Advanced Industrial Synthesis of Topiroxostat: A Cost-Effective Route for Global Supply Chains
The pharmaceutical industry is constantly seeking robust, scalable, and cost-efficient synthetic routes for high-value active pharmaceutical ingredients (APIs) and their intermediates. A significant breakthrough in the synthesis of Topiroxostat, a potent xanthine oxidase inhibitor used for treating hyperuricemia and gout, is detailed in Chinese patent CN113173916A. This patent discloses a novel five-step preparation method that fundamentally restructures the supply chain economics of this critical molecule. By shifting away from expensive, specialized starting materials towards commodity chemicals like 4-cyanopyridine and isonicotinic acid, this technology offers a compelling value proposition for global procurement teams. The method achieves high yields and purity through a sequence of hydrazinolysis, oxidation, condensation, cyanidation, and cyclization, addressing the long-standing challenges of cost and availability that have plagued previous manufacturing protocols.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of Topiroxostat has been hindered by reliance on costly and difficult-to-source precursors. For instance, the original route developed by Fuji Drug Co., Ltd. utilizes 4-cyanopyridine N-oxide as a key building block. As illustrated in the reaction scheme below, this starting material is not only commercially scarce but also commands a premium price, which inflates the final cost of the API significantly. Furthermore, alternative methods, such as those disclosed by Shanghai Pharmaceutical Industry Research Institute, involve complex oxidation sequences of isonicotinic acid methyl esters followed by hydrazinolysis under strict nitrogen protection at elevated temperatures. These processes are operationally sensitive, prone to efficiency fluctuations, and often require rigorous safety controls due to the handling of unstable intermediates.
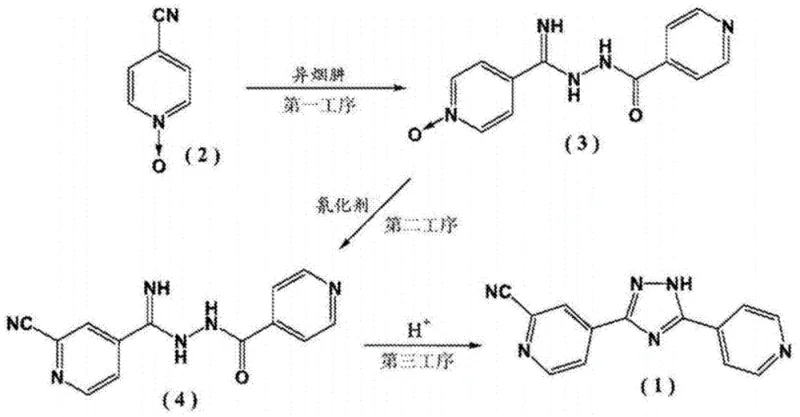
The Novel Approach
In stark contrast, the methodology presented in CN113173916A introduces a paradigm shift by utilizing 4-cyanopyridine and isonicotinic acid as the primary feedstocks. These materials are abundant, inexpensive, and widely available in the global chemical market. The innovation lies in the strategic timing of the N-oxide formation and the subsequent functional group transformations. Instead of purchasing the N-oxide, the process generates isonicotinic acid N-oxide in situ from the acid via a straightforward oxidation with hydrogen peroxide. This eliminates the supply chain bottleneck associated with specialized N-oxide reagents. Additionally, the hydrazinolysis of 4-cyanopyridine is conducted under mild alkaline conditions, avoiding the harsh thermal requirements of older methods. This streamlined approach not only simplifies the operational workflow but also significantly reduces the environmental footprint by minimizing waste generation.
Mechanistic Insights into the Five-Step Cascade Synthesis
The core of this technological advancement is a meticulously orchestrated five-step reaction cascade that ensures high atom economy and structural fidelity. The process begins with the hydrazinolysis of 4-cyanopyridine using hydrazine hydrate and sodium methoxide in ethanol at a controlled temperature of 25±5°C. This step selectively converts the nitrile group into an amidine functionality without compromising the pyridine ring integrity. Simultaneously, isonicotinic acid is oxidized using 30% hydrogen peroxide in acetic acid at 90°C to yield the corresponding N-oxide. The convergence of these two streams occurs in the condensation step, where the amidine intermediate couples with the N-oxide carboxylic acid derivative. This coupling is facilitated by modern peptide coupling reagents such as EDC/HOBt in DMF, ensuring high conversion rates and minimizing racemization or side-product formation.
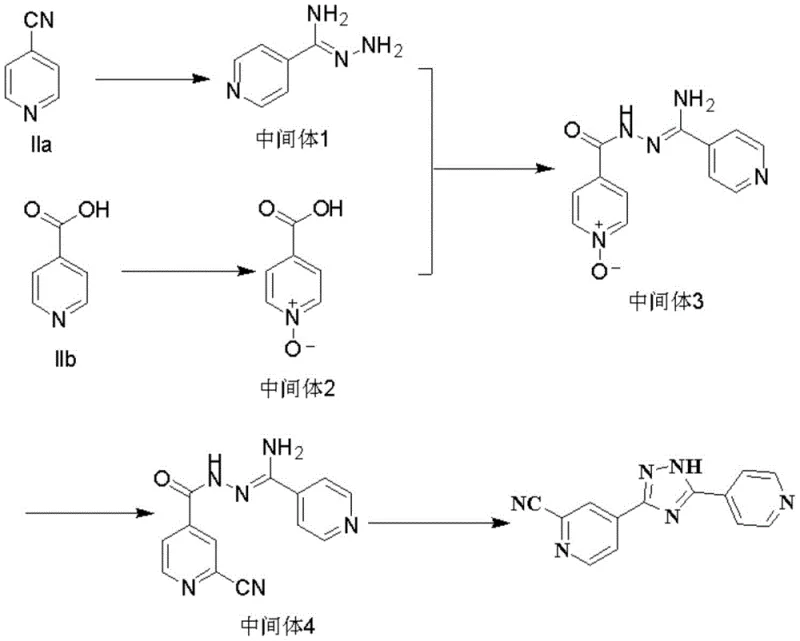
Following condensation, the critical cyanidation step transforms the N-oxide moiety into the requisite cyano group found in the final Topiroxostat structure. This is achieved using trimethylsilyl cyanide (TMSCN) or alkali metal cyanides under nitrogen protection, effectively replacing the oxygen atom on the pyridine ring. The final step involves an acid-catalyzed cyclization where the hydrazide and amidine moieties undergo intramolecular condensation to form the central 1,2,4-triazole ring. This cyclization is driven by refluxing in acetic acid, which promotes dehydration and ring closure. The result is a highly pure solid product that can be isolated via simple filtration, demonstrating the elegance and efficiency of this mechanistic design.
How to Synthesize Topiroxostat Efficiently
Implementing this synthesis route requires precise control over reaction parameters to maximize yield and safety. The process is divided into distinct operational units: the preparation of the amidine intermediate, the oxidation of the acid, the coupling reaction, the cyanide substitution, and the final ring closure. Each step has been optimized for industrial feasibility, utilizing common solvents and reagents that are easy to handle and recycle. The detailed standardized operating procedures, including specific molar ratios, temperature ramps, and workup protocols such as crystallization and washing steps, are essential for reproducibility. For a comprehensive guide on executing these steps in a GMP environment, please refer to the technical protocol outlined below.
- React 4-cyanopyridine with hydrazine hydrate and sodium methoxide in ethanol at 25°C to form the amidine intermediate.
- Oxidize 4-picolinic acid with hydrogen peroxide in acetic acid at 90°C to generate isonicotinic acid N-oxide.
- Condense the amidine intermediate with the N-oxide using EDC/HOBt coupling agents in DMF to form the hydrazide precursor.
- Perform nucleophilic substitution using trimethylsilyl cyanide to replace the N-oxide oxygen with a cyano group.
- Execute final cyclization under acidic reflux conditions to close the 1,2,4-triazole ring and isolate Topiroxostat.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthesis route translates into tangible strategic benefits beyond mere technical superiority. The primary advantage is the drastic reduction in raw material costs. By substituting expensive, niche starting materials with commodity chemicals like isonicotinic acid and 4-cyanopyridine, manufacturers can achieve substantial cost savings per kilogram of produced intermediate. This cost structure provides a buffer against market volatility and allows for more competitive pricing in the final API market. Furthermore, the reliance on widely available reagents mitigates the risk of supply disruptions, ensuring a more resilient and continuous supply chain for downstream pharmaceutical customers.
- Cost Reduction in Manufacturing: The elimination of 4-cyanopyridine N-oxide from the bill of materials removes a significant cost driver. Additionally, the use of efficient coupling agents and the high yield of the final cyclization step reduce the overall consumption of reagents and solvents. The process avoids the need for cryogenic cooling or high-pressure equipment, leading to lower energy consumption and reduced capital expenditure on specialized reactor infrastructure. These factors collectively contribute to a leaner, more cost-effective manufacturing model.
- Enhanced Supply Chain Reliability: Sourcing 4-cyanopyridine and isonicotinic acid is straightforward, as they are produced by multiple suppliers globally. This diversification of the supply base reduces dependency on single-source vendors and shortens lead times for raw material delivery. The robustness of the reaction conditions also means that production schedules are less likely to be impacted by minor variations in reagent quality or environmental conditions, ensuring consistent on-time delivery of the finished intermediate.
- Scalability and Environmental Compliance: The process generates fewer hazardous byproducts compared to traditional methods, simplifying waste treatment and disposal. The solvents used, such as ethanol and acetic acid, are relatively benign and can be recovered and recycled efficiently. The scalability of the process is evidenced by the use of standard unit operations like filtration and crystallization, which are easily transferred from pilot scale to multi-ton commercial production without significant re-engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Topiroxostat synthesis method. These answers are derived directly from the experimental data and beneficial effects described in the patent literature, providing clarity on the feasibility and advantages of this route for potential partners and licensees.
Q: Why is this new synthesis route more cost-effective than the Japanese Fuji method?
A: The conventional Japanese method relies on 4-cyanopyridine N-oxide as a starting material, which is commercially scarce and expensive. This new patent utilizes readily available 4-cyanopyridine and isonicotinic acid, generating the necessary N-oxide functionality in situ from cheap bulk chemicals, drastically reducing raw material costs.
Q: What are the critical quality control points in this manufacturing process?
A: Critical control points include the temperature maintenance during the hydrazinolysis step (25±5°C) to prevent side reactions, and the precise pH adjustment during the workup of the cyanidation step. The final acid-catalyzed cyclization requires strict reflux time control to ensure complete ring closure and high purity of the final API intermediate.
Q: Is this process scalable for multi-ton production?
A: Yes, the process is designed for industrial scalability. It avoids cryogenic conditions and uses common solvents like ethanol, DMF, and acetic acid. The exothermic reactions are manageable, and the isolation steps involve standard crystallization and filtration, making it highly suitable for large-scale commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Topiroxostat Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this advanced synthesis route for the global gout medication market. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and reliability. Our facilities are equipped with state-of-the-art reactors and stringent purity specifications are maintained through our rigorous QC labs, guaranteeing that every batch of Topiroxostat intermediate meets the highest international standards. We are committed to delivering high-purity pharmaceutical intermediates that empower your drug development and commercialization efforts.
We invite you to collaborate with us to leverage this cost-effective technology for your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can drive value and efficiency for your organization.
