Optimized Industrial Synthesis of Ranolazine Intermediates for Scalable Pharmaceutical Manufacturing
Optimized Industrial Synthesis of Ranolazine Intermediates for Scalable Pharmaceutical Manufacturing
The pharmaceutical landscape for antianginal therapies has been significantly shaped by the development of Ranolazine, a novel metabolic regulator that selectively inhibits partial fatty acid oxidation to improve myocardial efficiency. As global demand for cardiovascular medications rises, the efficiency of synthesizing high-purity Ranolazine intermediates becomes a critical bottleneck for supply chains. Patent CN1915982A, filed in early 2007, presents a transformative methodology that addresses the longstanding challenges of low yields and complex purification associated with traditional synthesis routes. This technical disclosure outlines a refined five-step process that optimizes reaction times, solvent systems, and crystallization protocols, offering a robust framework for commercial scale-up of complex pharmaceutical intermediates. By strictly controlling parameters such as distillation cuts and pH adjustments during workup, this method ensures consistent product quality while streamlining the manufacturing workflow for industrial applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
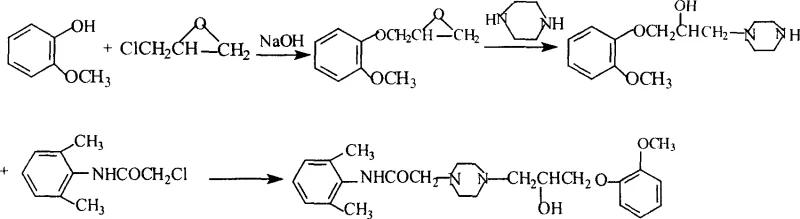
Historically, the synthesis of Ranolazine has relied on several distinct pathways, each plagued by specific inefficiencies that hinder large-scale production. Conventional Route 1, often cited in earlier literature, involves the amidation of 2,6-xylidine followed by N-monoalkylation with piperazine and subsequent coupling with an epoxide derivative. However, legacy implementations of this route frequently suffer from suboptimal yields due to incomplete reactions and the formation of difficult-to-remove side products. Furthermore, traditional purification methods often lack the specificity required to meet stringent pharmacopeial standards without significant material loss. Alternative pathways, such as Route 2 which reverses the order of piperazine introduction, or Route 3 which utilizes longer synthetic sequences involving azetidinone intermediates, introduce additional complexity. These older methods typically require harsher reaction conditions, multiple solvent exchanges, and extended reaction times, all of which contribute to elevated operational expenditures and reduced overall throughput in a manufacturing setting.
The Novel Approach
The methodology detailed in CN1915982A represents a strategic evolution of the conventional Route 1, introducing targeted modifications that dramatically enhance process efficiency. A key innovation lies in the optimization of the epoxide synthesis step, where the reflux time is rigorously controlled to exactly 2 hours, preventing degradation while ensuring complete conversion. The patent specifies a precise vacuum distillation cut at 121-124°C under 2KPa pressure, which isolates the 3-(2-methoxyphenoxy)-1,2-epoxypropane intermediate with a yield exceeding 80%. Furthermore, the introduction of a hexane refining step for the chloroacetamide intermediate serves as a critical quality gate, removing impurities that would otherwise propagate through the synthesis. In the final coupling stage, the exclusive use of methanol as a solvent simplifies the reaction matrix compared to mixed solvent systems, facilitating easier solvent recovery and reducing waste generation. This holistic optimization results in a streamlined process that balances high yield with operational simplicity.
Mechanistic Insights into Amidation and Epoxide Ring Opening
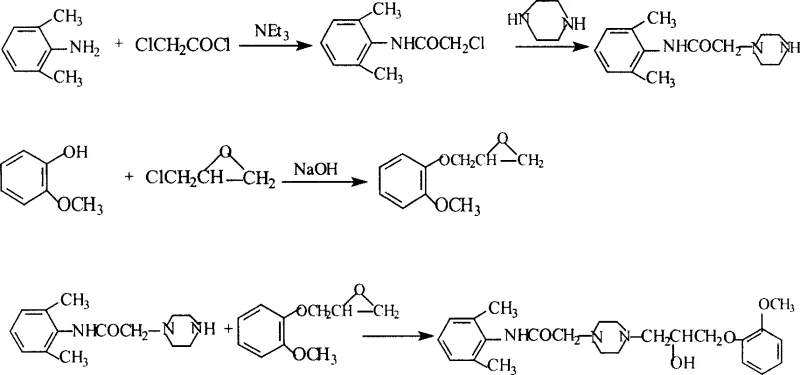
The core of this synthesis relies on two fundamental organic transformations: nucleophilic acyl substitution and epoxide ring opening. The initial formation of 2-chloro-N-(2,6-xylyl)ethanamide proceeds via a Schotten-Baumann type amidation, where 2,6-xylidine acts as the nucleophile attacking chloroacetyl chloride. The presence of triethylamine is crucial here, serving as an acid scavenger to neutralize the generated HCl, thereby driving the equilibrium forward and preventing the protonation of the aniline nitrogen which would deactivate it. The steric hindrance provided by the ortho-methyl groups on the xylidine ring necessitates careful temperature control, specifically maintaining the reaction below 0°C during the addition of the acid chloride to minimize di-acylation or polymerization side reactions. Following this, the nucleophilic displacement of the chloride by piperazine requires elevated temperatures (reflux in ethanol) to overcome the activation energy barrier, resulting in the formation of the secondary amine linkage essential for the drug's pharmacophore.
The final assembly of the Ranolazine molecule involves the ring-opening of the glycidyl ether derivative by the secondary amine of the piperazine intermediate. This reaction is mechanistically driven by the nucleophilic attack of the piperazine nitrogen on the less hindered carbon of the epoxide ring. The regioselectivity of this attack is influenced by the electronic properties of the adjacent methoxyphenoxy group, which stabilizes the transition state. The use of methanol as a solvent in this step is particularly advantageous; it acts not only as a medium but potentially participates in hydrogen bonding that stabilizes the developing alkoxide intermediate before protonation yields the final beta-hydroxy amine structure. The rigorous control of stoichiometry and the specific recrystallization protocol using ethanol and ethyl acetate ensure that the final product precipitates in its thermodynamically stable crystalline form, effectively excluding structural isomers and unreacted starting materials from the lattice.
How to Synthesize Ranolazine Efficiently
The execution of this optimized synthesis requires strict adherence to the defined operational parameters to replicate the high yields reported in the patent data. The process is divided into five distinct stages, beginning with the preparation of the epoxide building block and concluding with the final recrystallization of the active pharmaceutical ingredient. Each step has been engineered to maximize material throughput while minimizing the accumulation of impurities that could complicate downstream processing. Operators must pay particular attention to the vacuum distillation specifications and the pH adjustments during the aqueous workups, as these are critical control points for product quality. For a comprehensive breakdown of the exact reagent quantities, mixing rates, and safety protocols required for implementation, please refer to the standardized synthesis guide below.
- Synthesize 3-(2-methoxyphenoxy)-1,2-epoxypropane by reacting o-methoxyphenol with epichlorohydrin in dioxane/water with NaOH, followed by distillation at 121-124°C/2KPa.
- Prepare 2-chloro-N-(2,6-xylyl)ethanamide by reacting 2,6-xylidine with chloroacetyl chloride in toluene/triethylamine at 0°C, followed by hexane purification.
- Form N-(2,6-xylyl)-2-(1-piperazinyl)ethanamide by refluxing the chloroacetamide intermediate with excess piperazine in ethanol, adjusting pH to 8-9, and refining with ether.
- Couple the piperazine intermediate with the epoxide derivative in methanol under reflux for 3 hours to generate the crude Ranolazine product.
- Purify the crude product via recrystallization using an ethanol/ethyl acetate (2: 1) solvent system with activated carbon decolorization to achieve high purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this optimized synthesis route offers tangible strategic benefits beyond mere technical superiority. The simplification of the solvent profile, particularly the shift to methanol for the final coupling step, directly correlates to a reduction in raw material procurement complexity and cost. By eliminating the need for exotic or high-boiling solvents that require energy-intensive recovery processes, manufacturers can achieve significant operational savings. Furthermore, the enhanced purity of the intermediates, achieved through the novel hexane refining and specific recrystallization techniques, reduces the burden on quality control laboratories and minimizes the risk of batch rejection. This reliability translates into a more predictable production schedule, allowing supply chain planners to maintain leaner inventory levels without compromising on delivery commitments to downstream API manufacturers.
- Cost Reduction in Manufacturing: The process achieves cost efficiency primarily through solvent optimization and yield improvement. By utilizing methanol instead of more expensive solvent blends for the critical coupling reaction, the direct material cost per kilogram of product is lowered. Additionally, the high yield of the epoxide intermediate (>80%) and the efficient recovery of the final product via ethanol/ethyl acetate recrystallization mean that less starting material is wasted. The elimination of complex purification steps found in older routes further reduces labor hours and utility consumption, contributing to a substantially lower cost of goods sold (COGS) for the final intermediate.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as 2,6-xylidine, piperazine, and epichlorohydrin ensures that the supply chain is resilient to market fluctuations. These raw materials are widely available from multiple global suppliers, mitigating the risk of single-source bottlenecks. The robustness of the reaction conditions, which do not require cryogenic temperatures beyond simple ice baths or high-pressure equipment, allows for production in a wider range of manufacturing facilities. This flexibility enhances the continuity of supply, ensuring that reducing lead time for high-purity pharmaceutical intermediates becomes a achievable operational reality rather than just a goal.
- Scalability and Environmental Compliance: From an environmental and scaling perspective, the process is designed for seamless transition from pilot to commercial scale. The use of standard unit operations like filtration, distillation, and crystallization aligns with existing infrastructure in most fine chemical plants. Moreover, the reduction in solvent variety simplifies waste stream management, making it easier to implement effective recycling programs and adhere to increasingly strict environmental regulations. The high selectivity of the reactions minimizes the generation of hazardous byproducts, supporting a greener manufacturing footprint that aligns with modern corporate sustainability mandates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Ranolazine synthesis technology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for decision-making. Understanding these nuances is essential for R&D teams evaluating technology transfer and procurement officers assessing vendor capabilities. The answers highlight the specific advantages of this route over legacy methods, focusing on yield, purity, and operational feasibility.
Q: How does the optimized solvent system in Step 4 impact production costs?
A: The optimized process utilizes methanol exclusively as the solvent for the final coupling reaction in Step 4. This eliminates the need for more expensive or complex solvent mixtures used in conventional methods, significantly reducing raw material procurement costs and simplifying solvent recovery operations.
Q: What specific purification techniques are employed to ensure intermediate quality?
A: The protocol introduces a critical hexane refining step for the 2-chloro-N-(2,6-xylyl)ethanamide intermediate to remove impurities before the piperazine coupling. Additionally, the final Ranolazine product undergoes rigorous recrystallization using a specific ethanol/ethyl acetate ratio to ensure stringent purity specifications required for API manufacturing.
Q: Can this synthesis route be scaled for commercial API production?
A: Yes, the reaction conditions utilize standard industrial solvents like toluene, ethanol, and methanol, and operate at manageable temperatures (reflux or ice bath). The simplified workup procedures, such as direct filtration and standard distillation cuts, indicate high feasibility for scaling from pilot batches to multi-ton commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ranolazine Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires deep technical expertise and robust manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of optimized synthesis routes like CN1915982A are fully realized in practice. We maintain stringent purity specifications across all our product lines, supported by rigorous QC labs equipped with advanced analytical instrumentation to verify every batch against the highest industry standards. Our commitment to quality ensures that the Ranolazine intermediates we supply are perfectly suited for the demanding requirements of final API synthesis.
We invite potential partners to engage with our technical procurement team to discuss how our manufacturing capabilities can support your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic advantages of sourcing from our optimized production lines. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your volume requirements, ensuring a seamless integration of our high-quality intermediates into your global supply chain.
