Advanced Manufacturing of Caspofungin Intermediates via Safer Substituted Thiophenol Routes
Advanced Manufacturing of Caspofungin Intermediates via Safer Substituted Thiophenol Routes
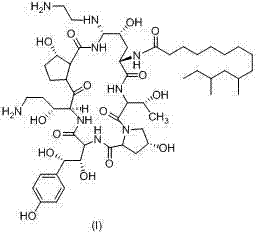
The pharmaceutical landscape for antifungal agents continues to evolve, with Caspofungin remaining a cornerstone therapy for invasive aspergillosis and candidiasis. The efficient and safe production of this complex azacyclohexapeptide is critical for global supply chains. Patent CN103483426A introduces a transformative preparation method that addresses long-standing safety and purification challenges inherent in previous synthetic routes. By leveraging substituted thiophenols instead of the highly toxic unsubstituted variant, this technology not only enhances operator safety but also fundamentally improves the physical properties of key intermediates, enabling robust crystallization and high-purity isolation without reliance on tedious chromatography. This report analyzes the technical merits and commercial implications of this novel pathway for stakeholders in the fine chemical and pharmaceutical sectors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the semi-synthetic production of Caspofungin from Pneumocandin B0 has been fraught with significant operational hazards and efficiency bottlenecks. Prior art, such as the methods disclosed in CN1127515C, relies heavily on unsubstituted thiophenol as a nucleophilic reagent to introduce the necessary side-chain modifications. This approach presents severe occupational health risks due to the extreme toxicity of thiophenol, which possesses a murine LD50 of less than 50mg/kg, necessitating rigorous and costly containment measures. Furthermore, the sulfide intermediates generated using unsubstituted thiophenol typically exist as oils or foams that resist solidification. This physical state forces manufacturers to employ column chromatography for purification, a technique that is notoriously difficult to scale, solvent-intensive, and prone to significant product loss, thereby driving up the cost of goods sold and limiting overall throughput capacity.
The Novel Approach
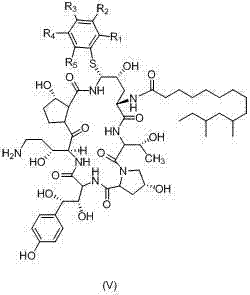
The methodology outlined in CN103483426A represents a paradigm shift by substituting the hazardous thiophenol with safer, substituted analogues such as p-toluene thiol or o-toluene thiol. These substituted reagents possess markedly lower toxicity profiles, with LD50 values exceeding 2.5g/kg, drastically reducing the safety burden on production facilities. More importantly from a process engineering perspective, the introduction of alkyl or aryl substituents on the phenyl ring alters the crystal lattice energy of the resulting intermediate (Formula V). This modification allows the intermediate to precipitate directly from the reaction solution as a solid upon the addition of water, bypassing the need for column chromatography entirely. The ability to purify via simple recrystallization not only streamlines the workflow but also ensures higher stability and purity of the intermediate, laying a solid foundation for the subsequent final coupling steps.
Mechanistic Insights into Borane-Mediated Reduction and Nucleophilic Substitution
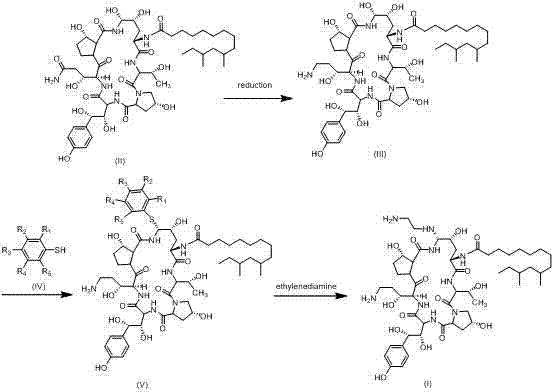
The synthesis initiates with a critical reduction step where the lactam moiety of the starting material (Formula II) is converted into the corresponding amine (Formula III). This transformation is achieved using a borane complex, such as borane-dimethyl sulfide, in a tetrahydrofuran (THF) solvent system. The choice of borane is strategic; it acts as a potent reducing agent capable of selectively opening the lactam ring under controlled temperatures (typically 0°C to room temperature) without compromising the stereochemical integrity of the adjacent chiral centers. Following the reduction, the reaction mixture is carefully quenched with hydrochloric acid to stabilize the amine product as a salt, which is then isolated via lyophilization. This step sets the stage for the subsequent nucleophilic attack by ensuring the availability of a reactive amine group while maintaining the structural fidelity required for the final bioactive conformation.
The core innovation lies in the subsequent substitution reaction where the Formula III amine reacts with a substituted thiophenol (Formula IV) under acidic catalysis. Conducted in acetonitrile with acids like trifluoroacetic acid, this reaction proceeds through an activated intermediate that facilitates the nucleophilic displacement. The specific choice of substituents (R groups) on the thiophenol ring is not merely a safety measure but a deliberate physicochemical tuning. For instance, the use of p-toluene thiol introduces a methyl group that enhances the hydrophobic interactions and packing efficiency of the molecule, promoting rapid crystallization upon workup. This mechanistic nuance ensures that the intermediate (Formula V) is obtained with high optical purity (ee value > 98%) and chemical purity (> 98%), effectively filtering out impurities through the crystal lattice formation rather than relying on downstream separation techniques.
How to Synthesize Caspofungin Efficiently
The synthesis of high-purity Caspofungin via this patented route involves a streamlined sequence that prioritizes safety and scalability. The process begins with the reduction of the cyclic peptide precursor, followed by the pivotal substitution with a substituted thiophenol to generate a crystalline intermediate, and concludes with an amination step to finalize the macrocycle. This approach eliminates the need for hazardous reagents and complex purification columns, making it ideally suited for GMP manufacturing environments. The detailed standardized synthetic steps, including specific molar ratios, temperature controls, and workup procedures, are outlined in the guide below to ensure reproducibility and quality compliance.
- Reduce the starting lactam compound (Formula II) using a borane complex in THF to obtain the amine intermediate (Formula III).
- React Formula III with a substituted thiophenol (Formula IV) under acidic conditions in acetonitrile to form the crystalline sulfide intermediate (Formula V).
- Treat the Formula V intermediate with ethylenediamine to displace the thiophenol group and cyclize, yielding the final Caspofungin product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers compelling economic and logistical benefits that extend beyond simple yield metrics. By replacing highly regulated toxic substances with safer alternatives, facilities can reduce the overhead costs associated with hazardous waste disposal and specialized safety infrastructure. The shift from chromatographic purification to crystallization significantly reduces solvent consumption and processing time, leading to a more agile production cycle. Furthermore, the enhanced stability of the crystalline intermediate minimizes the risk of degradation during storage and transport, ensuring a more reliable supply of high-quality material for downstream formulation.
- Cost Reduction in Manufacturing: The elimination of column chromatography is a primary driver for cost optimization in this process. Chromatography is a resource-intensive operation requiring large volumes of high-grade solvents and specialized silica media, both of which contribute significantly to variable costs. By enabling purification through recrystallization, the process drastically simplifies the workflow, reducing solvent recovery loads and minimizing product hold-up volume. Additionally, the use of less toxic thiophenol derivatives lowers the regulatory compliance costs and insurance premiums associated with handling extremely hazardous substances, contributing to a leaner cost structure for the final API.
- Enhanced Supply Chain Reliability: The robustness of the crystallization step directly translates to improved supply chain continuity. In traditional methods, the variability inherent in chromatographic separations can lead to batch failures or inconsistent throughput, creating bottlenecks. The predictable nature of crystallization allows for more accurate production planning and inventory management. Moreover, the intermediates produced via this method exhibit superior stability, reducing the likelihood of spoilage during warehousing or international shipping. This reliability ensures that downstream partners receive consistent quality material, mitigating the risk of production delays in the final drug product manufacturing.
- Scalability and Environmental Compliance: From an environmental and scaling perspective, this method aligns with modern green chemistry principles. The reduction in solvent usage and the avoidance of toxic thiophenol lower the environmental footprint of the manufacturing process, facilitating easier permitting and compliance with increasingly stringent environmental regulations. The process is inherently scalable because crystallization is a unit operation that scales linearly and predictably from pilot to commercial scales, unlike chromatography which often requires complex re-engineering for larger batches. This scalability ensures that the supply can grow in tandem with market demand for Caspofungin without requiring disproportionate capital investment in new equipment.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on the practical benefits of switching to substituted thiophenol reagents. Understanding these nuances is essential for technical teams evaluating the feasibility of technology transfer.
Q: Why are substituted thiophenols preferred over unsubstituted thiophenol in Caspofungin synthesis?
A: Substituted thiophenols, such as p-toluene thiol, exhibit significantly lower toxicity (LD50 > 2.5g/kg vs < 50mg/kg for thiophenol) and, crucially, impart crystallinity to the intermediate (Formula V), allowing for purification via recrystallization rather than difficult column chromatography.
Q: What represents the key yield improvement in this patented method?
A: The method achieves high yields (e.g., 92.5% for the intermediate step and 95% for the final step) by optimizing the substitution reaction conditions and utilizing the superior physical properties of the substituted intermediates to minimize loss during purification.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is designed for industrial suitability by eliminating highly toxic reagents, avoiding complex chromatographic separations in favor of crystallization, and using common solvents like acetonitrile and methanol, which facilitates safe scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Caspofungin Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic routes like the one described in CN103483426A to maintain competitiveness in the global pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this safer, crystallization-based process are fully realized in a GMP setting. We are committed to delivering high-purity Caspofungin intermediates that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities.
We invite potential partners to engage with our technical procurement team to discuss how this optimized route can enhance your supply chain resilience. Contact us today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. Our experts are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how we can support your long-term production goals with reliability and excellence.
