Advanced Metal-Free Synthesis of Triarylmethane Derivatives for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable, and cost-effective methodologies for constructing complex molecular scaffolds. Patent CN111484419B introduces a groundbreaking approach for the synthesis of triarylmethane derivatives, a class of compounds pivotal in medicinal chemistry, dye manufacturing, and material science. This innovation leverages an iodine-catalyzed, one-pot reaction strategy that merges substituted salicylaldehydes, aromatic boronic acids, and arenes into a unified synthetic pathway. Unlike conventional methods that often rely on harsh conditions or scarce resources, this technique operates under base-free and ligand-free conditions, completely bypassing the need for expensive transition metal catalysts. The result is a streamlined process that delivers high chemical selectivity and impressive yields, addressing critical pain points in the supply chain for high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of triarylmethane frameworks has been plagued by significant technical and economic hurdles that hinder efficient commercial production. Traditional Friedel-Crafts alkylation methods, while conceptually straightforward, frequently suffer from poor regioselectivity, leading to complex mixtures of isomeric by-products that are difficult and costly to separate. Furthermore, these reactions often require strong Lewis acids and严苛 conditions that limit the tolerance of sensitive functional groups, thereby restricting the diversity of accessible derivatives. Alternative approaches involving transition metal-catalyzed cross-coupling or carbon-hydrogen functionalization introduce another layer of complexity, necessitating the use of precious metals like palladium or platinum. The presence of these heavy metals mandates rigorous and expensive purification steps to meet stringent regulatory limits for residual metals in active pharmaceutical ingredients, ultimately inflating the cost of goods and extending lead times for reliable agrochemical intermediate supplier networks.
The Novel Approach
In stark contrast to these legacy techniques, the methodology disclosed in CN111484419B offers a transformative solution by utilizing inexpensive iodine species as the sole catalyst. This metal-free protocol enables the direct assembly of three distinct aryl groups in a single operational step, dramatically simplifying the synthetic route. The reaction proceeds smoothly in common organic solvents such as chlorobenzene at moderate temperatures ranging from 40°C to 120°C, ensuring energy efficiency and operational safety. By eliminating the need for external bases or specialized ligands, the process reduces reagent costs and minimizes waste generation, aligning perfectly with green chemistry principles. The versatility of this approach is exemplified by its ability to produce completely asymmetric triarylmethane derivatives with high fidelity, as demonstrated in the specific reaction scheme below which highlights the convergence of salicylaldehyde, phenylboronic acid, and N,N-dimethylaniline.
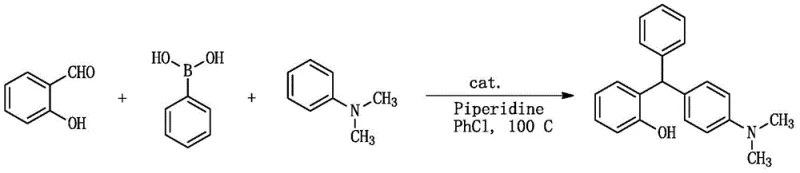
Mechanistic Insights into Iodine-Catalyzed Cyclization
The success of this synthetic strategy lies in the unique activation mode provided by the iodine catalyst, which facilitates electrophilic aromatic substitution without the oxidative addition steps typical of transition metal cycles. The mechanism likely involves the activation of the aldehyde carbonyl group by the iodine species, enhancing its electrophilicity towards the nucleophilic attack by the electron-rich arene component. Subsequent dehydration and further arylation by the boronic acid species, potentially activated by the amine additive or the solvent system, lead to the formation of the central methane carbon bridge. This cascade occurs with remarkable chemoselectivity, preserving sensitive functional groups such as halogens, nitro groups, and ethers that might otherwise be compromised under harsher acidic or basic conditions. The absence of transition metals also precludes the formation of metal-complexed impurities, resulting in a cleaner crude reaction profile that simplifies downstream processing.
Crucially, the structural integrity of the starting materials is maintained throughout the transformation, allowing for extensive diversification of the final product library. The salicylaldehyde component, depicted in the general structure below, can accommodate a wide array of substituents at the R1 position, including hydrogen, methyl, nitro, bromo, fluoro, and chloro groups. This flexibility is paramount for medicinal chemists aiming to optimize the pharmacokinetic properties of drug candidates by fine-tuning electronic and steric parameters. The tolerance for multiple substituents, such as 3,5-dichloro or 3,5-dibromo patterns, further underscores the robustness of the catalytic system. Such broad substrate scope ensures that this method is not merely a niche academic curiosity but a versatile platform technology capable of supporting the diverse needs of a reliable pharmaceutical intermediate supplier.
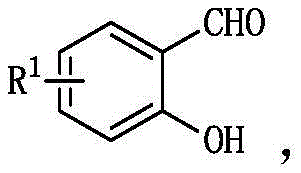
How to Synthesize Triarylmethane Derivatives Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to reagent stoichiometry and reaction monitoring to maximize yield and purity. The protocol dictates a molar ratio of approximately 1:1 to 1.5 for the aldehyde, boronic acid, and amine components, with a catalytic loading of iodine species between 0.5 and 1 equivalent relative to the limiting reagent. Reaction progress is typically tracked using thin-layer chromatography (TLC) to ensure complete consumption of the starting salicylaldehyde before proceeding to workup. Following the reaction, a standard aqueous workup involving ethyl acetate extraction, washing with water and saturated brine, and drying over anhydrous sodium sulfate effectively isolates the organic product. Final purification is achieved through column chromatography using a hexane and ethyl acetate eluent system, yielding the target triarylmethane derivative in high purity suitable for subsequent biological evaluation or chemical transformation.
- Mix substituted salicylaldehyde, aromatic boronic acid, arene derivative, piperidine, and an iodine-based catalyst (I2, KI, or TBAI) in a dry reaction vessel.
- Add a suitable solvent such as chlorobenzene or 1,4-dioxane and heat the mixture to between 40°C and 120°C while stirring for 20 to 80 minutes.
- Upon completion monitored by TLC, cool the reaction, extract with ethyl acetate, wash with water and brine, dry over sodium sulfate, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this iodine-catalyzed methodology presents a compelling value proposition centered on cost optimization and risk mitigation. The elimination of precious metal catalysts removes a significant volatile cost component from the bill of materials, shielding production budgets from fluctuations in the global markets for palladium and platinum. Moreover, the simplified purification process reduces the consumption of silica gel and solvents associated with heavy metal scavenging, leading to substantial cost savings in manufacturing overheads. The use of readily available, commodity-grade starting materials such as salicylaldehydes and boronic acids ensures a stable and resilient supply chain, minimizing the risk of production delays caused by sourcing bottlenecks for exotic reagents.
- Cost Reduction in Manufacturing: The replacement of expensive transition metal catalysts with inexpensive iodine salts directly lowers the raw material costs per kilogram of product. Additionally, the base-free and ligand-free nature of the reaction reduces the number of auxiliary chemicals required, further driving down the overall cost of goods sold. The simplified workup procedure also translates to reduced labor hours and utility consumption during the isolation and purification stages, contributing to a leaner and more economical manufacturing process.
- Enhanced Supply Chain Reliability: By relying on widely available commercial feedstocks, manufacturers can secure long-term supply contracts with multiple vendors, thereby enhancing supply chain continuity. The robustness of the reaction conditions means that production is less susceptible to disruptions caused by the need for specialized equipment or strict environmental controls often associated with handling pyrophoric or highly toxic reagents. This reliability is critical for maintaining consistent delivery schedules to downstream customers in the pharmaceutical and agrochemical sectors.
- Scalability and Environmental Compliance: The one-pot nature of the synthesis minimizes the number of unit operations, making the process inherently easier to scale from kilogram to multi-ton quantities without significant re-engineering. Furthermore, the absence of heavy metals simplifies waste stream management and disposal, ensuring compliance with increasingly stringent environmental regulations regarding hazardous waste. This environmental compatibility not only reduces disposal costs but also enhances the sustainability profile of the manufactured intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthetic route. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear understanding of the technology's capabilities and limitations. Understanding these details is essential for R&D teams evaluating the feasibility of integrating this method into their existing process development pipelines.
Q: What are the primary advantages of this iodine-catalyzed method over traditional transition metal catalysis?
A: The primary advantage is the complete elimination of expensive and toxic transition metals like palladium or platinum. This method utilizes inexpensive iodine catalysts under base-free and ligand-free conditions, significantly simplifying post-reaction purification and reducing heavy metal contamination risks in the final pharmaceutical intermediate.
Q: Does this synthesis method support a wide range of substrate functional groups?
A: Yes, the patent demonstrates excellent chemoselectivity and substrate tolerance. It successfully accommodates various substituents on the salicylaldehyde, boronic acid, and amine components, including electron-withdrawing groups like nitro and halogens, as well as electron-donating groups like methoxy and alkyl chains, without compromising yield.
Q: Is the process scalable for industrial production of triarylmethane derivatives?
A: The process is highly scalable due to its one-pot nature and simple workup procedure involving standard extraction and column chromatography. The use of readily available commercial reagents and mild reaction conditions (40-120°C) facilitates safe and efficient scale-up from laboratory to commercial manufacturing volumes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Triarylmethane Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the iodine-catalyzed synthesis described in CN111484419B for producing high-value triarylmethane derivatives. As a seasoned CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop discovery to full-scale manufacturing. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of intermediate delivered meets the exacting standards required by the global pharmaceutical industry.
We invite you to collaborate with our technical team to explore how this cost-effective and environmentally friendly synthesis can enhance your product portfolio. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how we can become your trusted partner in delivering high-purity pharmaceutical intermediates with unmatched reliability and speed.
