Advanced BDTT-im-Cd Cadmium Complex Dye Sensitizers for High-Efficiency Solar Applications
Advanced BDTT-im-Cd Cadmium Complex Dye Sensitizers for High-Efficiency Solar Applications
The landscape of renewable energy materials is undergoing a significant transformation with the advent of novel molecular architectures designed for dye-sensitized solar cells (DSSCs). A pivotal development in this sector is detailed in patent CN111943965A, which discloses a sophisticated D(-A-π-A)2-type pyridineimine derivative combined with an 8-hydroxyquinoline derivative cadmium complex, known as BDTT-im-Cd. This innovative sensitizer leverages a unique structural design where a benzodithiophene dithiophene (BDTT) unit functions as a potent electron donor, coupled with a cadmium complex acting as an auxiliary electron acceptor. The integration of these functional groups through a robust Heck coupling reaction results in a material that not only broadens the light absorption spectrum but also enhances intramolecular charge transfer efficiency. For industrial stakeholders, this represents a critical advancement in creating reliable optoelectronic material supplier networks capable of delivering high-performance components for next-generation photovoltaic devices.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional dye sensitizers often rely on simpler D-π-A (Donor-π-Acceptor) configurations, which, while effective, frequently encounter bottlenecks regarding spectral coverage and charge recombination rates. In many legacy systems, the balance between the electron-pushing capability of the donor and the electron-withdrawing strength of the acceptor is difficult to optimize without compromising molecular stability. Conventional organic dyes may suffer from narrow absorption bands, limiting their ability to harvest photons across the visible and near-infrared regions effectively. Furthermore, the lack of robust anchoring groups or auxiliary acceptors can lead to inefficient electron injection into the semiconductor conduction band, thereby capping the overall power conversion efficiency. These structural deficiencies often necessitate complex post-synthesis modifications or the use of expensive co-adsorbents to mitigate aggregation, driving up the cost reduction in dye-sensitized solar cell manufacturing efforts.
The Novel Approach
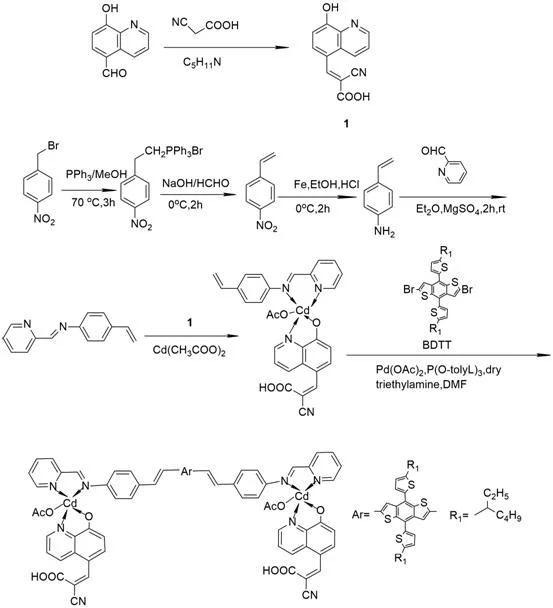
The methodology outlined in the patent introduces a paradigm shift by employing a D(-A-π-A)2 architecture, which strategically incorporates an auxiliary electron acceptor to modulate the energy levels of the dye. By utilizing a cadmium complex as this auxiliary acceptor alongside the primary cyanoacrylic acid anchor, the system effectively lowers the LUMO energy level, facilitating a more favorable energy gap for electron injection. The synthesis pathway, illustrated below, demonstrates the modular assembly of this complex, starting from simple precursors like 5-formyl-8-hydroxyquinoline and progressing through a series of high-yield transformations. This approach not only enhances the molar extinction coefficient but also improves the thermal robustness of the final product, addressing the stability issues prevalent in earlier generations of sensitizers.
Mechanistic Insights into Heck-Coupled Cadmium Coordination
The core of this technology lies in the precise orchestration of coordination chemistry and cross-coupling catalysis. The formation of the BDTT-im-Cd complex involves a critical Heck coupling reaction, where a palladium catalyst facilitates the carbon-carbon bond formation between the vinyl group of the cadmium-ligand precursor and the brominated positions on the BDTT core. This step is paramount for establishing the conjugated π-system that spans the entire molecule, allowing for delocalized electron density that is essential for rapid charge transport. The cadmium center itself plays a dual role; it acts as a structural node that rigidifies the molecular geometry, reducing non-radiative decay pathways, and simultaneously functions as an electronic bridge that accepts electron density from the BDTT donor before passing it to the anchoring group. This synergistic interaction ensures that the excited state lifetime is sufficiently long to allow for efficient electron injection into the TiO2 semiconductor matrix.
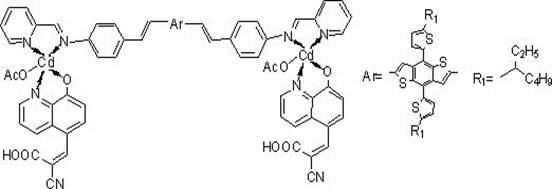
From an impurity control perspective, the synthetic route is designed to minimize side reactions that could generate non-emissive traps or recombination centers. The use of specific recrystallization steps, such as the purification of the 2-cyano-3-(8-hydroxyquinolin-5-yl) acrylic acid intermediate using ethanol, ensures that the final high-purity cadmium complex dye meets stringent specifications. The coordination of the cadmium ion with the nitrogen and oxygen donors of the ligands creates a stable chelate ring system that resists hydrolysis under operational conditions. This stability is further reinforced by the steric bulk provided by the BDTT unit, which shields the metal center from potential nucleophilic attacks by electrolyte components. Consequently, the resulting material exhibits a thermal decomposition temperature exceeding 300°C, a metric that is crucial for ensuring long-term device reliability in varying environmental conditions.
How to Synthesize BDTT-im-Cd Efficiently
The production of this advanced sensitizer follows a logical, stepwise progression that balances yield with purity, making it amenable to scale-up. The process begins with the Knoevenagel condensation to form the anchoring group, followed by the construction of the π-bridge and donor units. Each intermediate is rigorously purified to prevent the carryover of impurities that could poison the palladium catalyst in the final coupling step. The detailed standardized synthesis steps are provided in the guide below, offering a clear roadmap for technical teams aiming to replicate or adapt this chemistry for commercial production.
- Synthesize the 8-hydroxyquinoline anchor via Knoevenagel condensation of 5-formyl-8-hydroxyquinoline and cyanoacetic acid.
- Prepare the vinyl-aniline ligand through Wittig olefination of nitrobenzyl bromide followed by iron-mediated reduction.
- Form the Cd(II) intermediate complex and finalize the BDTT-im-Cd structure via Palladium-catalyzed Heck coupling with the BDTT donor.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of the BDTT-im-Cd synthesis route offers tangible strategic benefits beyond mere performance metrics. The reliance on commercially available starting materials, such as nitrobenzyl bromide and 8-hydroxyquinoline derivatives, mitigates the risk of raw material shortages that often plague specialty chemical supply chains. Furthermore, the reaction conditions employed, such as the use of standard solvents like DMF and ethanol, align well with existing infrastructure in fine chemical manufacturing plants, reducing the need for capital-intensive equipment upgrades. This compatibility significantly streamlines the transition from laboratory benchtop to pilot plant, ensuring a smoother ramp-up phase for new product introductions.
- Cost Reduction in Manufacturing: The synthetic strategy eliminates the need for exotic or prohibitively expensive catalysts in the early stages, relying instead on well-established organic transformations like the Wittig reaction and iron-mediated reductions. By optimizing the stoichiometry of the Heck coupling step and utilizing efficient purification methods like recrystallization rather than preparative HPLC, the overall cost of goods sold can be substantially lowered. Additionally, the high thermal stability of the final product reduces waste associated with material degradation during storage and transport, contributing to significant cost savings over the product lifecycle.
- Enhanced Supply Chain Reliability: The modular nature of the synthesis allows for the decoupling of intermediate production, meaning that key building blocks like the vinyl-aniline ligand can be stockpiled independently of the final coupling reaction. This flexibility enhances supply chain resilience, allowing manufacturers to buffer against fluctuations in demand or temporary disruptions in the availability of the palladium catalyst. The robustness of the intermediates also simplifies logistics, as they do not require the same stringent temperature controls as some fragile organometallic species, thereby reducing lead time for high-purity optoelectronic materials.
- Scalability and Environmental Compliance: The process avoids the use of highly toxic reagents where possible, substituting them with safer alternatives like iron powder for reductions, which aligns with increasingly strict environmental regulations. The ability to perform the final coupling in polar aprotic solvents that are easily recoverable supports sustainable manufacturing practices. Moreover, the high yield of the final Heck coupling step ensures that atom economy is maximized, minimizing the generation of hazardous waste streams and simplifying the permitting process for commercial scale-up of complex organic sensitizers.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this dye sensitizer technology. These insights are derived directly from the experimental data and structural analysis presented in the patent documentation, providing a factual basis for decision-making.
Q: What is the photoelectric conversion efficiency of the BDTT-im-Cd dye?
A: According to patent CN111943965A, the BDTT-im-Cd dye sensitizer achieves a photoelectric conversion efficiency (PCE) of 9.13% in dye-sensitized solar cells.
Q: How thermally stable is the BDTT-im-Cd complex?
A: The complex exhibits excellent thermal stability with a decomposition temperature exceeding 300°C, making it suitable for rigorous photovoltaic applications.
Q: What represents the key structural innovation in this dye?
A: The dye utilizes a D(-A-π-A)2 configuration where a benzodithiophene (BDTT) unit acts as the electron donor and a cadmium complex serves as the auxiliary electron acceptor.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable BDTT-im-Cd Supplier
As the demand for high-efficiency photovoltaic materials continues to surge, partnering with an experienced CDMO is essential for navigating the complexities of scale-up. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our facilities are equipped with rigorous QC labs capable of verifying the stringent purity specifications required for electronic grade chemicals, guaranteeing that every batch of BDTT-im-Cd performs identically to the patent benchmarks. We understand the critical nature of batch-to-batch reproducibility in the solar industry and have implemented robust process controls to maintain the highest standards of quality.
We invite you to engage with our technical procurement team to discuss how we can tailor this synthesis to your specific volume requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper visibility into how our optimized processes can reduce your overall material costs without compromising performance. We encourage potential partners to contact us for specific COA data and route feasibility assessments, allowing you to validate the technical viability of integrating BDTT-im-Cd into your next-generation solar cell designs with confidence.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
