Advanced Solvent-Free Manufacturing of High-Purity Triarylmethane Derivatives for Global Pharma
Advanced Solvent-Free Manufacturing of High-Purity Triarylmethane Derivatives for Global Pharma
The landscape of pharmaceutical intermediate manufacturing is undergoing a significant transformation driven by the urgent need for greener, more sustainable synthetic methodologies. Triarylmethane derivatives represent a cornerstone structural motif in medicinal chemistry, renowned for their diverse biological activities ranging from potent antibacterial and antiviral properties to significant anticancer potential. As illustrated in the structural diversity of bioactive scaffolds, these compounds serve as critical building blocks for next-generation therapeutics targeting complex diseases. 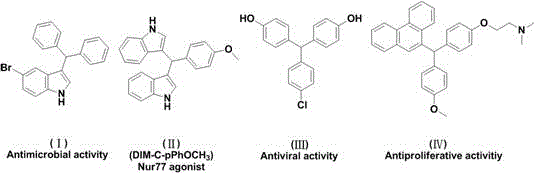 . A pivotal advancement in this domain is detailed in Chinese Patent CN111138299A, which discloses a revolutionary solvent-free green synthesis method. This technology leverages a 1,6-conjugate addition reaction between methylene benzoquinone and N,N-dimethylaniline under acidic conditions, effectively bypassing the environmental and safety hazards associated with traditional routes. For global supply chain leaders and R&D directors, this patent represents a paradigm shift towards high atom economy and operational simplicity, offering a robust pathway for the reliable triarylmethane supplier market to meet stringent regulatory and sustainability goals.
. A pivotal advancement in this domain is detailed in Chinese Patent CN111138299A, which discloses a revolutionary solvent-free green synthesis method. This technology leverages a 1,6-conjugate addition reaction between methylene benzoquinone and N,N-dimethylaniline under acidic conditions, effectively bypassing the environmental and safety hazards associated with traditional routes. For global supply chain leaders and R&D directors, this patent represents a paradigm shift towards high atom economy and operational simplicity, offering a robust pathway for the reliable triarylmethane supplier market to meet stringent regulatory and sustainability goals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the triarylmethane skeleton has relied heavily on classical Friedel-Crafts alkylation or arylation reactions, which are fraught with significant operational and environmental drawbacks. Traditional protocols typically necessitate the use of strong Lewis acids or corrosive Brønsted acids in conjunction with volatile organic solvents to drive the reaction to completion. These processes often demand rigorous anhydrous and oxygen-free conditions to prevent catalyst deactivation and side reactions, thereby increasing the complexity and cost of the manufacturing infrastructure. Furthermore, the reliance on stoichiometric amounts of metal-based reagents introduces severe challenges in downstream processing, particularly regarding the removal of trace heavy metal impurities to meet pharmaceutical grade standards. The generation of substantial quantities of acidic wastewater and solvent waste not only escalates disposal costs but also poses significant environmental compliance risks for large-scale facilities. Consequently, the conventional approach suffers from low atom utilization and poor safety profiles, making it increasingly untenable for modern cost reduction in pharmaceutical intermediates manufacturing where efficiency and sustainability are paramount.
The Novel Approach
In stark contrast to these legacy methods, the novel approach outlined in the patent utilizes a direct 1,6-conjugate addition strategy that fundamentally simplifies the synthetic workflow. By employing p-methylenebenzoquinone and N,N-dimethylaniline as readily available starting materials, the reaction proceeds efficiently under solvent-free conditions, eliminating the need for bulk organic solvents entirely. The use of mild protonic acids, such as p-toluenesulfonic acid, as catalysts allows the reaction to occur at moderate temperatures around 95°C without the necessity for inert atmospheres. This methodology not only enhances the safety profile by removing flammable solvents but also drastically simplifies the work-up procedure, as the product can often be isolated through straightforward purification techniques like column chromatography. The absence of metal reagents ensures a cleaner impurity profile, reducing the burden on quality control laboratories. This innovative route exemplifies how green chemistry principles can be successfully integrated into the commercial scale-up of complex pharmaceutical intermediates, delivering a process that is both economically viable and environmentally responsible.
Mechanistic Insights into Bronsted Acid-Catalyzed 1,6-Conjugate Addition
The core of this technological breakthrough lies in the precise mechanistic pathway of the acid-catalyzed 1,6-conjugate addition. The reaction initiates with the activation of the p-methylenebenzoquinone electrophile by the Bronsted acid catalyst, which increases the electrophilicity of the exocyclic methylene group. Subsequently, the electron-rich aromatic ring of N,N-dimethylaniline acts as a nucleophile, attacking the activated methylene carbon to form a new carbon-carbon bond. This step is followed by a proton transfer and rearomatization sequence that stabilizes the triarylmethane framework. 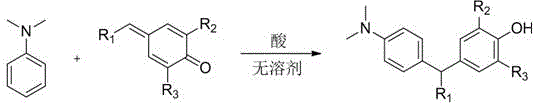 . The beauty of this mechanism is its tolerance to various electronic environments; electron-donating groups on the quinone ring enhance the stability of the intermediate, while the dimethylamino group on the aniline ring provides sufficient nucleophilicity to drive the reaction forward even without harsh activation. The catalytic cycle is efficient, requiring only 300 mol% of the acid promoter to achieve high conversion rates, demonstrating excellent turnover and minimizing reagent consumption. This mechanistic clarity allows chemists to predict reactivity patterns and optimize conditions for diverse substrate combinations with high confidence.
. The beauty of this mechanism is its tolerance to various electronic environments; electron-donating groups on the quinone ring enhance the stability of the intermediate, while the dimethylamino group on the aniline ring provides sufficient nucleophilicity to drive the reaction forward even without harsh activation. The catalytic cycle is efficient, requiring only 300 mol% of the acid promoter to achieve high conversion rates, demonstrating excellent turnover and minimizing reagent consumption. This mechanistic clarity allows chemists to predict reactivity patterns and optimize conditions for diverse substrate combinations with high confidence.
From an impurity control perspective, this mechanism offers distinct advantages over radical-based or metal-catalyzed alternatives. The absence of transition metals eliminates the risk of metal-catalyzed oxidative coupling byproducts or residual metal contamination, which are critical failure points in API synthesis. The reaction specificity is governed by the electronic match between the quinone methide and the aniline derivative, leading to high regioselectivity and minimizing the formation of poly-alkylated side products. Furthermore, the mild thermal conditions (95°C) prevent thermal degradation of sensitive functional groups, ensuring that the final product retains its structural integrity. The solvent-free nature of the reaction medium also reduces the likelihood of solvent-participation side reactions, such as etherification or solvolysis, which can complicate purification. For R&D teams focused on purity and impurity profiling, this clean reaction profile translates to simpler analytical validation and faster regulatory approval timelines for new drug candidates incorporating this scaffold.
How to Synthesize Triarylmethane Derivatives Efficiently
The practical implementation of this synthesis route is designed for ease of execution, making it accessible for both laboratory discovery and pilot plant operations. The protocol involves a straightforward one-pot procedure where the amine and quinone precursors are mixed directly with the acid catalyst. Detailed standard operating procedures for this transformation, including precise stoichiometric ratios and temperature controls, are essential for reproducibility. The following guide outlines the critical steps derived from the patent examples to ensure optimal yield and purity.
- Charge N,N-dimethylaniline and p-methylenebenzoquinone into a reaction vessel at a molar ratio of 1.2: 1.
- Add a Bronsted acid catalyst, such as p-toluenesulfonic acid (300 mol%), and stir the mixture at 95°C.
- Monitor reaction progress via TLC until completion, then purify the crude product using column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this solvent-free technology presents a compelling value proposition centered on cost efficiency and operational resilience. The elimination of organic solvents removes a major variable cost component, as there is no longer a need for purchasing, storing, and recovering large volumes of volatile chemicals. This reduction in material usage directly correlates to lower raw material expenditures and decreased logistics burdens associated with hazardous chemical transport. Moreover, the simplified reaction setup reduces the capital expenditure required for specialized reactor vessels equipped with solvent recovery distillation columns. The process inherently lowers the E-factor (environmental factor) of the manufacturing line, aligning corporate operations with increasingly strict global environmental regulations and sustainability mandates. These factors collectively contribute to a more robust and cost-effective supply chain capable of withstanding market fluctuations in solvent prices.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the drastic simplification of the downstream processing workflow. By avoiding the use of metal catalysts and toxic solvents, the expensive and time-consuming steps of metal scavenging and solvent distillation are rendered unnecessary. This streamlining of the production line reduces energy consumption and labor hours, leading to substantial cost savings per kilogram of product. Additionally, the high atom economy of the 1,6-conjugate addition ensures that a greater proportion of the starting mass is converted into the desired product, minimizing waste disposal fees. The use of inexpensive, commodity-grade Bronsted acids further depresses the reagent cost base compared to precious metal catalysts. These cumulative efficiencies allow for a highly competitive pricing structure for high-purity triarylmethane intermediates in the global market.
- Enhanced Supply Chain Reliability: Supply chain continuity is significantly bolstered by the reliance on widely available, non-specialized raw materials. N,N-dimethylaniline and substituted phenols are commodity chemicals produced at massive scales globally, reducing the risk of supply shortages that often plague exotic reagents. The robustness of the reaction conditions, which do not require stringent anhydrous or inert gas environments, means that production can be maintained even in facilities with standard utility infrastructure. This flexibility reduces the dependency on specialized equipment maintenance and minimizes downtime due to technical failures. Furthermore, the stability of the reagents allows for longer shelf-life and easier inventory management, ensuring that production schedules can be met consistently without interruption. This reliability is crucial for maintaining just-in-time delivery commitments to downstream pharmaceutical partners.
- Scalability and Environmental Compliance: Scaling this process from gram to tonnage levels is facilitated by the absence of solvent-mediated heat transfer limitations, allowing for safer exotherm management in larger reactors. The solvent-free nature inherently reduces the volume of hazardous waste generated, simplifying compliance with environmental discharge permits and reducing the carbon footprint of the manufacturing site. The mild reaction temperatures and atmospheric pressure operation eliminate the need for high-pressure autoclaves or cryogenic cooling systems, lowering the barrier to scale-up. This ease of scalability ensures that supply can be rapidly ramped up to meet surging demand for clinical trial materials or commercial launch volumes. The alignment with green chemistry principles also enhances the corporate sustainability profile, appealing to eco-conscious stakeholders and investors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this solvent-free synthesis technology. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity for technical decision-makers. Understanding these nuances is vital for assessing the feasibility of integrating this route into existing manufacturing portfolios.
Q: What are the primary advantages of this solvent-free method over conventional Friedel-Crafts reactions?
A: Unlike conventional methods that require toxic solvents, anhydrous conditions, and harsh Lewis acids, this patented process operates under solvent-free conditions using mild Bronsted acids. This eliminates the need for complex solvent recovery systems and reduces hazardous waste generation significantly.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the process is highly suitable for scale-up. It utilizes commercially available raw materials, does not require strict anhydrous or oxygen-free environments, and involves a simple one-pot reaction procedure that facilitates easy separation and purification of the final triarylmethane products.
Q: What types of substituents are tolerated in this 1,6-conjugate addition reaction?
A: The reaction demonstrates broad substrate scope, tolerating various substituents on the aromatic rings including halogens (fluoro, chloro, bromo), alkyl groups (methyl, tert-butyl, isopropyl), and alkoxy groups (methoxy), allowing for the synthesis of diverse triarylmethane derivatives.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Triarylmethane Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this solvent-free synthesis technology for the production of high-value pharmaceutical intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop discovery to full-scale manufacturing. Our facilities are equipped with state-of-the-art rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of triarylmethane derivative meets the highest international quality standards. We are committed to leveraging our technical expertise to optimize this green chemistry route, maximizing yield while minimizing environmental impact for our global clientele.
We invite you to collaborate with us to unlock the full commercial potential of your triarylmethane-based projects. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to contact us today to request specific COA data and comprehensive route feasibility assessments. By partnering with us, you gain access to a supply chain that prioritizes innovation, sustainability, and reliability, positioning your organization for success in the competitive pharmaceutical marketplace.
