Advanced Stereoselective Inversion Strategy for Commercial Ursodesoxycholic Acid Production
Advanced Stereoselective Inversion Strategy for Commercial Ursodesoxycholic Acid Production
The pharmaceutical industry continuously seeks robust synthetic pathways for high-value active pharmaceutical ingredients, particularly for hepatoprotective agents like ursodesoxycholic acid (UDCA). Patent CN102746359A introduces a transformative methodology that shifts the paradigm from traditional reduction techniques to a highly selective stereochemical inversion strategy starting from chenodeoxycholic acid. This novel approach addresses critical bottlenecks in purity and yield that have long plagued conventional manufacturing processes. By leveraging specific functional group protections and precise activation of the C-7 hydroxyl group, the technology enables the production of high-purity ursodesoxycholic acid with superior optical integrity. For global procurement teams and R&D directors, this represents a significant opportunity to secure a reliable ursodesoxycholic acid supplier capable of delivering consistent quality at scale. The strategic implementation of this chemistry not only enhances product specifications but also aligns with modern green chemistry principles by reducing the reliance on harsh reducing agents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of ursodesoxycholic acid has relied heavily on the reduction of 7-ketolithocholic acid using sodium metal in alcoholic solvents. This legacy process is fundamentally flawed due to its inherent lack of stereoselectivity during the hydro-reduction phase. Typically, only about 80% of the ketone precursor is successfully reduced to the desired 7-beta-hydroxy configuration, while the remaining 20% reverts to the 7-alpha-hydroxy chenodeoxycholic acid isomer. This formation of unwanted byproducts necessitates rigorous and costly downstream purification steps to isolate the target molecule, drastically impacting the overall process efficiency. Furthermore, the final yield of pure ursodesoxycholic acid from the starting material often hovers around a mere 60%, representing a substantial loss of valuable raw materials. The use of sodium metal also introduces significant safety hazards and operational complexities, making the process less attractive for modern, safety-conscious manufacturing facilities seeking cost reduction in pharmaceutical intermediates manufacturing.
The Novel Approach
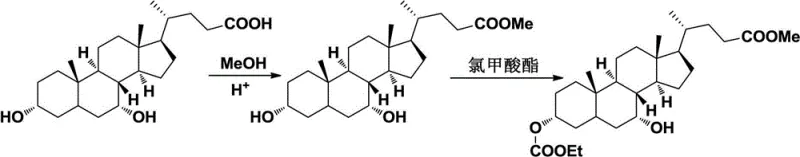
In stark contrast, the methodology disclosed in the patent utilizes a sophisticated sequence of protection, activation, and inversion to bypass the selectivity issues of reduction. The process begins with the esterification of the carboxyl group and the selective protection of the 3-hydroxyl group, effectively masking these sites to prevent side reactions. The core innovation lies in the activation of the 7-hydroxyl group, converting it into a superior leaving group such as a methanesulfonate, tosylate, or nitrobenzenesulfonate ester. Subsequent nucleophilic attack induces a clean stereochemical inversion at the C-7 position, transforming the alpha-configuration directly into the therapeutically active beta-configuration. This route eliminates the generation of the unwanted chenodeoxycholic acid epimer during the key transformation step, thereby simplifying purification. The result is a streamlined workflow that offers mild reaction conditions, straightforward operations, and significantly enhanced efficiency, positioning it as a preferred choice for the commercial scale-up of complex bile acid derivatives.
Mechanistic Insights into Stereoselective Inversion at C-7
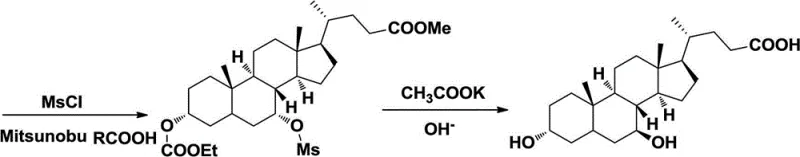
The success of this synthetic route hinges on the precise manipulation of stereochemistry at the C-7 position of the steroid nucleus through an SN2-type nucleophilic substitution mechanism. By first protecting the 3-hydroxyl group with an ethyl carbonate moiety, the chemist ensures that the subsequent activation reagents react exclusively with the 7-hydroxyl group. When methanesulfonyl chloride is introduced, it converts the 7-hydroxyl into a methanesulfonate ester, creating an excellent leaving group that is primed for displacement. The introduction of a nucleophile, such as potassium acetate, attacks the C-7 carbon from the opposite face of the leaving group, forcing the inversion of configuration from the 7-alpha to the 7-beta orientation. This mechanistic certainty is what guarantees the high optical purity of the final product, as the reaction pathway is energetically favorable for inversion rather than retention or racemization. Understanding this mechanism is crucial for R&D teams aiming to optimize reaction parameters and ensure batch-to-batch consistency in high-purity ursodesoxycholic acid production.
Furthermore, the choice of protecting groups and activation reagents plays a pivotal role in controlling the impurity profile of the synthesis. The use of vinyl chloroformate for 3-OH protection provides a stable intermediate that withstands the conditions required for mesylation and subsequent inversion. Following the inversion step, the removal of the protecting groups under basic hydrolysis conditions restores the free hydroxyl groups and the carboxylic acid without affecting the newly established stereochemistry. This careful orchestration of functional group transformations minimizes the formation of degradation products or rearrangement byproducts that often complicate bile acid chemistry. For quality control laboratories, this translates to a cleaner crude product that requires less intensive purification, directly supporting the goal of reducing lead time for high-purity pharmaceutical intermediates while maintaining stringent regulatory compliance standards.
How to Synthesize Ursodesoxycholic Acid Efficiently
The synthesis of ursodesoxycholic acid via this inversion pathway involves a logical sequence of esterification, selective protection, activation, inversion, and deprotection. The process starts by dissolving chenodeoxycholic acid in methanol with an acid catalyst to form the methyl ester, followed by reaction with vinyl chloroformate to protect the 3-position. The 7-hydroxyl is then activated using methanesulfonyl chloride in pyridine to form the mesylate intermediate. This intermediate undergoes nucleophilic substitution with potassium acetate in DMF to invert the stereochemistry, and finally, basic hydrolysis removes all protecting groups to yield the target acid. Detailed standardized operating procedures and specific molar ratios for each step are critical for maximizing yield and purity. For a comprehensive breakdown of the exact experimental conditions and workup procedures, please refer to the technical guide below.
- Protect the carboxyl group via esterification and selectively protect the 3-hydroxyl group using vinyl chloroformate.
- Activate the 7-hydroxyl group by converting it into a leaving group such as a methanesulfonate ester.
- Perform nucleophilic substitution to invert the configuration at C-7, followed by deprotection to yield pure ursodesoxycholic acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this inversion-based synthesis offers profound strategic benefits beyond mere chemical elegance. The primary advantage lies in the drastic simplification of the purification train; by avoiding the formation of the chenodeoxycholic acid epimer during the key step, the need for expensive and time-consuming chromatographic separations is largely eliminated. This reduction in downstream processing complexity directly correlates to lower operational expenditures and reduced solvent consumption, driving significant cost reduction in pharmaceutical intermediates manufacturing. Additionally, the starting material, chenodeoxycholic acid, is a widely available natural product derived from bile, ensuring a stable and resilient supply chain that is not subject to the volatility of synthetic precursor markets. The mild reaction conditions further reduce energy consumption and equipment wear, contributing to a more sustainable and economically viable production model.
- Cost Reduction in Manufacturing: The elimination of the sodium-alcohol reduction step removes the need for handling hazardous metals and the associated safety infrastructure costs. More importantly, the high stereoselectivity of the inversion step means that a much larger proportion of the starting material is converted into the desired product, significantly improving the mass balance of the process. This efficiency gain reduces the cost of goods sold by minimizing waste and maximizing the throughput of existing reactor capacity. Qualitatively, the avoidance of difficult epimer separations translates to substantial savings in solvent usage and labor hours, making the overall process far more cost-effective than traditional reduction methods.
- Enhanced Supply Chain Reliability: Relying on chenodeoxycholic acid as a feedstock leverages an established agricultural supply chain, reducing the risk of raw material shortages common with specialized synthetic intermediates. The reagents used in this process, such as methanesulfonyl chloride and potassium acetate, are commodity chemicals available from multiple global suppliers, preventing single-source bottlenecks. This diversification of the supply base ensures continuity of supply even during market fluctuations, providing procurement teams with greater negotiating power and stability. The robustness of the synthetic route also means that production schedules are less likely to be disrupted by unexpected yield failures or purification delays.
- Scalability and Environmental Compliance: The process operates under mild conditions without the need for extreme temperatures or pressures, facilitating easier scale-up from pilot plant to commercial tonnage production. The absence of heavy metal catalysts or hazardous reducing agents simplifies wastewater treatment and waste disposal, aligning with increasingly strict environmental regulations. This environmental compatibility reduces the regulatory burden and potential liability associated with hazardous waste management. Consequently, manufacturers can achieve faster regulatory approvals and maintain a stronger corporate social responsibility profile, which is increasingly important for partnerships with major pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of ursodesoxycholic acid using this advanced inversion technology. These insights are derived directly from the patent specifications and are designed to clarify the operational benefits for potential partners. Understanding these details is essential for evaluating the feasibility of integrating this route into your existing supply chain or R&D pipeline.
Q: Why is the inversion method superior to traditional reduction for UDCA synthesis?
A: Traditional reduction of 7-ketolithocholic acid using sodium and alcohol suffers from poor stereoselectivity, often yielding a mixture of ursodesoxycholic acid and chenodeoxycholic acid that requires difficult separation. The inversion method described in patent CN102746359A utilizes selective protection and nucleophilic substitution to achieve high optical purity without generating significant amounts of the unwanted epimer.
Q: What are the key advantages of this synthetic route for industrial scale-up?
A: This route offers mild reaction conditions and avoids the use of hazardous sodium metal reductions. By eliminating the need for complex chromatographic separations of epimers and utilizing readily available chenodeoxycholic acid as a starting material, the process significantly simplifies operations and enhances overall production efficiency.
Q: How does this method impact the cost structure of bile acid manufacturing?
A: The method reduces costs by improving yield through better stereocontrol and minimizing waste associated with separating isomers. The use of standard reagents like methanesulfonyl chloride and potassium acetate allows for predictable pricing and reliable sourcing, contributing to a more stable supply chain for high-value pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ursodesoxycholic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and pure synthetic routes for vital hepatoprotective agents like ursodesoxycholic acid. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical advantages of this inversion chemistry are fully realized in large-scale manufacturing. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, guaranteeing that every batch supports the efficacy and safety of your final pharmaceutical formulations. Our capability to adapt and optimize complex steroid syntheses makes us an ideal partner for companies seeking to secure a stable supply of high-quality intermediates.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis can optimize your supply chain dynamics. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits specific to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project needs, ensuring a seamless transition to a more efficient and reliable sourcing strategy.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
