Advanced Synthesis of Atorvastatin Calcium: High-Yield Route for Commercial API Production
Introduction to Advanced Atorvastatin Calcium Synthesis
The global demand for effective lipid-lowering agents continues to drive innovation in the synthesis of Atorvastatin Calcium, a cornerstone medication in the statin class. Patent CN102796036B introduces a transformative preparation method that addresses critical inefficiencies found in legacy manufacturing processes. By fundamentally reordering the hydrolysis steps—specifically prioritizing basic hydrolysis before acid treatment—this technology achieves a total yield exceeding 90%, significantly outperforming traditional methods that typically struggle to reach 83%. For R&D directors and procurement managers, this breakthrough represents a pivotal opportunity to enhance supply chain reliability and reduce the cost of goods sold (COGS) for this high-volume active pharmaceutical ingredient (API). The method not only simplifies the purification landscape but also ensures the production of high-purity amorphous Atorvastatin Calcium, meeting stringent regulatory standards for modern pharmaceutical formulations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Atorvastatin Calcium has relied on a sequence where acid hydrolysis is performed prior to basic hydrolysis. As illustrated in the conventional reaction pathway, this approach exposes the sensitive intermediate to acidic conditions while the tert-butyl ester protection is still intact or during the initial deprotection phase. This sequence is chemically precarious; the acidic environment often catalyzes unwanted side reactions, including the degradation of the pyrrole ring or the formation of difficult-to-remove impurities. Consequently, prior art documents, such as CN1980890A, report overall yields ranging merely between 64% and 83%. Furthermore, the resulting crude product often requires rigorous and costly purification steps, such as column chromatography, to remove these acid-generated byproducts, creating a bottleneck in large-scale manufacturing and inflating solvent consumption.
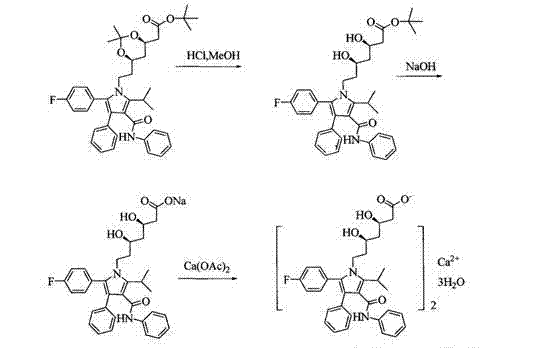
The Novel Approach
In stark contrast, the methodology disclosed in CN102796036B inverts this logical sequence to maximize chemical stability and yield. The novel approach initiates with basic hydrolysis using sodium hydroxide in a tetrahydrofuran (THF) system. This step selectively cleaves the tert-butyl ester to form a stable sodium carboxylate salt while leaving the acetonide protecting group on the dihydroxy side chain intact. By forming the sodium salt first, the molecule becomes more resistant to the degradative pathways observed in the acid-first route. Subsequent acid hydrolysis is then employed solely to remove the acetonide protection under controlled conditions. This strategic reordering eliminates the generation of major side products, thereby streamlining the downstream purification process and enabling a direct path to high-purity material without the need for extensive chromatographic separation.
Mechanistic Insights into Base-First Hydrolysis Strategy
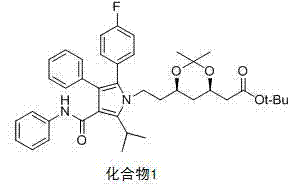
The core mechanistic advantage of this process lies in the differential stability of the protecting groups under varying pH conditions. The starting material, identified as Compound 1 in the patent literature, features both a tert-butyl ester and an acetonide (isopropylidene) protecting group. Under the optimized conditions, the addition of 20% NaOH solution at a controlled pH of approximately 11 facilitates the saponification of the ester. Crucially, acetonide groups are generally stable in basic media, allowing the ester to be hydrolyzed selectively without exposing the sensitive diol functionality prematurely. This results in the formation of a robust sodium salt intermediate, (4R-cis)-6-[2-[2-(4-fluorophenyl)-5-(1-sec.-propyl)-3-phenyl-4-[(aniline) carbonyl]-1H-pyrroles-1-yl] ethyl]-2,2-dimethyl-1,3-dioxane-4-sodium acetate, which serves as a stable platform for the subsequent transformation.
Following the formation of the sodium salt, the process transitions to acid hydrolysis using dilute hydrochloric acid. In this second stage, the acidic conditions are specifically tuned to cleave the acetonide protecting group, revealing the essential 3,5-dihydroxy heptanoic acid side chain required for HMG-CoA reductase inhibition. Because the carboxylic acid moiety is already present as a sodium salt, the molecule maintains better solubility characteristics in the aqueous workup compared to the free acid form generated in conventional routes. This solubility profile aids in the efficient removal of organic impurities via extraction with methyl tert-butyl ether. The final conversion to the calcium salt is achieved by reacting the purified dihydroxy acid sodium salt with calcium acetate, precipitating the final API in its amorphous form, which is pharmacologically preferred for its dissolution properties.
How to Synthesize Atorvastatin Calcium Efficiently
The implementation of this synthesis route requires precise control over reaction parameters, particularly pH and temperature, to ensure the selective deprotection sequence functions as intended. The process begins with the dissolution of the protected precursor in THF, followed by the careful addition of alkali to maintain a pH of 11 at 30-35°C. Once the sodium salt is formed and isolated, it is subjected to acid hydrolysis at similar temperatures to remove the acetonide group. The detailed operational parameters, including specific solvent ratios, stirring times, and crystallization conditions for the final calcium salt, are critical for reproducing the >90% yield reported in the patent. For process chemists looking to adopt this technology, strict adherence to the base-first protocol is essential to avoid reverting to the lower-yielding side reactions of the past.
- Perform basic hydrolysis on the tert-butyl ester precursor using NaOH in THF to form the stable sodium salt intermediate while retaining acetonide protection.
- Conduct acid hydrolysis using HCl to remove the acetonide protecting group, revealing the dihydroxy side chain without degrading the core structure.
- React the purified dihydroxy acid sodium salt with calcium acetate to precipitate the final amorphous Atorvastatin Calcium product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this base-first hydrolysis method offers tangible economic and logistical benefits that extend beyond simple yield improvements. By eliminating the side reactions inherent to the conventional acid-first approach, the process drastically reduces the burden on downstream purification units. The removal of complex chromatographic steps, which are often resource-intensive and difficult to scale, translates directly into reduced solvent consumption and lower waste disposal costs. Furthermore, the simplified workflow enhances batch cycle times, allowing manufacturing facilities to increase throughput without requiring additional capital investment in reactor volume. This efficiency gain is crucial for maintaining competitive pricing in the generic pharmaceutical market.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the substantial increase in overall yield, moving from a historical average of roughly 70% to over 90%. This improvement means that for every ton of raw material purchased, the output of saleable API increases significantly, effectively lowering the unit cost of production. Additionally, the avoidance of column chromatography—a step frequently cited in prior art for purification—eliminates the need for large quantities of silica gel and specialized elution solvents. This reduction in material usage, combined with decreased waste treatment requirements for hazardous solvents, results in a leaner, more cost-effective manufacturing profile that aligns with modern green chemistry initiatives.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by complex processes that are prone to failure or variability. The robust nature of the sodium salt intermediate formed in the first step provides a stable checkpoint in the synthesis, reducing the risk of batch rejection due to impurity profiles. Because the reagents used—such as sodium hydroxide, hydrochloric acid, and calcium acetate—are commodity chemicals with stable global supply lines, the risk of raw material shortage is minimized. This reliability ensures that production schedules can be met consistently, reducing lead times for high-purity pharmaceutical intermediates and providing a buffer against market volatility.
- Scalability and Environmental Compliance: Scaling chemical processes from the laboratory to commercial production often reveals hidden complexities, but this method utilizes standard unit operations such as liquid-liquid extraction and precipitation. The use of common solvents like THF, ethyl acetate, and hexane allows for straightforward integration into existing multi-purpose API manufacturing plants. Moreover, the reduction in side products means less chemical waste is generated per kilogram of product, simplifying environmental compliance and wastewater treatment. This scalability ensures that the process can be seamlessly expanded from pilot batches of 100 kgs to full commercial production of 100 MT annually without compromising quality or safety standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. Understanding these details is vital for technical teams evaluating the feasibility of technology transfer and for procurement specialists assessing the long-term value proposition of this manufacturing route. The answers are derived directly from the experimental data and claims presented in the patent documentation, ensuring accuracy and relevance for decision-makers.
Q: Why is the base-first hydrolysis method superior to conventional acid-first routes?
A: Conventional methods initiate with acid hydrolysis, which often triggers side reactions and degradation of the sensitive pyrrole ring or ester moieties, leading to lower yields (64%-83%). The base-first approach stabilizes the molecule as a sodium salt initially, preventing these side reactions and achieving total yields exceeding 90%.
Q: What is the expected purity and yield of this synthesis method?
A: According to patent CN102796036B, this optimized protocol delivers a total reaction yield higher than 90%. The process includes specific purification steps, such as extraction with ethyl acetate and hexane, ensuring high product purity suitable for pharmaceutical applications.
Q: Does this process produce the amorphous or crystalline form of Atorvastatin Calcium?
A: The final step of the described method involves stirring the purified calcium salt in water at room temperature followed by drying, which specifically yields the amorphous form of Atorvastatin Calcium, known for its favorable solubility characteristics compared to certain crystalline forms.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Atorvastatin Calcium Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthesis routes in the competitive landscape of generic pharmaceuticals. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the base-first hydrolysis method for Atorvastatin Calcium can be successfully translated from patent to plant. We are committed to delivering high-purity intermediates and APIs that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. Our infrastructure is designed to handle complex chemistries with precision, guaranteeing supply continuity for our global partners.
We invite you to collaborate with us to leverage this advanced technology for your supply chain. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, ensuring that your next project is built on a foundation of chemical excellence and commercial viability.
