into tangible strategic advantages beyond mere technical specifications. The primary benefit is the significant reduction in manufacturing costs driven by the elimination of multiple solvent-intensive recrystallization cycles. By streamlining the purification into a continuous or semi-continuous ion-exchange process, manufacturers can drastically reduce solvent consumption and waste treatment expenses, leading to a more sustainable and cost-effective production model. Furthermore, the high yield reported in the patent examples, often exceeding 85%, ensures that raw material utilization is optimized, directly impacting the bottom line and allowing for more competitive pricing structures in the global market.
- Cost Reduction in Manufacturing: The transition to an ion-exchange based purification route eliminates the need for expensive and hazardous organic solvents typically used in repeated recrystallizations. This shift not only lowers the direct cost of goods sold (COGS) by reducing solvent purchase requirements but also significantly decreases the operational expenditure associated with solvent recovery and hazardous waste disposal. Additionally, the ability to regenerate the ion exchange resin for multiple cycles further amortizes the cost of consumables, providing a long-term economic advantage over traditional batch purification methods that rely on single-use filtration media.
- Enhanced Supply Chain Reliability: The robustness of this purification method enhances supply chain stability by reducing the risk of batch failures due to purity issues. Since the process effectively removes heavy metals and organic impurities that are common causes of regulatory rejection, manufacturers can maintain a consistent flow of compliant material to their clients. The use of commercially available resins and standard chromatographic equipment means that the supply chain is not dependent on exotic or hard-to-source catalysts, mitigating the risk of supply disruptions and ensuring that production schedules can be met reliably even during periods of high market demand.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this method is inherently superior for large-scale production. Ion exchange columns can be easily scaled up by increasing bed volume or operating in parallel, allowing for seamless transition from pilot batches to multi-ton annual production capacities. The aqueous nature of the primary purification steps reduces the volatile organic compound (VOC) emissions associated with the process, aligning with increasingly strict environmental regulations globally. This compliance reduces the regulatory burden on the manufacturer and ensures long-term operational continuity without the threat of environmental shutdowns.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and sourcing of high-purity Rosuvastatin Calcium. These insights are derived directly from the technical specifications and experimental data provided in the underlying patent literature, offering clarity on the feasibility and benefits of this specific manufacturing route. Understanding these details is crucial for partners evaluating the technical capability of their potential suppliers.
Q: How does the ion exchange method improve Rosuvastatin Calcium purity compared to traditional crystallization?
A: The ion exchange method utilizes strong-acid resins that not only facilitate ionic conversion but also adsorb non-electrolytic impurities and colored by-products, achieving purity levels exceeding 99.5% which is difficult with standard recrystallization.
Q: What are the advantages of using calcium hydroxide or calcium bicarbonate for elution?
A: Using these calcium sources allows for the direct regeneration of the calcium salt without introducing new counter-ions; additionally, the release of carbon dioxide gas during the reaction with bicarbonate helps drive the equilibrium and prevents impurity entrapment.
Q: Is this purification process suitable for large-scale industrial production?
A: Yes, the process employs fixed-bed or continuous ion exchange columns and standard chromatographic techniques, which are inherently scalable and utilize commercially available resins, making it highly suitable for industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Rosuvastatin Calcium Supplier
At NINGBO INNO PHARMCHEM, we recognize that the quality of the final medication depends entirely on the integrity of the intermediate supply. Our technical team has extensively analyzed advanced purification technologies, including the ion-exchange methods described in CN102070537B, to ensure our production capabilities meet the highest global standards. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that whether you require clinical trial materials or full-scale commercial volumes, our facility is equipped to deliver. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that utilize state-of-the-art HPLC and spectroscopic analysis to verify every batch.
We invite pharmaceutical partners to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements. By leveraging our optimized purification processes, we can help you reduce the overall cost of your lipid-lowering drug portfolio without compromising on quality. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our advanced manufacturing capabilities can support your long-term supply chain goals.
