Optimizing Vilazodone Intermediate Production via Novel Fischer Indole Cyclization Technology
Optimizing Vilazodone Intermediate Production via Novel Fischer Indole Cyclization Technology
The pharmaceutical industry is constantly seeking more efficient and safer pathways for the production of critical active pharmaceutical ingredients (APIs) and their precursors. A significant breakthrough in this domain is documented in patent CN102964287A, which details a novel synthesis method for 3-(4-chlorobutyl)-5-cyanoindole, a pivotal intermediate in the manufacture of Vilazodone hydrochloride. Vilazodone is a widely recognized antidepressant known for its rapid onset of action and favorable side-effect profile, making its supply chain stability crucial for global mental health treatment. The traditional methods for synthesizing this key indole derivative have long been plagued by safety concerns and operational complexities, but this new technology offers a transformative approach. By leveraging a direct Fischer indole cyclization strategy, the process achieves high purity and yield while drastically simplifying the operational workflow. This report analyzes the technical merits of this innovation, providing R&D directors and procurement leaders with a comprehensive understanding of its potential to redefine manufacturing standards for high-purity pharmaceutical intermediates.
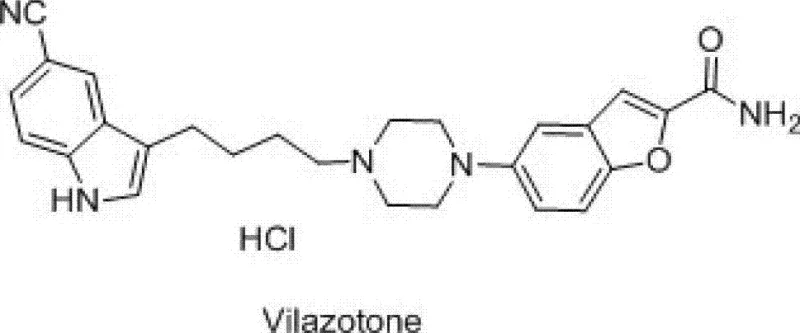
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to the introduction of the methodology described in CN102964287A, the standard industrial route for producing 3-(4-chlorobutyl)-5-cyanoindole relied heavily on a multi-step sequence involving Friedel-Crafts acylation followed by a rigorous reduction step. As illustrated in the reaction scheme below, this conventional pathway typically begins with the acylation of 5-cyanoindole using 4-chlorobutyryl chloride in the presence of anhydrous aluminum trichloride at cryogenic temperatures ranging from -5°C to 5°C. This initial step is not only energy-intensive due to the cooling requirements but also generates significant amounts of hazardous waste associated with aluminum salts. Furthermore, the subsequent reduction of the carbonyl group necessitates the use of sodium borohydride in combination with additional aluminum trichloride at temperatures between 0°C and -15°C. The reliance on sodium borohydride introduces severe safety risks, as it is highly reactive with moisture and can cause fires or explosions upon contact with water or acidic conditions. Additionally, anhydrous aluminum trichloride is extremely hygroscopic and corrosive, posing significant handling challenges and requiring specialized equipment to prevent degradation and ensure operator safety. These factors collectively contribute to higher production costs, extended lead times, and a larger environmental footprint, making the conventional route increasingly unsustainable for modern green chemistry initiatives.
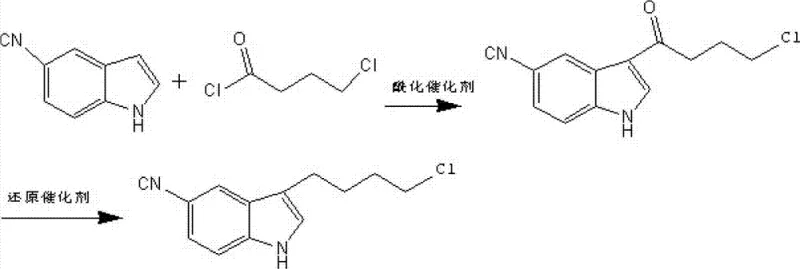
The Novel Approach
In stark contrast to the cumbersome traditional methods, the innovative process outlined in patent CN102964287A utilizes a direct Fischer indole synthesis to construct the core heterocyclic ring in a single, streamlined operation. This novel approach reacts 1,1-dimethoxy-6-chlorohexane with 4-cyanophenylhydrazine hydrochloride in a mixed solvent system comprising alcohol and water. The reaction proceeds under relatively mild thermal conditions, typically maintained between 65°C and 75°C, which eliminates the need for expensive cryogenic cooling infrastructure. The use of a homogeneous alcohol-water solvent system is particularly advantageous as it leverages the principle of like dissolves like, ensuring that both the organic alkylating agent and the inorganic hydrazine salt are fully soluble in the same phase. This homogeneity significantly enhances the collision frequency between reactant molecules, thereby driving the reaction to completion with greater efficiency and consistency. Moreover, the product, 3-(4-chlorobutyl)-5-cyanoindole, exhibits temperature-dependent solubility in ethanol, allowing for easy isolation via simple cooling and crystallization rather than complex extraction procedures. This one-pot strategy not only reduces the number of unit operations but also minimizes solvent consumption and waste generation, representing a paradigm shift towards more sustainable and cost-effective pharmaceutical manufacturing.
Mechanistic Insights into Fischer Indole Cyclization
The core of this technological advancement lies in the mechanistic elegance of the Fischer indole synthesis, which facilitates the formation of the indole ring through an acid-catalyzed rearrangement of arylhydrazones. In this specific application, the reaction initiates with the condensation of 4-cyanophenylhydrazine hydrochloride with the ketone equivalent generated in situ from 1,1-dimethoxy-6-chlorohexane. Under the acidic conditions provided by the hydrazine hydrochloride salt and the thermal energy supplied at 65°C to 75°C, the acetal functionality of the starting material is hydrolyzed to reveal the reactive carbonyl group. This carbonyl species immediately undergoes nucleophilic attack by the hydrazine nitrogen to form a hydrazone intermediate. Subsequent protonation of the hydrazone nitrogen triggers a critical [3,3]-sigmatropic rearrangement, often referred to as the Fischer rearrangement, which establishes the carbon-carbon bond necessary for ring closure. Following the rearrangement, the loss of ammonia and subsequent aromatization yield the final indole structure. The presence of the cyano group on the phenyl ring is well-tolerated under these conditions, demonstrating the robustness of the method for functionalized substrates. This mechanism avoids the harsh Lewis acid conditions required in Friedel-Crafts chemistry, thereby preserving the integrity of sensitive functional groups and minimizing the formation of polymeric by-products that often complicate purification in traditional routes.
From an impurity control perspective, the choice of solvent and reaction parameters plays a pivotal role in ensuring the high purity of the final product. The patent data indicates that controlling the dropwise addition rate of the hydrazine solution is critical; adding the reagent too rapidly can lead to localized exotherms and uncontrolled reaction kinetics, potentially generating side products such as regioisomers or oligomers. Conversely, adding the reagent too slowly may prolong the exposure of intermediates to thermal stress, also risking degradation. By optimizing the addition rate to approximately 0.5 to 1 mL/min and maintaining a strict temperature window of 70°C to 75°C, the process maximizes the selectivity for the desired 3-substituted indole. Furthermore, the recrystallization step utilizing a 50% to 60% ethanol-water mixture exploits the differential solubility of the product versus impurities. As the solution cools from the reaction temperature to room temperature, the target molecule precipitates out with high fidelity, leaving soluble impurities in the mother liquor. This inherent purification capability embedded within the workup procedure contributes to the reported purity levels exceeding 99%, which is essential for meeting the stringent quality specifications required for pharmaceutical intermediates destined for human consumption.
How to Synthesize 3-(4-Chlorobutyl)-5-Cyanoindole Efficiently
Implementing this synthesis route requires careful attention to solvent ratios and thermal management to replicate the high yields reported in the patent literature. The process begins with the dissolution of the alkylating agent in a pre-heated alcohol-water mixture, ensuring a clear homogeneous phase before the introduction of the hydrazine component. The subsequent addition must be controlled precisely to maintain the reaction exotherm within safe limits while promoting the cyclization kinetics. Detailed operational parameters, including specific molar ratios and stirring speeds, are critical for scaling this reaction from laboratory benchtop to pilot plant volumes. For a comprehensive guide on the standardized execution of this protocol, please refer to the technical instructions provided below.
- Dissolve 1,1-dimethoxy-6-chlorohexane in a heated mixture of alcohol and water under inert gas protection to ensure complete solubility.
- Slowly add a solution of 4-cyanophenylhydrazine hydrochloride in alcohol and water to the reaction mixture while maintaining a temperature between 65°C and 75°C.
- Maintain the reaction temperature for 0.5 to 2 hours, then cool to room temperature to precipitate the product, followed by suction filtration and recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis method presents a compelling value proposition centered on risk mitigation and operational efficiency. The elimination of hazardous reagents such as sodium borohydride and anhydrous aluminum trichloride fundamentally alters the safety profile of the manufacturing facility. Without the need to store and handle pyrophoric or highly corrosive materials, the regulatory burden associated with hazardous waste disposal and worker safety protocols is substantially reduced. This translates directly into lower compliance costs and reduced insurance premiums, contributing to a more lean and agile supply chain. Furthermore, the simplified one-pot nature of the reaction reduces the overall cycle time by removing intermediate isolation and purification steps that are characteristic of the older acylation-reduction sequence. Fewer processing steps mean less equipment occupancy time, lower labor requirements, and decreased utility consumption, all of which drive down the cost of goods sold (COGS) without compromising on quality. The use of benign solvents like ethanol and water also simplifies wastewater treatment processes, aligning production with increasingly strict environmental regulations and enhancing the sustainability credentials of the final product.
- Cost Reduction in Manufacturing: The economic benefits of this process are derived primarily from the simplification of the synthetic route and the substitution of expensive, dangerous reagents with commodity chemicals. By removing the requirement for cryogenic cooling and specialized corrosion-resistant reactors needed for aluminum chloride handling, capital expenditure (CAPEX) for new production lines can be significantly optimized. Additionally, the high atom economy of the Fischer indole synthesis ensures that a greater proportion of raw materials are converted into the final product, minimizing waste and maximizing yield. The avoidance of complex workup procedures, such as multiple extractions and hazardous quenching steps, further reduces the consumption of auxiliary solvents and processing time. These cumulative efficiencies result in a robust cost structure that allows for competitive pricing in the global market for pharmaceutical intermediates, providing a distinct advantage in tender negotiations and long-term supply contracts.
- Enhanced Supply Chain Reliability: Supply chain resilience is critically dependent on the availability and stability of raw materials. The new method relies on 1,1-dimethoxy-6-chlorohexane and 4-cyanophenylhydrazine hydrochloride, which are commercially available and stable compounds, unlike the moisture-sensitive reagents used in the conventional route. This stability reduces the risk of supply disruptions caused by transportation restrictions on hazardous goods or storage degradation. Moreover, the robustness of the reaction conditions—operating at moderate temperatures with tolerant solvent systems—means that the process is less susceptible to minor fluctuations in utility supplies or environmental conditions. This reliability ensures consistent batch-to-batch quality and on-time delivery performance, which are key metrics for maintaining trust with downstream API manufacturers. The ability to scale this process seamlessly from kilogram to tonne quantities without significant re-engineering further secures the supply chain against demand volatility.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces unforeseen challenges related to heat transfer and mixing efficiency, but the homogeneous nature of this alcohol-water system mitigates many of these risks. The exothermicity of the reaction is manageable through controlled addition rates, making it suitable for large-scale batch reactors without the need for complex continuous flow setups initially. From an environmental standpoint, the replacement of chlorinated solvents and heavy metal catalysts with aqueous ethanol solutions dramatically reduces the toxicity of the effluent stream. This facilitates easier treatment and potential recycling of solvents, supporting a circular economy approach within the manufacturing plant. Compliance with green chemistry principles not only future-proofs the operation against tightening environmental legislation but also enhances the brand reputation of the supplier as a responsible partner in the pharmaceutical value chain, appealing to eco-conscious stakeholders and investors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and claims presented in patent CN102964287A, offering clarity on the practical aspects of adopting this method for industrial production. Understanding these nuances is essential for technical teams evaluating the feasibility of technology transfer and for commercial teams assessing the strategic fit within their existing portfolio.
Q: What are the primary safety advantages of this new synthesis method compared to conventional routes?
A: The novel method eliminates the use of hazardous reducing agents like sodium borohydride and corrosive catalysts such as anhydrous aluminum trichloride, significantly reducing explosion risks and handling hazards.
Q: How does the solvent system in patent CN102964287A improve reaction efficiency?
A: By utilizing a miscible alcohol-water co-solvent system, the process ensures that both organic and inorganic reactants remain in a single homogeneous phase, thereby enhancing reaction kinetics and simplifying downstream processing.
Q: Is this synthetic route suitable for large-scale industrial production?
A: Yes, the one-pot nature of the reaction, combined with mild operating temperatures and the absence of dangerous reagents, makes it highly scalable and compliant with strict environmental and safety regulations for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-(4-Chlorobutyl)-5-Cyanoindole Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced synthetic methodologies requires a partner with deep technical expertise and proven manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patent CN102964287A are fully realized in a commercial setting. We operate state-of-the-art facilities equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of 3-(4-chlorobutyl)-5-cyanoindole meets the highest international standards. Our commitment to quality assurance extends beyond simple compliance; we actively engage in process optimization to enhance yield and reduce impurity profiles, delivering a superior product that supports the efficacy and safety of the final antidepressant medication.
We invite pharmaceutical companies and contract manufacturing organizations to collaborate with us to leverage this cutting-edge synthesis technology. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical needs. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you secure a sustainable, cost-effective, and reliable supply of this critical intermediate, empowering your organization to lead in the competitive landscape of mental health therapeutics.
