Advanced Asymmetric Reduction Technology for High-Purity Mitiglinide Intermediate Manufacturing
The pharmaceutical industry continuously seeks more efficient pathways for synthesizing complex chiral intermediates, particularly for anti-diabetic agents like Mitiglinide. Patent CN101346352B introduces a groundbreaking asymmetric reduction method that fundamentally alters the economic and technical landscape of producing (2S)-2-benzyl-3-(cis-hexahydro-2-isoindolinylcarbonyl)propionic acid. This technology leverages a novel class of urea-type pyrrolidine diphosphine ligands coordinated with rhodium to achieve unprecedented catalytic efficiency. Unlike previous iterations that struggled with slow kinetics and excessive noble metal consumption, this innovation enables high conversion rates and exceptional optical purity under mild conditions. For global procurement teams and R&D directors, this represents a critical opportunity to optimize the supply chain for type II diabetes medications by reducing reliance on expensive catalysts while maintaining stringent quality standards required for regulatory approval.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of optically active benzylsuccinic acid derivatives, which serve as the backbone for Mitiglinide, relied heavily on transition metal complexes such as those utilizing BPPM (2S,4S)-N-(tert-butoxycarbonyl)-4-diphenylphosphino-2-diphenylphosphinomethylpyrrolidine. While functional, these conventional carbamate-type ligands exhibited significant drawbacks when scaled for industrial application. The primary bottleneck was the sluggish reaction kinetics, which necessitated prolonged reaction times to reach acceptable conversion levels, thereby reducing overall plant throughput. Furthermore, these methods typically required low substrate-to-catalyst (S/C) molar ratios, forcing manufacturers to utilize large quantities of costly rhodium precursors to drive the reaction to completion. This inefficiency not only inflated raw material costs but also complicated downstream purification processes, as removing residual heavy metals from the final API became a resource-intensive burden. Additionally, the optical purity achieved was often insufficient for direct pharmaceutical use, requiring additional recrystallization steps that further eroded yield and increased waste generation.
The Novel Approach
The methodology disclosed in the patent data revolutionizes this process by substituting the traditional carbamate moiety with a urea-type structure within the pyrrolidine diphosphine ligand framework. This structural modification dramatically enhances the electronic and steric environment around the rhodium center, resulting in superior substrate specificity and catalytic turnover. By employing ligands such as PCPPM or DCPCPPM, the reaction proceeds with remarkable velocity, often achieving near-total conversion within approximately 4 hours even at high substrate concentrations. Crucially, this approach supports exceptionally high S/C ratios, ranging from 5,000 to 20,000, which drastically reduces the molar requirement for the precious metal catalyst. This shift allows for the production of high-purity Mitiglinide intermediates with enantiomeric excess values consistently exceeding 95% e.e., effectively eliminating the need for extensive post-reaction purification and streamlining the path from raw material to finished pharmaceutical salt.
Mechanistic Insights into Rhodium-Catalyzed Asymmetric Hydrogenation
The core of this technological advancement lies in the precise coordination chemistry between the chiral urea-type ligand and the rhodium atom. The ligand, defined by general formula (I), features a rigid pyrrolidine backbone with specific stereochemical configurations at the 2 and 4 positions, denoted as (2S, 4S). This chirality is transferred to the substrate during the hydrogenation of the carbon-carbon double bond in 2-benzylidene-3-(cis-hexahydro-2-isoindolinylcarbonyl)propionic acid. The urea linkage at the nitrogen atom of the pyrrolidine ring plays a pivotal role in stabilizing the active catalytic species and facilitating the approach of the hydrogen molecule and the olefinic substrate in a highly organized transition state. This organization ensures that hydrogen addition occurs predominantly from one face of the double bond, yielding the desired (2S) enantiomer with high fidelity. The robustness of this catalytic system is evident in its tolerance to various reaction conditions, maintaining high activity even when the catalyst loading is minimized to trace levels relative to the substrate mass.
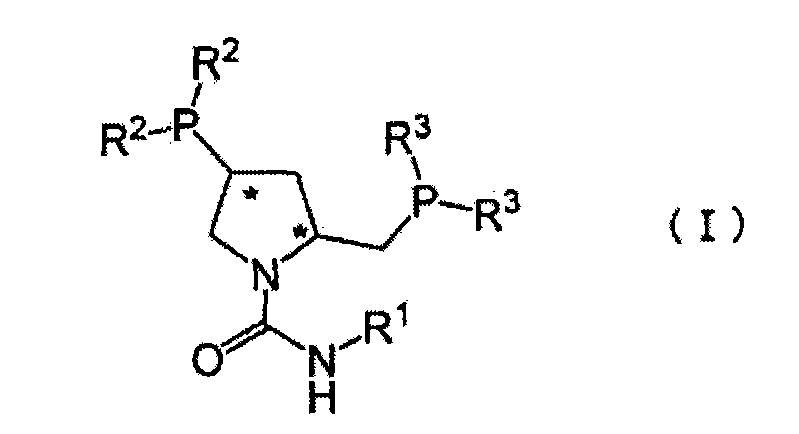
Furthermore, the mechanism accounts for the impressive impurity control observed in experimental trials. The high selectivity of the catalyst minimizes the formation of side products or the undesired (2R) enantiomer, which are common contaminants in less optimized asymmetric reductions. The ability to operate at moderate hydrogen pressures, typically between 0.1 MPa and 2 MPa, and temperatures ranging from 10°C to 50°C, suggests that the activation energy barrier for the desired pathway is significantly lowered by the unique electronic properties of the urea-substituted phosphine groups. This mechanistic efficiency translates directly to process reliability, as the reaction is less susceptible to stalling or deactivation, ensuring consistent batch-to-batch quality. For technical teams, understanding this mechanism validates the feasibility of scaling the process without the fear of unpredictable exotherms or catalyst degradation that often plague sensitive hydrogenation reactions.
How to Synthesize Mitiglinide Intermediate Efficiently
Implementing this synthesis route requires careful attention to catalyst preparation and reaction parameters to fully realize the benefits outlined in the patent literature. The process begins with the in situ or pre-formation of the active rhodium complex by combining the selected urea-type pyrrolidine diphosphine compound with a rhodium precursor such as rhodium-1,5-cyclooctadiene-chloro complex. This mixture is then introduced to a solution of the olefinic substrate in a protic solvent like methanol or ethanol. The reaction vessel is pressurized with hydrogen gas, and the mixture is agitated under controlled thermal conditions until HPLC analysis confirms the disappearance of the starting material. Following the reduction, the free acid can be directly converted into its pharmaceutically acceptable salt, such as the calcium salt dihydrate, by treatment with a base like calcium hydroxide, bypassing the need for intermediate isolation. Detailed standardized synthetic steps see the guide below.
- Prepare the asymmetric catalyst by mixing a urea-type pyrrolidine diphosphine compound (Formula I) with a rhodium compound such as rhodium-1,5-cyclooctadiene-chloro complex in a suitable solvent.
- Conduct the catalytic reduction of 2-benzylidene-3-(cis-hexahydro-2-isoindolinylcarbonyl)propionic acid under hydrogen pressure (0.1-15 MPa) at temperatures between 0°C and 150°C.
- React the resulting free acid with a basic substance like calcium hydroxide to form the final mitiglinide calcium salt dihydrate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain strategists, the adoption of this asymmetric reduction technology offers profound advantages that extend beyond simple technical metrics. The most immediate impact is seen in the drastic reduction of catalyst consumption, which directly correlates to lower variable costs per kilogram of produced intermediate. By enabling S/C ratios as high as 20,000, the process decouples production volume from the volatile pricing of noble metals, providing a more stable cost structure for long-term contracts. Additionally, the shortened reaction times enhance asset utilization, allowing manufacturing facilities to increase throughput without capital expenditure on new reactors. The high conversion rates and optical purity reduce the burden on quality control laboratories and minimize the loss of material during purification, contributing to a leaner and more sustainable manufacturing operation that aligns with modern green chemistry principles.
- Cost Reduction in Manufacturing: The ability to utilize extremely low loadings of rhodium catalyst represents a significant financial advantage, as precious metals constitute a major portion of raw material expenses in asymmetric synthesis. By increasing the substrate-to-catalyst ratio from typical industrial standards to levels exceeding 10,000, the cost attributed to the catalyst per unit of product is substantially diminished. This efficiency eliminates the economic necessity for complex catalyst recovery systems in many scenarios, further simplifying the process flow. Moreover, the high selectivity reduces the generation of chiral impurities, lowering the costs associated with chromatographic separation or multiple recrystallization steps, thereby improving the overall yield and profitability of the manufacturing campaign.
- Enhanced Supply Chain Reliability: The robustness of this catalytic system ensures consistent production schedules, mitigating the risk of delays caused by slow reaction kinetics or catalyst failure. Since the reaction proceeds rapidly to near-complete conversion, the lead time for producing batches of the Mitiglinide intermediate is significantly compressed, allowing for more responsive inventory management. The use of readily available solvents like methanol and standard hydrogenation equipment means that the process can be easily transferred between different manufacturing sites without requiring specialized or proprietary hardware. This flexibility strengthens the supply chain against disruptions, ensuring a continuous flow of high-quality intermediates to downstream API manufacturers.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated to work effectively under conditions that are safe and manageable on a large industrial scale. The moderate pressure and temperature requirements reduce energy consumption and safety risks associated with high-pressure hydrogenation. Furthermore, the high atom economy and reduced waste generation from fewer purification steps contribute to a smaller environmental footprint, aiding compliance with increasingly stringent environmental regulations. The potential for catalyst immobilization also opens avenues for continuous flow processing, which could further enhance sustainability and operational efficiency in future commercial implementations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this asymmetric reduction technology for Mitiglinide production. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a reliable basis for decision-making. Understanding these details is crucial for evaluating the feasibility of integrating this method into existing manufacturing portfolios.
Q: What represents the key advantage of the urea-type ligand over conventional BPPM ligands?
A: The urea-type pyrrolidine diphosphine ligands described in patent CN101346352B demonstrate significantly higher catalytic activity, allowing for much higher substrate-to-catalyst ratios (S/C up to 20,000) and faster reaction completion times compared to traditional carbamate-type ligands.
Q: What optical purity levels can be achieved with this asymmetric reduction method?
A: Experimental data indicates that this method consistently achieves high optical purity, with enantiomeric excess (e.e.) values ranging from 94.4% to 96.6%, ensuring the production of pharmaceutical-grade intermediates suitable for diabetes treatment formulations.
Q: Is this process scalable for industrial commercial production?
A: Yes, the process is designed for industrial applicability, utilizing robust reaction conditions such as moderate hydrogen pressures (0.5 MPa) and ambient to moderate temperatures, alongside the potential for catalyst immobilization to facilitate reuse and separation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Mitiglinide Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to maintain competitiveness in the global pharmaceutical market. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the urea-type ligand catalyzed reduction are seamlessly translated from the laboratory to the manufacturing floor. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that employ state-of-the-art analytical techniques to verify enantiomeric excess and chemical purity. Our infrastructure is designed to handle complex chiral syntheses with the utmost precision, guaranteeing a supply of intermediates that meet the exacting standards of international regulatory bodies.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific supply chain requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic implications of switching to this high-efficiency catalyst system. We encourage you to contact us to obtain specific COA data and route feasibility assessments tailored to your project timelines, ensuring that your development programs proceed without interruption and with the highest quality materials available.
