Advanced Catalytic Oxidation Strategy for Scalable Pyroxasulfone Manufacturing
The global demand for high-efficiency pre-emergence herbicides continues to drive innovation in agrochemical intermediate manufacturing, specifically for compounds like Pyroxasulfone (Sulfuryl pyraflufen). A significant technological breakthrough in this domain is detailed in patent CN111393427B, which discloses a robust synthetic method achieving a total yield of 31-38%. This process addresses critical bottlenecks in traditional manufacturing by introducing a novel catalytic oxidation system and safer chlorination protocols. For R&D directors and supply chain managers, understanding this pathway is essential for securing a reliable agrochemical intermediate supplier capable of delivering consistent quality. The core innovation lies in replacing hazardous reagents with commodity chemicals while enhancing reaction selectivity, thereby setting a new standard for cost reduction in herbicide manufacturing. By leveraging sodium tungstate-catalyzed oxidation, the process eliminates difficult-to-remove byproducts, ensuring that the final active ingredient meets stringent regulatory purity requirements without extensive downstream processing.
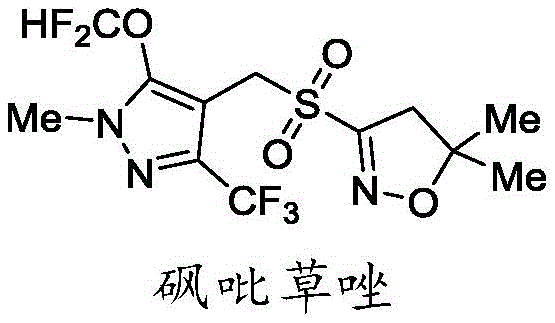
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Pyroxasulfone has been plagued by significant safety and economic inefficiencies, as illustrated in prior art such as WO2007071900a1. Traditional routes often rely on the use of isobutene gas for the construction of the dihydroisoxazole ring, presenting severe explosion hazards and requiring specialized pressure equipment that complicates commercial scale-up of complex heterocyclic intermediates. Furthermore, these legacy methods frequently employ expensive halogenating agents like N-bromosuccinimide (NBS) or N-chlorosuccinimide (NCS), which inflate raw material costs and generate stoichiometric amounts of succinimide waste. Perhaps most critically, the use of sodium methyl mercaptide introduces toxic, malodorous sulfur species that pose occupational health risks and require rigorous scrubbing systems. The reliance on m-chloroperoxybenzoic acid (mCPBA) for the final oxidation step further exacerbates cost issues due to the generation of m-chlorobenzoic acid byproducts, necessitating complex purification steps to achieve pharmaceutical-grade purity.
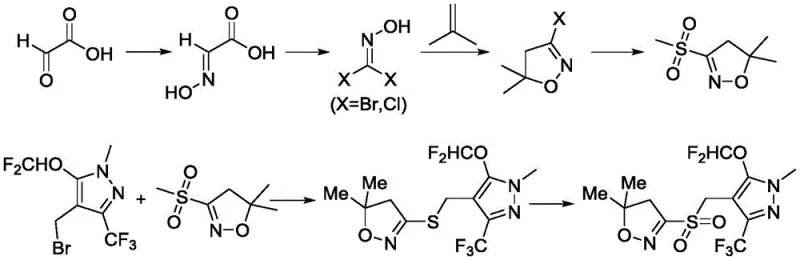
The Novel Approach
In stark contrast, the methodology outlined in CN111393427B offers a transformative solution by restructuring the synthetic logic to prioritize safety and atom economy. The new route utilizes phosphorus pentachloride (PCl5) or phosphorus oxychloride (POCl3) for the chlorination step, reagents that are not only significantly cheaper than NBS but also exhibit superior reactivity profiles that facilitate enol tautomerization. This allows for high-yield chlorination followed by simple distillation workups, effectively reducing lead time for high-purity agrochemical intermediates. The coupling strategy employs a thiourea-mediated substitution to generate the necessary sulfur linkage, avoiding the handling of volatile mercaptans. Most notably, the final oxidation step utilizes a green chemistry approach with hydrogen peroxide and a sodium tungstate catalyst. This system selectively oxidizes the thioether directly to the sulfone, bypassing the formation of sulfoxide intermediates and acidic byproducts, thus streamlining the isolation of the final white solid product with exceptional purity.
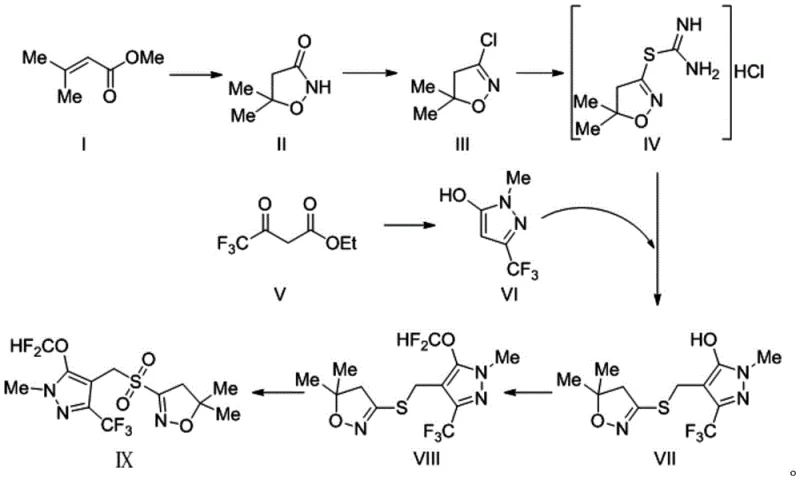
Mechanistic Insights into Sodium Tungstate-Catalyzed Oxidation
The mechanistic elegance of this synthesis is best observed in the final oxidation stage, where the conversion of intermediate VIII to the target Pyroxasulfone (IX) occurs. In this system, sodium tungstate acts as a phase-transfer-like catalyst that activates hydrogen peroxide, generating a peroxotungstate species in situ. This active oxidant possesses sufficient potential to drive the oxygenation of the sulfide bond directly to the sulfone state without over-oxidizing sensitive functional groups on the pyrazole or isoxazole rings. The presence of an acid co-catalyst, such as sulfuric acid, further stabilizes the oxidative environment, ensuring that the reaction proceeds rapidly at mild temperatures ranging from 0 to 70°C. This controlled oxidation prevents the accumulation of the sulfoxide byproduct, a common impurity in non-catalytic peroxide oxidations, thereby simplifying the impurity profile. For process chemists, this mechanism represents a significant advancement in selectivity, as it eliminates the need for chromatographic purification or recrystallization to remove m-chlorobenzoic acid, a persistent contaminant in mCPBA-based routes.
Equally important is the chlorination mechanism involving phosphorus pentachloride in the early stages of the synthesis. The interaction between PCl5 and the amide structure of intermediate II promotes rapid enolization, creating a highly reactive enol phosphate or chloride intermediate that readily accepts the chlorine atom. This pathway is kinetically favored over alternative halogenation methods, resulting in yields as high as 90% for intermediate III compared to significantly lower yields with other reagents. The subsequent reaction with thiourea proceeds through a nucleophilic attack on the chloromethyl group, forming an isothiouronium salt which is then hydrolyzed or rearranged to establish the carbon-sulfur bond required for the final scaffold. Understanding these mechanistic nuances is vital for scaling the process, as it highlights the importance of strict temperature control during the exothermic chlorination and the precise stoichiometric balance of the tungstate catalyst to maintain oxidative efficiency throughout the batch.
How to Synthesize Pyroxasulfone Efficiently
The practical implementation of this synthesis requires careful attention to reaction conditions, particularly during the chlorination and oxidation phases where exotherms and gas evolution may occur. The process begins with the cyclization of ethyl 3,3-dimethacrylate with hydroxyurea under basic conditions to form the isoxazolone ring, followed by the critical chlorination step using PCl5 in a solvent like 1,2-dichloroethane. Subsequent steps involve the coupling of the sulfur-containing fragment with the trifluoromethyl-pyrazole moiety via a Mannich-type reaction with formaldehyde, followed by difluoromethoxylation to install the ether group. The final oxidation is performed in methanol or dichloromethane, where the addition of hydrogen peroxide must be controlled to manage heat release. Detailed standardized operating procedures regarding mixing rates, temperature ramps, and quenching protocols are essential for reproducibility. For a comprehensive breakdown of the specific molar ratios, solvent choices, and workup techniques validated in the patent examples, please refer to the technical guide below.
- Cyclize compound I with hydroxyurea to form intermediate II, followed by chlorination using phosphorus pentachloride to yield intermediate III.
- React intermediate III with thiourea to obtain hydrochloride intermediate IV, while separately synthesizing pyrazole intermediate VI from ethyl trifluoroacetoacetate.
- Couple intermediates IV and VI with formaldehyde to form VII, perform difluoromethoxylation to get VIII, and finally oxidize using hydrogen peroxide and sodium tungstate to obtain Pyroxasulfone.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the adoption of this synthetic route offers substantial strategic advantages by decoupling production from volatile specialty reagent markets. The substitution of expensive halogenating agents like NBS with commodity phosphorus chlorides results in a drastic reduction in bill of materials (BOM) costs, making the final herbicide more competitive in price-sensitive agricultural markets. Moreover, the elimination of isobutene gas removes the need for specialized high-pressure reactors and associated safety audits, lowering capital expenditure (CAPEX) requirements for manufacturing facilities. The simplified workup procedures, particularly the ability to isolate the final product via filtration rather than energy-intensive distillation or crystallization sequences, translate directly into reduced utility consumption and shorter cycle times. These efficiencies collectively enhance the overall supply chain resilience, ensuring that manufacturers can maintain consistent output even during periods of raw material scarcity.
- Cost Reduction in Manufacturing: The replacement of m-chloroperoxybenzoic acid (mCPBA) with a hydrogen peroxide and sodium tungstate system eliminates the cost of the oxidant itself and the downstream costs associated with removing m-chlorobenzoic acid byproducts. Since the catalytic system allows for solvent recycling and avoids the generation of stoichiometric organic waste, the environmental disposal costs are significantly minimized. This leaner chemical footprint means that the cost per kilogram of the active ingredient is optimized, providing a clear margin advantage for generic herbicide producers looking to maximize profitability without compromising on quality standards.
- Enhanced Supply Chain Reliability: By relying on widely available bulk chemicals such as thiourea, phosphorus pentachloride, and hydrogen peroxide, the supply chain becomes less susceptible to disruptions caused by the shortage of niche intermediates. The avoidance of toxic sodium methyl mercaptide also simplifies logistics, as it removes the requirement for hazardous material transport certifications and specialized storage infrastructure. This accessibility ensures that production schedules can be maintained with greater predictability, reducing the risk of stockouts and enabling manufacturers to respond more agilely to seasonal demand spikes in the agrochemical sector.
- Scalability and Environmental Compliance: The process is inherently designed for scale, utilizing liquid-phase reactions that are easily monitored and controlled in large-scale stirred tank reactors. The absence of gaseous reagents like isobutene simplifies the engineering controls needed for containment, making technology transfer to multipurpose plants straightforward. Furthermore, the green chemistry attributes of the oxidation step align with increasingly stringent environmental regulations regarding organic waste discharge. The ability to produce high-purity material without generating persistent organic pollutants facilitates easier permitting and compliance, future-proofing the manufacturing asset against evolving regulatory landscapes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, aiming to clarify the operational benefits for potential licensees and manufacturing partners. Understanding these details is crucial for evaluating the feasibility of integrating this route into existing production lines.
Q: How does the new catalytic oxidation method improve safety compared to conventional routes?
A: The novel route eliminates the use of dangerous isobutene gas and toxic sodium methyl mercaptide found in prior art. Furthermore, it replaces hazardous oxidants like m-chloroperoxybenzoic acid (mCPBA) with a safer hydrogen peroxide and sodium tungstate system, significantly reducing industrial safety risks.
Q: What are the cost advantages of using phosphorus pentachloride for chlorination?
A: Using phosphorus pentachloride (PCl5) instead of expensive halogenating agents like NBS or NCS drastically reduces raw material costs. Additionally, PCl5 facilitates enol tautomerization, allowing for simpler post-treatment via distillation rather than complex purification, further lowering operational expenses.
Q: Can this synthesis method achieve high purity without recrystallization?
A: Yes, the optimized oxidation system utilizing sodium tungstate stabilizes hydrogen peroxide, enabling direct conversion of the thioether to the sulfone. This specificity avoids the formation of sulfoxide byproducts and m-chlorobenzoic acid, allowing the final product to reach over 99% content without the need for final recrystallization.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyroxasulfone Supplier
As the agrochemical industry evolves towards more sustainable and cost-efficient manufacturing paradigms, having a partner with deep technical expertise in heterocyclic chemistry is paramount. NINGBO INNO PHARMCHEM stands at the forefront of this transition, leveraging advanced catalytic technologies to deliver high-value intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale optimization to full-scale manufacturing is seamless. We operate stringent purity specifications and maintain rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of Pyroxasulfone meets the exacting standards required for global registration and formulation.
We invite procurement leaders and R&D directors to engage with us to explore how this optimized synthesis route can be tailored to your specific supply chain needs. By collaborating with our technical procurement team, you can request a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this greener methodology. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your herbicide portfolio remains competitive and compliant in the global market.
