Advanced BDTT-im-Cd Cadmium Complex Dye Sensitizer for Next-Generation Solar Cell Manufacturing
Advanced BDTT-im-Cd Cadmium Complex Dye Sensitizer for Next-Generation Solar Cell Manufacturing
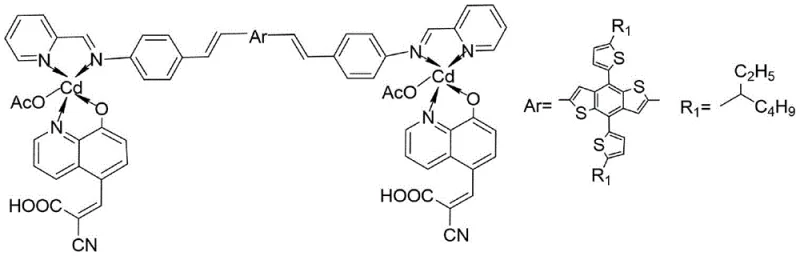
The rapid evolution of renewable energy technologies has placed significant emphasis on the development of high-performance materials for dye-sensitized solar cells (DSSCs). A pivotal advancement in this field is documented in patent CN111943965B, which introduces a novel pyridine imine derivative and 8-hydroxyquinoline derivative cadmium complex dye sensitizer, known as BDTT-im-Cd. This innovative compound utilizes a sophisticated D(-A-π-A)2 molecular architecture, designed to optimize intramolecular charge transfer and broaden the absorption spectrum. By integrating a benzodithiophene (BDTT) unit as a potent electron donor and a cadmium complex as an auxiliary electron acceptor, this material addresses critical limitations in current photovoltaic efficiency. For R&D directors and procurement specialists seeking reliable electronic chemical supplier partnerships, understanding the synthesis and application of such high-purity dye sensitizers is essential for driving the next generation of cost-effective solar energy solutions.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional dye sensitizers often rely on simpler D-π-A configurations which, while functional, frequently suffer from narrow absorption bands and insufficient thermal stability for long-term commercial deployment. Many organic dyes degrade rapidly under the thermal stress encountered during device operation or outdoor exposure, leading to a precipitous drop in power conversion efficiency over time. Furthermore, conventional synthesis routes for complex organometallic dyes can be plagued by low yields, difficult purification processes, and the use of hazardous solvents that complicate waste management. These factors collectively increase the cost reduction in display & optoelectronic materials manufacturing barriers, making it challenging to scale production without compromising on the purity required for high-performance electronic applications. The inability to effectively balance electron donor and acceptor strengths in older designs also limits the open-circuit voltage and short-circuit current density achievable in the final device.
The Novel Approach
The methodology outlined in the patent presents a transformative solution by employing a D(-A-π-A)2 structural motif that significantly enhances electron transport capabilities. By introducing the cadmium complex as a secondary electron acceptor alongside the primary anchoring group, the energy gap between the LUMO and HOMO levels is effectively reduced, facilitating broader light absorption and more efficient charge injection into the semiconductor substrate. This design not only improves the photoelectric conversion efficiency to a remarkable 9.13 percent but also imparts exceptional thermal stability, with decomposition temperatures exceeding 300 degrees Celsius. The use of a Heck coupling reaction to link the BDTT donor unit ensures a robust conjugated system that maintains structural integrity under operational stress. This approach represents a significant leap forward for manufacturers aiming for the commercial scale-up of complex organometallic dyes, offering a pathway to more durable and efficient photovoltaic modules.
Mechanistic Insights into Heck Coupling and Coordination Chemistry
The synthesis of BDTT-im-Cd is underpinned by precise coordination chemistry and transition metal catalysis, specifically leveraging the versatility of the Heck coupling reaction. In the final synthetic step, the cadmium-containing intermediate, which acts as the electron acceptor and anchoring unit, is coupled with the electron-rich benzodithiophene (BDTT) derivative. This reaction is catalyzed by palladium acetate in the presence of tris(2-tolyl)phosphine as a ligand and triethylamine as a base, typically conducted in DMF solvent at temperatures above 90 degrees Celsius for over 48 hours. The mechanism involves the oxidative addition of the aryl halide on the BDTT unit to the palladium center, followed by migratory insertion of the alkene from the cadmium complex and subsequent beta-hydride elimination to form the new carbon-carbon bond. This process creates an extended pi-conjugated system that is crucial for efficient electron delocalization across the molecule.
Furthermore, the coordination environment of the cadmium ion plays a pivotal role in defining the electronic properties of the dye. The cadmium center is coordinated by nitrogen atoms from the pyridine imine ligand and oxygen atoms from the 8-hydroxyquinoline derivative, forming a stable chelate structure. This coordination not only stabilizes the metal center against leaching or degradation but also tunes the energy levels of the frontier orbitals to match the conduction band of the titanium dioxide semiconductor commonly used in DSSCs. The auxiliary acceptor nature of the cadmium complex facilitates a cascade of electron transfer from the BDTT donor through the pi-bridge to the anchoring group, minimizing charge recombination losses. Understanding these mechanistic details is vital for quality control teams ensuring the batch-to-batch consistency required for high-purity dye sensitizer production.
How to Synthesize BDTT-im-Cd Efficiently
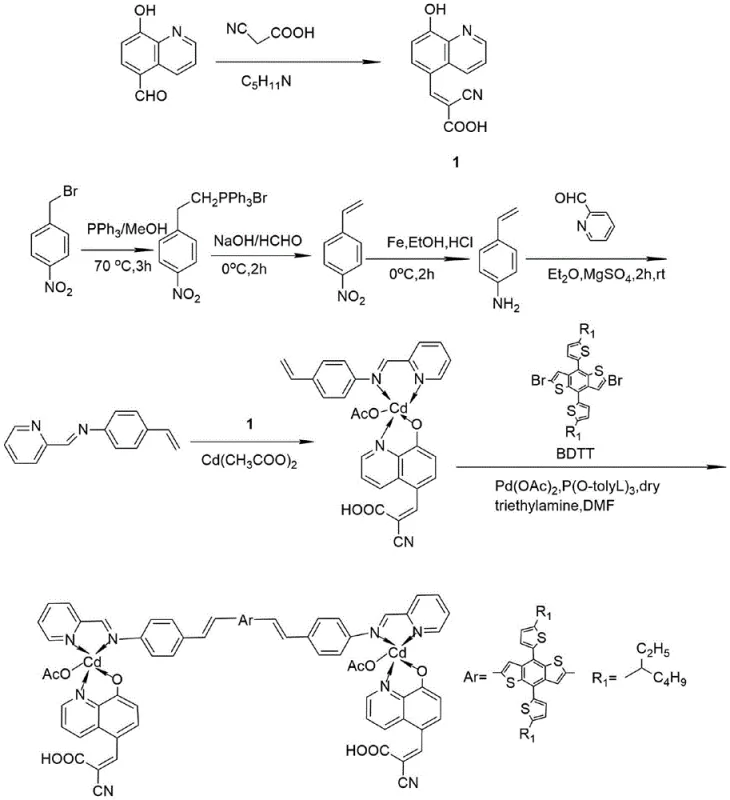
The preparation of this advanced dye sensitizer involves a multi-step sequence that requires careful control of reaction conditions to maximize yield and purity. The process begins with the synthesis of key ligands, including the 2-cyano-3-(8-hydroxyquinolin-5-yl)acrylic acid and the vinyl-functionalized pyridine imine, before proceeding to metal complexation and the final coupling. Each step demands rigorous purification, often involving recrystallization and column chromatography, to remove trace metal catalysts and organic impurities that could act as recombination centers in the solar cell. The detailed standardized synthesis steps provided below outline the specific reagents, stoichiometry, and thermal profiles necessary to replicate the high performance reported in the patent literature.
- Prepare the 2-cyano-3-(8-hydroxyquinolin-5-yl)acrylic acid ligand via Knoevenagel condensation of 5-formyl-8-hydroxyquinoline and cyanoacetic acid.
- Synthesize the pyridine imine ligand by reacting 4-vinylaniline with 2-pyridinecarboxaldehyde, followed by complexation with cadmium acetate.
- Perform the final Heck coupling reaction between the cadmium complex intermediate and the BDTT donor derivative using a palladium catalyst at 90°C.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of the BDTT-im-Cd synthesis route offers distinct strategic advantages rooted in process robustness and material stability. The reliance on well-established reactions such as the Wittig reaction, Knoevenagel condensation, and Heck coupling means that the manufacturing process can be easily scaled using existing infrastructure without requiring exotic or hard-to-source equipment. The starting materials, such as 5-formyl-8-hydroxyquinoline and various styrene derivatives, are commercially available fine chemicals, reducing the risk of supply chain bottlenecks associated with proprietary or custom-synthesized precursors. This accessibility translates directly into enhanced supply chain reliability, ensuring that production schedules can be met consistently even in fluctuating market conditions.
- Cost Reduction in Manufacturing: The synthetic pathway eliminates the need for extremely low-temperature cryogenic conditions or ultra-high vacuum systems, which are often cost-prohibitive in large-scale chemical manufacturing. By utilizing standard reflux conditions and common solvents like ethanol, methanol, and DMF, the operational expenditure associated with energy consumption and solvent recovery is significantly minimized. Additionally, the high thermal stability of the final product reduces waste generated from material degradation during storage and transport, further contributing to substantial cost savings. The efficient use of catalysts, particularly the palladium system in the final step, allows for optimized loading rates that balance reaction speed with economic feasibility.
- Enhanced Supply Chain Reliability: The modular nature of the synthesis allows for the stockpiling of key intermediates, such as the cadmium complex im-Cd, which can be produced in bulk and stored for extended periods due to its stability. This decoupling of synthesis stages provides flexibility in production planning, enabling manufacturers to respond rapidly to surges in demand for photovoltaic materials. Furthermore, the use of robust chemical bonds in the final D(-A-π-A)2 structure ensures that the product maintains its integrity during shipping and handling, reducing the incidence of returns or claims related to product quality. This reliability is critical for maintaining long-term contracts with downstream solar module manufacturers.
- Scalability and Environmental Compliance: The process is amenable to continuous flow chemistry adaptations, which can further improve safety profiles by minimizing the inventory of reactive intermediates at any given time. Waste streams are primarily composed of standard organic solvents and salt byproducts, which can be managed through established recycling and treatment protocols, aligning with increasingly stringent environmental regulations. The high yield of the final Heck coupling step, combined with effective purification methods like recrystallization, ensures that the final product meets the rigorous purity specifications required for electronic applications without generating excessive hazardous waste. This scalability positions the technology as a viable candidate for gigawatt-scale solar manufacturing initiatives.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of the BDTT-im-Cd dye sensitizer. These insights are derived directly from the experimental data and structural analysis provided in the patent documentation, offering clarity on performance metrics and synthesis feasibility. Understanding these aspects is crucial for stakeholders evaluating the integration of this material into their existing product lines or research pipelines.
Q: What is the photoelectric conversion efficiency of the BDTT-im-Cd dye sensitizer?
A: According to patent CN111943965B, the BDTT-im-Cd dye sensitizer achieves a Photoelectric Conversion Efficiency (PCE) of 9.13% when tested in dye-sensitized solar cells, demonstrating superior performance compared to many conventional dyes.
Q: How does the thermal stability of BDTT-im-Cd compare to standard organic dyes?
A: The BDTT-im-Cd complex exhibits exceptional thermal stability with a decomposition temperature exceeding 300°C. This high thermal resistance ensures long-term operational stability and durability in photovoltaic devices under varying environmental conditions.
Q: What is the key reaction mechanism used to assemble the final dye molecule?
A: The final assembly of the BDTT-im-Cd molecule relies on a Palladium-catalyzed Heck coupling reaction. This robust cross-coupling method efficiently links the electron-rich BDTT donor unit with the electron-deficient cadmium complex acceptor unit.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable BDTT-im-Cd Supplier
As the global demand for high-efficiency renewable energy materials continues to surge, partnering with an experienced chemical manufacturer is paramount for success. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of BDTT-im-Cd delivered meets the exacting standards required for high-performance dye-sensitized solar cells. We understand the critical nature of impurity profiles in electronic materials and employ advanced analytical techniques to ensure product integrity.
We invite you to collaborate with our technical team to explore how this advanced cadmium complex dye can enhance your photovoltaic product portfolio. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to support your R&D and manufacturing goals, ensuring a seamless transition from laboratory innovation to industrial reality.
