Scalable Synthesis of Mitiglinide Intermediate: Technical Breakthroughs and Commercial Viability
Introduction to Advanced Mitiglinide Intermediate Manufacturing
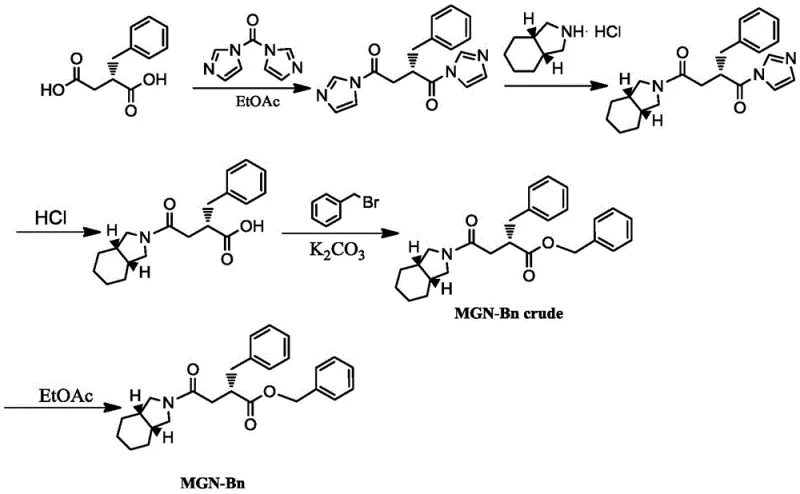
The global demand for effective type 2 diabetes treatments continues to drive innovation in the synthesis of key pharmaceutical intermediates, specifically those required for meglitinide class drugs like Mitiglinide. As a rapid-acting insulin secretagogue, Mitiglinide requires precise stereochemical control and high purity to ensure therapeutic efficacy and safety. The patent CN106631978B introduces a robust and industrially viable synthesis process for the critical intermediate, benzyl 2-(S)-benzyl-4-oxo-(cis-perhydroisoindol-2-yl) butyrate. This technical disclosure represents a significant evolution in process chemistry, moving away from hazardous solvents and complex purification trains towards a streamlined, ethyl acetate-based protocol. For R&D directors and procurement strategists, understanding the nuances of this pathway is essential for securing a reliable pharmaceutical intermediate supplier capable of delivering consistent quality at scale.
This synthesis route leverages 1,1'-carbonyldiimidazole (CDI) as a superior coupling agent, facilitating the formation of the amide bond under mild conditions while preserving the chiral integrity of the S-benzylsuccinic acid precursor. The subsequent esterification and purification steps are meticulously designed to minimize impurity carryover, addressing a common bottleneck in API manufacturing. By optimizing reaction temperatures and employing a strategic acid-base workup, the process achieves high yields without compromising on environmental safety or operational complexity. This report analyzes the technical merits of this approach and its implications for cost-effective, large-scale production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for peptidomimetic intermediates often rely on carbodiimide coupling reagents such as DCC (dicyclohexylcarbodiimide) or EDC, frequently in conjunction with additives like HOBt or DMAP. While effective on a laboratory scale, these methods present substantial challenges when translated to commercial manufacturing. The primary drawback is the generation of insoluble urea byproducts, such as dicyclohexylurea (DCU), which are notoriously difficult to remove completely from the reaction mixture. Residual DCU can act as a persistent impurity, complicating downstream crystallization and potentially affecting the safety profile of the final API. Furthermore, conventional processes often utilize chlorinated solvents like dichloromethane or polar aprotic solvents like DMF, which pose significant environmental and occupational health risks, increasing the cost of waste disposal and solvent recovery.
The Novel Approach
The methodology outlined in patent CN106631978B offers a transformative alternative by utilizing CDI in ethyl acetate, a greener and more manageable solvent system. The activation of S-2-benzylsuccinic acid with CDI generates an acyl imidazole intermediate that is highly reactive towards nucleophilic attack by cis-perhydroisoindole, yet produces imidazole as a water-soluble byproduct. This fundamental shift in chemistry eliminates the need for difficult filtrations of solid ureas and simplifies the aqueous workup significantly. The process operates at controlled low temperatures (0-5°C) during activation to prevent racemization, followed by a温和 warming phase for coupling, ensuring optimal kinetics without thermal degradation. This novel approach not only enhances the purity profile of the crude product but also aligns with modern green chemistry principles, making it an attractive option for cost reduction in API manufacturing.
Mechanistic Insights into CDI-Mediated Amide Coupling and Esterification
The core of this synthesis lies in the efficient formation of the amide bond via an acyl imidazole intermediate. When S-2-benzylsuccinic acid reacts with 1,1'-carbonyldiimidazole, the carboxylic acid attacks the carbonyl carbon of the CDI, displacing one molecule of imidazole and forming a mixed anhydride-like species that rapidly cyclizes to the stable acyl imidazole. This activated species is electrophilic enough to react with the secondary amine of cis-perhydroisoindole hydrochloride but stable enough to allow for controlled addition, minimizing side reactions. The presence of the hydrochloride salt necessitates careful pH management or the presence of a base, although the reaction conditions described suggest the intrinsic basicity of the released imidazole or the amine itself may facilitate the deprotonation. Crucially, the mild conditions preserve the stereocenter at the benzylic position, preventing epimerization which would lead to diastereomeric impurities that are difficult to separate.
Following amide formation, the process employs a sophisticated purification strategy before the final esterification. The reaction mixture undergoes a series of pH-adjusted extractions: first acidified to remove excess amine and imidazole into the aqueous layer, then basified to extract any unreacted acid, and finally re-acidified to isolate the pure amide-acid intermediate. This 'ping-pong' extraction technique is highly effective at scrubbing ionic impurities. The final step involves the alkylation of the carboxylic acid with benzyl bromide using potassium carbonate as a base. The use of large-particle anhydrous potassium carbonate ensures a heterogeneous reaction environment that minimizes hydrolysis of the benzyl bromide while driving the SN2 substitution to completion. The resulting benzyl ester is then crystallized directly from the reaction solvent, leveraging the solubility differences between the product and residual salts to achieve high purity.
How to Synthesize Mitiglinide Intermediate Efficiently
The synthesis of benzyl 2-(S)-benzyl-4-oxo-(cis-perhydroisoindol-2-yl) butyrate requires precise control over stoichiometry and temperature to maximize yield and optical purity. The patented procedure details a specific sequence of additions and phase separations that are critical for success. Operators must adhere strictly to the temperature ranges, particularly during the initial activation phase where exothermicity must be managed to prevent racemization. The workup procedure is equally critical, as the removal of imidazole and unreacted starting materials dictates the quality of the crude material entering the crystallization stage. For a detailed breakdown of the operational parameters, reagent ratios, and specific handling instructions, please refer to the standardized synthesis guide below.
- Activate S-2-benzylsuccinic acid with 1,1'-carbonyldiimidazole (CDI) in ethyl acetate at 0-5°C, followed by coupling with cis-perhydroisoindole hydrochloride.
- Perform rigorous acid-base workup involving pH adjustments to remove imidazole byproducts and unreacted starting materials.
- Conduct esterification using benzyl bromide and potassium carbonate under reflux, followed by crystallization and recrystallization from ethyl acetate.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthesis route offers tangible benefits for supply chain resilience and cost management. The shift to ethyl acetate as the primary solvent is a strategic move that reduces reliance on regulated chlorinated solvents, thereby lowering environmental compliance costs and simplifying solvent recovery operations. Ethyl acetate is widely available, has a favorable boiling point for distillation, and is generally recognized as a safer alternative in large-scale processing. This transition supports substantial cost savings in waste treatment and aligns with the increasingly stringent environmental regulations faced by chemical manufacturers globally. Furthermore, the high efficiency of the purification steps reduces the number of unit operations required, shortening the overall production cycle time.
- Cost Reduction in Manufacturing: The elimination of expensive coupling additives and the avoidance of difficult-to-remove solid byproducts like DCU significantly streamline the production process. By using CDI, the process generates water-soluble imidazole which is easily washed away, reducing the need for extensive chromatography or multiple recrystallizations. This efficiency translates directly into lower operational expenditures and higher throughput. Additionally, the high yield reported in the patent examples suggests a robust material balance, minimizing the loss of valuable chiral starting materials and maximizing the return on investment for raw material procurement.
- Enhanced Supply Chain Reliability: The reagents utilized in this process, including S-2-benzylsuccinic acid, CDI, and benzyl bromide, are commodity chemicals with established global supply chains. Unlike specialized catalysts or exotic reagents that may face availability bottlenecks, these materials can be sourced from multiple vendors, ensuring continuity of supply. The robustness of the reaction conditions, which tolerate slight variations in temperature and addition rates without catastrophic failure, further enhances manufacturing reliability. This stability allows for flexible production scheduling and reduces the risk of batch failures that could disrupt downstream API synthesis timelines.
- Scalability and Environmental Compliance: The process is inherently scalable due to its reliance on standard unit operations such as liquid-liquid extraction and crystallization. The use of ethyl acetate facilitates easy solvent recycling, contributing to a reduced carbon footprint and lower disposal costs. The absence of heavy metal catalysts or toxic reagents simplifies the regulatory filing process for the final drug substance, as residual solvent and impurity profiles are easier to control. This environmental compatibility positions the manufacturer as a responsible partner, capable of meeting the sustainability goals of major pharmaceutical clients while maintaining competitive pricing structures.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of this Mitiglinide intermediate. These answers are derived from the specific technical disclosures and beneficial effects outlined in the patent literature, providing clarity on process capabilities and quality standards. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this intermediate into their supply chain.
Q: What is the primary advantage of using CDI over DCC in this synthesis?
A: CDI activation avoids the formation of insoluble dicyclohexylurea (DCU) byproducts, simplifying filtration and purification while maintaining high stereochemical integrity of the S-enantiomer.
Q: How does the process ensure high purity of the Mitiglinide intermediate?
A: The process utilizes a multi-stage pH-controlled extraction strategy to selectively remove acidic and basic impurities, followed by a specific recrystallization protocol in ethyl acetate that yields >99% purity.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the use of ethyl acetate as a primary solvent, combined with mild temperature controls (0-30°C) and standard reagents like potassium carbonate, makes the process highly scalable and safe for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Mitiglinide Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development of antidiabetic therapies. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless. We are committed to delivering Mitiglinide intermediates with stringent purity specifications, utilizing rigorous QC labs to verify every batch against the highest industry standards. Our facility is equipped to handle the specific solvent systems and reaction conditions required by this advanced synthesis route, guaranteeing a consistent supply of material that meets your exacting requirements.
We invite you to collaborate with us to optimize your supply chain for Mitiglinide production. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific volume needs and logistical constraints. By partnering with us, you gain access to a transparent and responsive technical procurement team capable of supplying specific COA data and route feasibility assessments upon request. Let us help you secure a stable, cost-effective source of this vital pharmaceutical building block.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
