Advanced Catalytic Hydrogenation Route for High-Purity Caspofungin Manufacturing
Advanced Catalytic Hydrogenation Route for High-Purity Caspofungin Manufacturing
The pharmaceutical industry continuously seeks robust and scalable methodologies for the production of critical antifungal agents, particularly echinocandins like caspofungin. Patent CN101305018B introduces a transformative approach to synthesizing azacyclic hexapeptide compounds, specifically targeting the production of caspofungin with enhanced efficiency and purity. This innovation addresses long-standing challenges in the field by utilizing novel nitrile-containing intermediates that facilitate a highly selective catalytic hydrogenation step. Unlike conventional methods that struggle with low yields and complex purification requirements, this patented process leverages the unique reactivity of nitrile groups to achieve reaction yields ranging from approximately 80% to 90%. For R&D directors and procurement managers, understanding this technological shift is crucial for securing a reliable pharmaceutical intermediates supplier capable of delivering high-quality active ingredients consistently.
The significance of this patent extends beyond mere chemical novelty; it represents a strategic advancement in supply chain stability for antifungal medications. By introducing a stable, crystallizable intermediate (Formula VIa), the process allows for rigorous quality control checkpoints before the final reduction step. This capability is essential for mitigating risks associated with batch-to-batch variability and ensuring that the final product meets stringent regulatory standards. Furthermore, the method reduces reliance on extensive chromatographic purification, which is often a bottleneck in large-scale manufacturing. As we delve into the technical specifics, it becomes evident that this route offers a compelling value proposition for stakeholders focused on cost reduction in pharmaceutical intermediate manufacturing and the commercial scale-up of complex peptides.
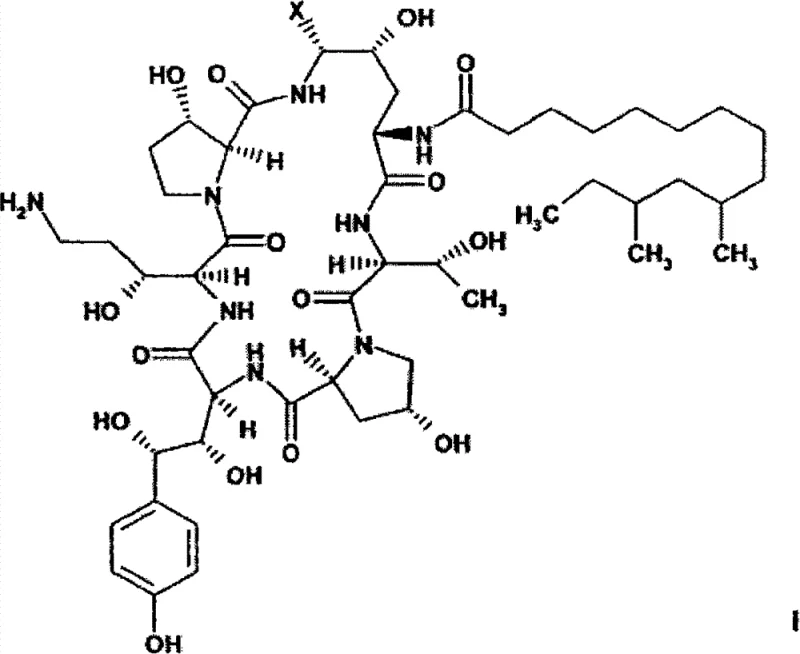
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of caspofungin from Pneumocandin B0 has been plagued by inefficiencies inherent in direct amide reduction strategies. Prior art, such as that described in US5,552,521 and WO96/24613, typically involves reducing the primary amide functional group directly to an amine, a transformation that often suffers from poor conversion rates. Reported yields for these reduction steps hover around 47%, necessitating the processing of significantly larger quantities of starting material to achieve desired output volumes. Moreover, these conventional routes frequently require the use of protecting groups, such as phenylboronic esters, to manage regioselectivity, adding multiple synthetic steps and increasing the overall cost of goods. The need for strictly anhydrous conditions and the reliance on multiple chromatographic purification stages further exacerbate the economic and operational burdens, making these methods suboptimal for industrial-scale production where throughput and consistency are paramount.
The Novel Approach
In stark contrast, the methodology outlined in CN101305018B circumvents these bottlenecks by converting the amide functionality into a nitrile group prior to reduction. This strategic modification transforms the chemical landscape of the intermediate, enabling a catalytic hydrogenation process that is both highly selective and efficient. The novel route proceeds through a dehydrated intermediate (Formula IV), which is subsequently functionalized to introduce the necessary side chain before the final reduction. This sequence allows for reaction yields to reach impressive levels of 80% to 90%, nearly doubling the efficiency of older techniques. Additionally, the process eliminates the strict requirement for anhydrous environments during the critical reduction phase, utilizing solvent systems like isopropanol and water mixtures. This flexibility not only simplifies operational protocols but also significantly enhances the safety and scalability of the manufacturing process, positioning it as a superior choice for modern pharmaceutical production facilities.
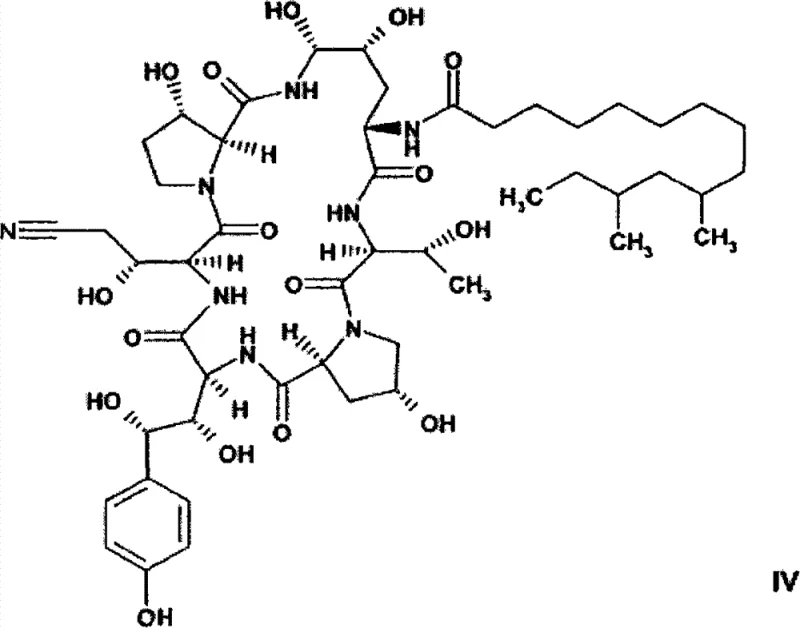
Mechanistic Insights into Selective Nitrile Hydrogenation
The core of this technological breakthrough lies in the selective catalytic hydrogenation of the nitrile group in the presence of other sensitive functionalities, specifically the aminal group at the C5-ornithine position. Traditional reduction methods often struggle to differentiate between various reducible groups, leading to side reactions and impurity formation. However, the patented process utilizes specific catalysts, such as Rh/Al2O3 or palladium on carbon, which demonstrate remarkable chemoselectivity. Under optimized conditions involving hydrogen pressure ranging from atmospheric pressure to 20 bar, the nitrile moiety is reduced to the primary amine without compromising the integrity of the macrocyclic structure. The presence of acids like acetic acid in the reaction mixture further suppresses the formation of unwanted by-products, ensuring a clean conversion profile. This mechanistic precision is critical for maintaining the stereochemical integrity of the complex peptide backbone, which is essential for the biological activity of the final antifungal agent.
Furthermore, the process incorporates a sophisticated impurity control mechanism centered around the crystallization of intermediate salts. The formation of the monoacetate salt of Formula VI (Formula VIa) serves as a pivotal purification step. Crystallization effectively removes difficult-to-separate impurities, such as the C-35 epimer at the homotyrosine moiety, which are notoriously challenging to eliminate via chromatography alone. By leveraging the differential solubility properties of the desired intermediate versus its stereoisomers, the process achieves a high degree of purity before the final synthetic step. This proactive approach to impurity management reduces the burden on downstream purification processes and ensures that the final caspofungin product meets rigorous quality specifications. For technical teams, this highlights the importance of intermediate isolation strategies in achieving overall process robustness and product consistency.
How to Synthesize Caspofungin Efficiently
The synthesis of caspofungin via this novel pathway involves a sequence of well-defined chemical transformations starting from the natural product Pneumocandin B0. The initial step entails the dehydration of the primary amide to form the corresponding nitrile intermediate using a dehydrating agent such as cyanuric chloride. Following this, the nitrile intermediate undergoes nucleophilic substitution with thiophenol and subsequent displacement with ethylenediamine to install the requisite side chain. The resulting precursor is then subjected to catalytic hydrogenation to yield the final active pharmaceutical ingredient. This streamlined workflow minimizes the number of unit operations and maximizes material throughput. Detailed standardized synthesis steps see the guide below.
- Dehydrate Pneumocandin B0 using cyanuric chloride to form the nitrile intermediate (Formula IV).
- React the nitrile intermediate with thiophenol and subsequently with ethylenediamine to generate the stable amine precursor (Formula VI).
- Perform selective catalytic hydrogenation of the nitrile group using Rh/Al2O3 or Pd/C to obtain Caspofungin with high yield.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this advanced synthesis route translates into tangible operational benefits that extend far beyond the laboratory. The primary advantage lies in the substantial improvement in reaction yields, which directly correlates to a more efficient utilization of raw materials. By achieving yields of 80% to 90% compared to the historical 47%, manufacturers can significantly reduce the volume of starting material required per kilogram of final product. This efficiency gain drives down the cost of goods sold and mitigates the risk associated with the sourcing of expensive fermentation-derived precursors like Pneumocandin B0. Additionally, the elimination of complex protecting group strategies simplifies the supply chain for reagents, reducing the number of specialized chemicals that need to be sourced, stored, and managed, thereby streamlining inventory logistics.
- Cost Reduction in Manufacturing: The economic impact of this process is profound, primarily driven by the drastic simplification of the purification workflow. Conventional methods often rely on multiple chromatographic steps to achieve acceptable purity levels, which are both time-consuming and expensive due to the high cost of chromatography media and solvents. In contrast, the ability to purify key intermediates through crystallization offers a far more cost-effective alternative. Crystallization is inherently scalable and consumes fewer resources than column chromatography, leading to significant operational expenditure savings. Furthermore, the higher reaction yields mean less waste generation, which reduces the costs associated with waste disposal and environmental compliance, contributing to a leaner and more sustainable manufacturing model.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by complex processes that are sensitive to minor variations in conditions. The robustness of this new method, particularly its tolerance for aqueous solvent systems during the reduction step, makes it less prone to failure due to moisture ingress compared to strictly anhydrous protocols. This resilience ensures more consistent batch success rates, reducing the likelihood of production delays. Moreover, the availability of a stable, isolable intermediate (Formula VIa) allows manufacturers to build strategic stockpiles of semi-finished goods. This decoupling of synthesis stages provides a buffer against supply disruptions, ensuring that the final API can be produced on demand even if there are temporary fluctuations in the availability of upstream materials.
- Scalability and Environmental Compliance: Scaling chemical processes from the bench to commercial production often reveals hidden complexities, but this route is designed with industrial applicability in mind. The use of common catalysts like palladium on carbon and rhodium on alumina, which are widely available and well-understood in the industry, facilitates straightforward technology transfer. The reduction in solvent usage and the shift away from hazardous protecting groups align with green chemistry principles, easing the regulatory burden associated with environmental permits. This alignment not only future-proofs the manufacturing site against tightening environmental regulations but also enhances the corporate sustainability profile, which is increasingly important for partnerships with major pharmaceutical companies committed to responsible sourcing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding these nuances is vital for evaluating the feasibility of adopting this process within existing manufacturing frameworks.
Q: What is the primary advantage of the nitrile reduction route over traditional amide reduction?
A: The nitrile reduction route described in CN101305018B achieves significantly higher reaction yields (approximately 80% to 90%) compared to traditional amide reduction methods which typically yield around 47% to 61%, while also simplifying purification.
Q: How does this process ensure high purity of the final API?
A: The process utilizes a crystallizable intermediate (Formula VIa monoacetate) which allows for the effective removal of difficult impurities like the C-35 epimer through crystallization rather than relying solely on chromatography.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the method is designed for industrial scalability, utilizing robust catalytic hydrogenation conditions and avoiding strictly anhydrous environments, which facilitates easier scale-up and cost reduction in pharmaceutical intermediate manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Caspofungin Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic routes to maintain competitiveness in the global pharmaceutical market. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN101305018B are fully realized in practice. We are equipped with rigorous QC labs and adhere to stringent purity specifications to guarantee that every batch of caspofungin intermediate or API meets the highest international standards. Our commitment to quality is backed by state-of-the-art infrastructure capable of handling complex catalytic hydrogenation and crystallization processes safely and efficiently.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific supply chain requirements. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the potential economic advantages tailored to your volume needs. We encourage you to contact us for specific COA data and route feasibility assessments, allowing you to make informed decisions based on concrete technical evidence. Partnering with us ensures access to a reliable caspofungin supplier dedicated to innovation, quality, and long-term supply security.
