Advanced Manufacturing Strategies for High-Purity Ranolazine Intermediates
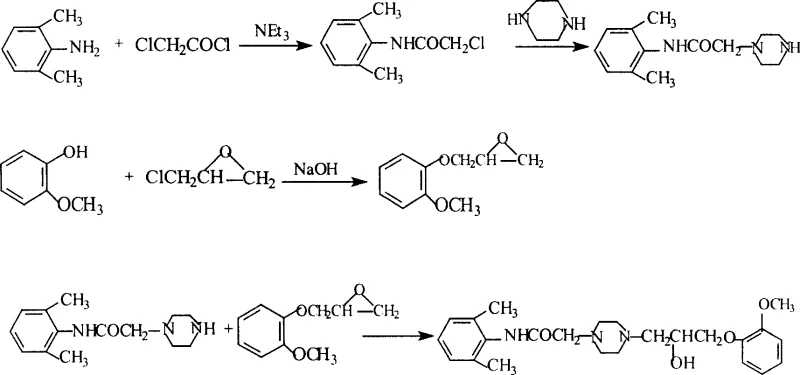
The pharmaceutical landscape for antianginal therapies continues to evolve, with Ranolazine standing out as a pivotal metabolic modulator for treating chronic angina. Patent CN1915982A introduces a refined synthetic methodology that addresses historical bottlenecks in the production of this critical active pharmaceutical ingredient (API). By optimizing the classical amidation and nucleophilic substitution pathways, this technology offers a robust framework for manufacturing high-purity Ranolazine intermediates. The core innovation lies in the strategic manipulation of reaction times, solvent systems, and purification protocols, specifically targeting the conversion of 2,6-dimethylaniline into the final piperazine-linked structure. For R&D directors and process chemists, this patent represents a significant opportunity to enhance process mass intensity (PMI) while maintaining stringent quality standards required for global regulatory submission.
Furthermore, the detailed operational parameters provided in the patent, such as specific vacuum distillation cuts and recrystallization solvent ratios, provide a clear roadmap for scaling this chemistry from laboratory benchtop to commercial production. The emphasis on minimizing side reactions during the epoxide ring-opening step is particularly noteworthy, as it directly impacts the impurity profile of the final drug substance. As a reliable Ranolazine intermediate supplier, understanding these nuanced process improvements is essential for ensuring supply chain continuity and cost-effectiveness in the competitive cardiovascular therapeutic market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Ranolazine has been plagued by inefficiencies inherent in early-generation routes, often characterized by excessive reaction steps and suboptimal yield profiles. Conventional methods, such as those relying on the initial formation of complex chloroethyl amines followed by lengthy cyclization sequences, introduce unnecessary complexity and increase the risk of generating difficult-to-remove impurities. These traditional pathways often suffer from poor atom economy, requiring large volumes of solvents and generating significant waste streams that complicate environmental compliance and drive up manufacturing costs. Additionally, the lack of specific purification steps for key intermediates in older protocols frequently leads to carryover impurities that poison downstream catalysts or interfere with final crystallization, resulting in inconsistent batch quality and extended production lead times.
The Novel Approach
In contrast, the methodology outlined in CN1915982A streamlines the synthesis by focusing on the optimization of the most direct route: the sequential amidation of 2,6-dimethylaniline followed by piperazine displacement and epoxide coupling. This novel approach drastically simplifies the workflow by eliminating redundant protection and deprotection steps found in alternative syntheses. A key differentiator is the introduction of a hexane refinement step for the chloroacetamide intermediate, which effectively removes unreacted starting materials and byproducts before they can interfere with the sensitive piperazine substitution. Furthermore, the utilization of methanol as the sole solvent for the final coupling reaction represents a significant departure from more expensive or toxic solvent systems, aligning the process with green chemistry principles while simultaneously reducing raw material procurement costs.
Mechanistic Insights into Nucleophilic Substitution and Epoxide Ring Opening
The chemical elegance of this synthesis relies heavily on the precise control of nucleophilic substitution reactions, particularly the displacement of the chloride in 2-chloro-N-(2,6-xylyl)acetamide by piperazine. Mechanistically, this step requires careful management of stoichiometry and temperature to prevent over-alkylation or polymerization of the piperazine ring. The patent specifies the use of excess piperazine in an ethanol medium under reflux, which drives the equilibrium towards the desired mono-substituted product while suppressing the formation of bis-alkylated impurities. The presence of the electron-withdrawing amide carbonyl adjacent to the chloromethyl group enhances the electrophilicity of the alpha-carbon, facilitating the nucleophilic attack by the secondary amine of the piperazine ring. This electronic activation is crucial for achieving high conversion rates without the need for harsh basic conditions that could degrade the sensitive aromatic amine moiety.
Following the formation of the piperazine intermediate, the subsequent ring-opening of the 3-(2-methoxyphenoxy)-1,2-epoxypropane epoxide is the defining step that constructs the final pharmacophore. This reaction proceeds via a nucleophilic attack by the secondary nitrogen of the piperazine ring on the less hindered terminal carbon of the epoxide. The regioselectivity of this ring opening is vital, as attack at the internal carbon would yield a regioisomer with different pharmacological properties. The use of methanol as a solvent likely assists in stabilizing the transition state through hydrogen bonding, while the reflux conditions provide the necessary thermal energy to overcome the activation barrier of the strained three-membered ring. Rigorous control of this step ensures that the hydroxyl group is positioned correctly at the beta-position relative to the ether oxygen, a structural feature essential for the drug's metabolic modulation activity.
How to Synthesize Ranolazine Efficiently
The synthesis of Ranolazine via this optimized pathway involves a sequence of five distinct operational stages, beginning with the preparation of the epoxide building block and concluding with a highly specific recrystallization protocol. The process is designed to maximize throughput while minimizing the accumulation of genotoxic impurities and heavy metal residues. Detailed below is the strategic overview of the synthesis, highlighting the critical control points identified in the patent literature. For process engineers and scale-up teams, adhering to the specified temperature ranges and solvent ratios is paramount to replicating the high yields reported in the experimental examples. The following guide outlines the standardized procedure for executing this chemistry effectively.
- Prepare 3-(2-methoxyphenoxy)-1,2-epoxypropane by reacting o-methoxyphenol with epichlorohydrin under alkaline conditions, followed by vacuum distillation.
- Synthesize 2-chloro-N-(2,6-xylyl)acetamide via low-temperature amidation of 2,6-dimethylaniline with chloroacetyl chloride, purifying the crude product with hexane.
- React the chloroacetamide intermediate with excess piperazine in ethanol to form the piperazine derivative, followed by extraction and ether refining.
- Couple the piperazine intermediate with the epoxide derivative in methanol under reflux to generate the crude Ranolazine base.
- Purify the final product through recrystallization using an ethanol and ethyl acetate mixture to achieve high pharmaceutical grade purity.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this optimized synthetic route offers tangible benefits that extend beyond mere chemical yield. The reliance on commodity chemicals such as 2,6-dimethylaniline, chloroacetyl chloride, and piperazine ensures a stable and diversified supply base, mitigating the risks associated with sourcing exotic or single-source reagents. The simplification of the solvent system, particularly the shift towards methanol and ethanol/ethyl acetate mixtures, facilitates easier solvent recovery and recycling, which significantly lowers the total cost of ownership for the manufacturing process. Moreover, the robustness of the purification steps reduces the likelihood of batch failures, thereby enhancing overall supply chain reliability and ensuring consistent delivery schedules for downstream API manufacturers.
- Cost Reduction in Manufacturing: The elimination of complex multi-step sequences and the use of cost-effective solvents like methanol for the coupling reaction contribute to a leaner manufacturing process. By removing the need for expensive catalysts or specialized reagents often required in alternative routes, the overall variable cost per kilogram of product is substantially decreased. Additionally, the improved yield in the epoxide formation step, achieved through optimized reflux times and vacuum distillation parameters, maximizes the utility of raw materials, further driving down the cost of goods sold (COGS) without compromising on quality standards.
- Enhanced Supply Chain Reliability: The use of widely available starting materials reduces dependency on niche suppliers, creating a more resilient supply chain capable of withstanding market fluctuations. The streamlined nature of the synthesis, with fewer unit operations and shorter reaction times, allows for faster batch turnover and increased production capacity within existing infrastructure. This agility enables manufacturers to respond more rapidly to changes in market demand, ensuring that critical cardiovascular medications remain available to patients without interruption due to production bottlenecks.
- Scalability and Environmental Compliance: The process is inherently scalable, as demonstrated by the use of standard unit operations like reflux, filtration, and distillation that are easily transferred from pilot plant to commercial scale. The reduction in solvent diversity and the avoidance of halogenated solvents in the final steps simplify waste stream management and lower the environmental footprint of the facility. This alignment with green chemistry principles not only reduces disposal costs but also ensures compliance with increasingly stringent environmental regulations, safeguarding the long-term viability of the manufacturing site.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Ranolazine synthesis technology. These insights are derived directly from the experimental data and process descriptions provided in the patent documentation, offering clarity on critical process parameters and quality control measures. Understanding these details is essential for technical teams evaluating the feasibility of adopting this route for commercial production.
Q: What are the critical purification steps in this Ranolazine synthesis route?
A: The patent highlights two critical purification stages: the refinement of 2-chloro-N-(2,6-xylyl)acetamide using hexane to remove impurities before the piperazine step, and the final recrystallization of Ranolazine using a specific ethanol/ethyl acetate (2:1) ratio to maximize yield and crystal quality.
Q: How does this method improve upon conventional Ranolazine manufacturing?
A: This method optimizes the conventional Route 1 by shortening the reflux time for epoxide synthesis to 2 hours and utilizing methanol as a cost-effective solvent for the final coupling step, thereby reducing overall processing time and solvent costs compared to longer, multi-step alternatives.
Q: What are the key reaction conditions for the epoxide intermediate formation?
A: The formation of 3-(2-methoxyphenoxy)-1,2-epoxypropane requires a reflux reaction of 2 hours followed by vacuum distillation collecting the fraction at 121-124°C under 2KPa pressure, which ensures high purity suitable for subsequent nucleophilic attacks.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ranolazine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and compliant manufacturing processes in the pharmaceutical industry. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical advantages of patents like CN1915982A are fully realized in practical application. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of Ranolazine intermediate meets the highest global standards for safety and efficacy. Our commitment to technical excellence allows us to navigate the complexities of process optimization, delivering products that support the development of life-saving cardiovascular therapies.
We invite potential partners to engage with our technical procurement team to discuss how our manufacturing capabilities can support your specific supply chain needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our optimized processes can reduce your overall procurement expenses. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project requirements, ensuring a seamless transition from development to commercial supply.
