Revolutionizing Atorvastatin Calcium Production with Green Organometallic Chemistry
The global demand for lipid-lowering agents continues to drive innovation in the synthesis of Atorvastatin Calcium, the active ingredient in the blockbuster drug Lipitor. Patent CN109503542B introduces a groundbreaking methodology for synthesizing key intermediates, specifically targeting the side chain precursor ATS-9, which has historically been a bottleneck in manufacturing due to safety and cost concerns. This invention proposes a green, environmentally friendly synthesis route that fundamentally alters the chemical landscape of statin production by eliminating highly toxic reagents. The structural complexity of Atorvastatin Calcium Trihydrate requires precise stereochemical control, as depicted in the molecular architecture below, necessitating robust and scalable intermediate synthesis strategies.
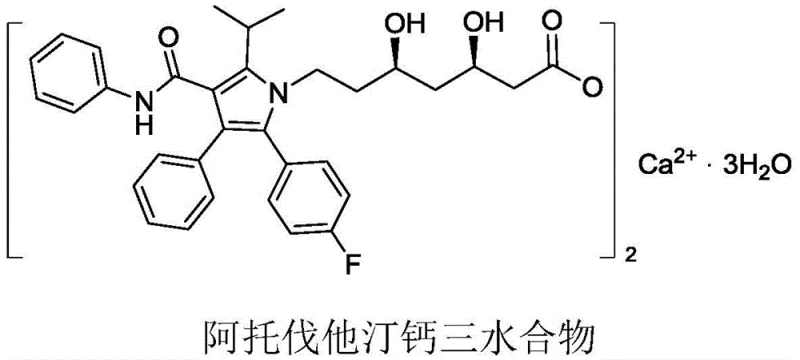
As a leading entity in fine chemical manufacturing, understanding the shift from hazardous legacy processes to sustainable organometallic coupling is critical for maintaining supply chain resilience. The patent details a novel pathway that not only enhances purity profiles but also aligns with increasingly stringent global environmental regulations. For procurement and technical teams, this represents a significant opportunity to optimize the cost of goods sold (COGS) while mitigating regulatory risks associated with cyanide and lithium waste streams. The following analysis dissects the technical merits and commercial implications of this patented technology for the pharmaceutical industry.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
The historical industrial standard for synthesizing the Atorvastatin side chain, often referred to as the Paal-Knorr pyrrole route, relies heavily on the intermediate ATS-9. However, the prevailing synthesis methods for ATS-9, such as those disclosed in earlier patents like WO9932434, are fraught with significant operational hazards and economic inefficiencies. As illustrated in the conventional scheme below, the process typically necessitates the use of sodium cyanide (NaCN), a substance of extreme toxicity that poses severe health risks to operators and creates complex wastewater treatment challenges. Furthermore, these legacy routes depend on Lithium Diisopropylamide (LDA), a strong base that requires preparation from expensive lithium metal and diisopropylamine.
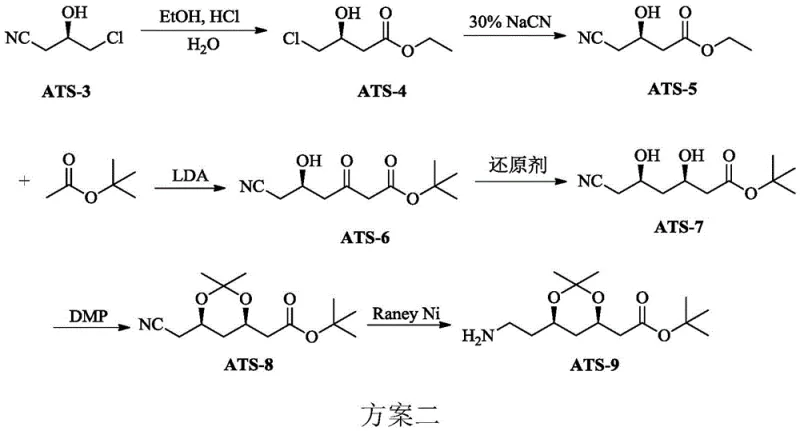
The reliance on LDA introduces substantial logistical burdens, including the need for cryogenic cooling with liquid nitrogen to manage the exothermic nature of its formation and usage. This requirement for deep freezing temperatures (often below -40°C) results in excessive energy consumption and complicates the engineering design of large-scale reactors. Additionally, the quenching of lithium-based reactions generates vast quantities of inorganic salt waste, such as lithium bromide and chloride, alongside organic amine salts, which significantly inflates the cost of three-waste treatment. The combination of high toxicity, extreme thermal requirements, and expensive raw materials makes the conventional approach increasingly untenable for modern, sustainability-focused manufacturing facilities.
The Novel Approach
In stark contrast to the hazardous legacy methods, the invention disclosed in CN109503542B presents a streamlined and safer synthetic strategy that circumvents the use of cyanides and organolithium reagents entirely. The core innovation lies in the utilization of readily available magnesium or zinc metals to generate organometallic intermediates under mild reflux conditions, eliminating the need for energy-intensive cryogenic cooling. This shift not only reduces the direct cost of raw materials by substituting expensive lithium with abundant magnesium or zinc but also dramatically simplifies the reactor infrastructure requirements. The process operates at near-ambient or moderate temperatures, thereby enhancing process safety and reducing the risk of thermal runaway incidents common with pyrophoric lithium reagents.
Furthermore, the new route shortens the overall synthetic sequence, improving the total yield of the critical intermediate ATS-8 to approximately 79.5%, compared to roughly 67.7% in existing mainstream processes. By avoiding the generation of heavy metal waste and toxic cyanide byproducts, the environmental footprint of the manufacturing process is substantially reduced, aligning with green chemistry principles. This approach ensures a more reliable supply of high-purity intermediates, as the simplified workflow minimizes the potential for impurity carryover and batch-to-batch variability, offering a distinct competitive advantage for generic drug manufacturers seeking cost-effective and compliant production methods.
Mechanistic Insights into Organometallic Coupling and Stereoselective Reduction
The heart of this novel synthesis lies in the construction of the carbon skeleton via an organometallic coupling reaction, specifically transforming a protected halogenated ester (Formula II) into a tricarbonyl precursor (Formula I). The mechanism initiates with the formation of a Grignard or organozinc reagent from a halogenated dioxolane derivative. Unlike the violent and uncontrollable nature of lithiation, the insertion of magnesium or zinc into the carbon-halogen bond proceeds smoothly in etheral solvents like tetrahydrofuran (THF). This organometallic species then acts as a nucleophile, attacking an electrophilic cyano-containing substrate, such as malononitrile or cyanoacetate derivatives. This carbon-carbon bond-forming step is pivotal, as it establishes the linear chain required for the eventual statin pharmacophore without introducing toxic cyanide ions into the reaction matrix.
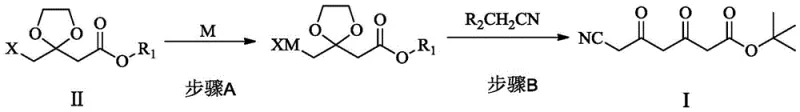
Following the coupling, the resulting tricarbonyl compound undergoes a critical stereoselective reduction to establish the requisite (3R, 5R) chirality. The patent highlights the versatility of this step, allowing for either chemical reduction using chiral boranes or, more preferably, enzymatic reduction using carbonyl reductases. The enzymatic approach is particularly noteworthy for its high stereospecificity and environmental benignity, operating efficiently in aqueous buffer systems with cofactor regeneration. This biocatalytic step ensures the formation of the dihydroxy intermediate (ATS-7) with high optical purity, which is subsequently protected as an acetonide. The final conversion to the amine (ATS-9) is achieved through catalytic hydrogenation, a robust and scalable unit operation. This mechanistic progression from organometallic coupling to biocatalysis represents a sophisticated integration of classical and modern synthetic techniques to achieve superior process metrics.
How to Synthesize Atorvastatin Intermediate Efficiently
The synthesis of the key intermediate involves a sequence of protection, organometallic coupling, reduction, and deprotection steps that are optimized for high yield and purity. The process begins with the protection of a keto-ester using ethylene glycol to prevent side reactions during the metal insertion phase. Subsequently, the organometallic reagent is generated in situ and reacted with a cyano-source to extend the carbon chain. The detailed operational parameters, including solvent choices, temperature profiles, and workup procedures, are critical for maximizing the efficiency of this transformation. For a comprehensive understanding of the specific reaction conditions and stoichiometry required to replicate this high-yielding process, please refer to the standardized synthesis guide provided below.
- Protect the ketone group of the starting halogenated ester using ethylene glycol and an acid catalyst to form the dioxolane intermediate.
- Generate an organometallic reagent (Grignard or Organozinc) by reacting the protected halide with Magnesium or Zinc in THF.
- Couple the organometallic species with a cyano-containing electrophile (like malononitrile or cyanoacetate) to form the tricarbonyl precursor.
- Perform stereoselective reduction using carbonyl reductase or chiral boranes, followed by acetonide protection and nitrile reduction to yield ATS-9.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route offers transformative benefits that extend beyond mere technical feasibility. The primary value proposition lies in the drastic simplification of the supply chain for raw materials. By eliminating the dependency on specialized and tightly regulated reagents like sodium cyanide and lithium metal, manufacturers can source inputs from a broader, more stable vendor base. This diversification reduces the risk of supply disruptions caused by regulatory crackdowns on hazardous chemical transport or production quotas on lithium. Furthermore, the removal of cryogenic requirements liberates the production schedule from the constraints of liquid nitrogen availability and the high energy costs associated with maintaining ultra-low temperatures, leading to significant operational expenditure reductions.
- Cost Reduction in Manufacturing: The economic impact of switching from lithium-based chemistry to magnesium or zinc protocols is profound. Lithium reagents are inherently expensive due to the scarcity of the metal and the energy-intensive processes required for their refinement and handling. In contrast, magnesium and zinc are commodity metals with stable pricing and widespread availability. Additionally, the elimination of liquid nitrogen for cryogenic cooling removes a substantial utility cost burden. The process also reduces waste disposal costs significantly, as the effluent no longer contains toxic cyanides or complex lithium salts, simplifying wastewater treatment and lowering environmental compliance fees.
- Enhanced Supply Chain Reliability: Reliability in the pharmaceutical supply chain is paramount, and this process enhances it by removing high-risk bottlenecks. The use of less hazardous reagents means that transportation and storage logistics are less constrained by dangerous goods regulations, facilitating smoother movement of materials across borders. The robustness of the reaction conditions, which do not require extreme sensitivity to moisture or oxygen compared to organolithium chemistry, reduces the likelihood of batch failures due to minor operational deviations. This stability ensures a consistent output of high-quality intermediates, securing the continuity of API production for downstream customers.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies safety risks, particularly with exothermic reactions involving pyrophoric reagents. This novel method mitigates such risks by operating under mild thermal conditions, making the transition from pilot plant to commercial tonnage much safer and more predictable. The alignment with green chemistry principles—specifically the reduction of hazardous substances and energy consumption—positions manufacturers favorably against tightening global environmental regulations. This proactive compliance strategy future-proofs the production facility against potential regulatory bans on older, dirtier technologies, ensuring long-term operational viability.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this green synthesis technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, aiming to clarify the practical benefits for industry stakeholders. Understanding these nuances is essential for evaluating the feasibility of technology transfer and the potential return on investment for upgrading existing manufacturing lines.
Q: How does this new process improve EHS safety compared to traditional methods?
A: The traditional industrial process relies heavily on sodium cyanide (NaCN) and Lithium Diisopropylamide (LDA), both of which pose severe toxicity and pyrophoric risks. This new patent eliminates the use of NaCN and expensive lithium reagents, replacing them with safer magnesium or zinc organometallics, significantly lowering the EHS risk profile and waste treatment costs.
Q: What are the cost advantages of using magnesium or zinc reagents over lithium?
A: Lithium metal and organic lithium reagents like LDA are expensive and require cryogenic conditions (liquid nitrogen) for safe handling, driving up energy and raw material costs. The disclosed method utilizes abundant and cheaper metals like magnesium and zinc, operating under mild reflux conditions, which drastically simplifies the thermal management and reduces overall production expenses.
Q: Can this synthetic route be scaled for commercial API production?
A: Yes, the process is specifically designed for industrial scalability. By avoiding cryogenic temperatures and highly hazardous reagents, the reaction conditions are much more forgiving on a large scale. The patent reports high yields (e.g., 79.5% total yield for key steps) and uses standard solvents like THF and ethanol, making it highly suitable for commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Atorvastatin Intermediate Supplier
The technological advancements detailed in patent CN109503542B underscore the evolving landscape of pharmaceutical intermediate manufacturing, where efficiency and safety are paramount. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, leveraging our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to bring such innovative processes to market. Our state-of-the-art facilities are equipped to handle complex organometallic and biocatalytic reactions with stringent purity specifications, ensuring that every batch meets the rigorous quality standards required by global regulatory bodies. With our rigorous QC labs and commitment to continuous process improvement, we are uniquely positioned to support your supply chain with high-purity Atorvastatin intermediates.
We invite you to collaborate with us to explore how this green synthesis route can be integrated into your production strategy to achieve substantial cost savings and sustainability goals. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and regional compliance needs. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us partner with you to secure a more efficient and sustainable future for your pharmaceutical manufacturing operations.
