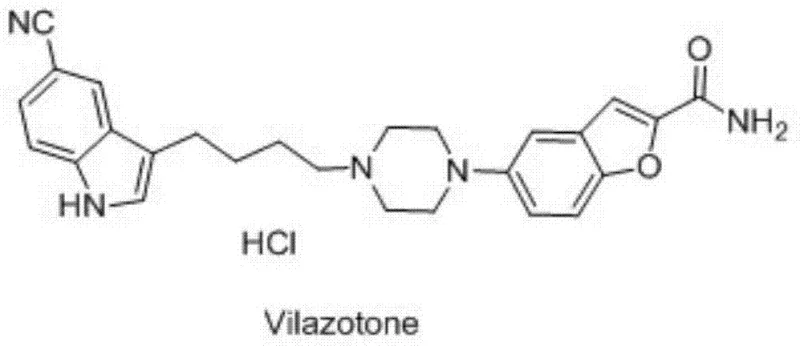
Advanced Fischer Indole Synthesis for High-Purity Vilazodone Intermediates and Commercial Scalability
Advanced Fischer Indole Synthesis for High-Purity Vilazodone Intermediates and Commercial Scalability
The pharmaceutical landscape for antidepressant therapies continues to evolve, with Vilazodone hydrochloride standing out as a potent serotonin partial agonist and reuptake inhibitor. As detailed in patent CN102964287B, the efficient production of its key precursor, 3-(4-chlorobutyl)-5-cyanoindole, represents a critical bottleneck that this new technology successfully addresses. This patent introduces a groundbreaking synthetic methodology that shifts away from hazardous traditional reduction protocols toward a greener, more robust Fischer indole cyclization strategy. For R&D directors and procurement specialists, understanding this shift is vital, as it directly impacts the safety profile, cost structure, and scalability of the final Active Pharmaceutical Ingredient (API). The structural complexity of Vilazodone, featuring a benzofuran moiety linked to a substituted indole via a piperazine bridge, demands intermediates of exceptional purity to meet stringent regulatory standards.
The innovation lies not merely in the chemical transformation but in the holistic re-engineering of the process flow to eliminate high-risk unit operations. By leveraging a one-pot reaction system utilizing benign alcohol-water binary solvents, the inventors have achieved a synthesis route that is inherently safer and more environmentally compliant. This approach mitigates the severe safety hazards associated with moisture-sensitive catalysts and pyrophoric reducing agents found in legacy methods. Furthermore, the process demonstrates remarkable operational simplicity, allowing for tighter control over impurity profiles, which is a paramount concern for quality assurance teams managing global supply chains. The ability to produce this intermediate with yields exceeding 78% and purity levels approaching 99.5% underscores the commercial viability of this patented technique for large-scale manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3-(4-chlorobutyl)-5-cyanoindole has relied on a two-step sequence involving Friedel-Crafts acylation followed by a harsh reduction step, a pathway fraught with significant operational and safety challenges. As illustrated in the prior art reaction schemes, the initial acylation of 5-cyanoindole with 4-chlorobutyryl chloride requires anhydrous aluminum trichloride, a highly corrosive Lewis acid that generates substantial acidic waste streams upon quenching. More critically, the subsequent reduction of the carbonyl group traditionally employs a mixture of sodium borohydride and anhydrous aluminum trichloride at cryogenic temperatures ranging from 0°C to -15°C. This combination presents extreme safety liabilities; sodium borohydride is prone to violent decomposition upon contact with moisture or acids, releasing flammable hydrogen gas, while the exothermic nature of the reaction requires energy-intensive cooling infrastructure to prevent thermal runaway.
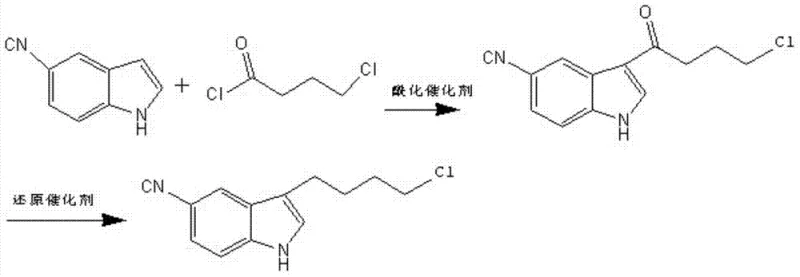
Beyond the immediate safety risks, the conventional method suffers from inherent inefficiencies that degrade the economic feasibility of the process. The requirement for strictly anhydrous conditions and low-temperature control significantly increases capital expenditure (CAPEX) and operating expenditure (OPEX) due to the need for specialized reactors and refrigeration systems. Moreover, the use of chlorinated organic solvents such as dichloromethane or carbon tetrachloride, often mandated by the solubility requirements of the aluminum complexes, creates severe environmental compliance burdens regarding volatile organic compound (VOC) emissions and waste disposal. The cumulative effect of these factors is a process with lower overall yields, higher impurity loads requiring extensive purification, and a supply chain vulnerable to disruptions caused by the strict regulation of hazardous precursors.
The Novel Approach
In stark contrast to the perilous legacy routes, the methodology disclosed in patent CN102964287B utilizes a direct Fischer indole synthesis, fundamentally simplifying the molecular construction of the target intermediate. This novel approach condenses 4-cyanophenylhydrazine hydrochloride with 1,1-dimethoxy-6-chlorohexane in a single pot, effectively bypassing the need for separate acylation and reduction steps. The reaction proceeds smoothly in a mixed solvent system of alcohol (ethanol or methanol) and water, eliminating the necessity for toxic chlorinated solvents and moisture-sensitive reagents. The visual representation of this streamlined pathway highlights the elegance of the design, where the carbon chain is introduced pre-functionalized, and the indole ring is constructed de novo around it, ensuring high regioselectivity and minimal by-product formation.
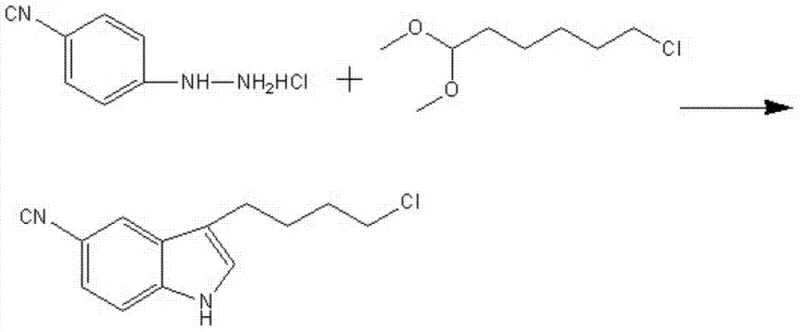
The operational advantages of this new route are profound, offering a dramatic reduction in process complexity and hazard potential. By conducting the reaction at moderate temperatures between 65°C and 75°C, the process eliminates the need for energy-intensive cryogenic cooling, thereby reducing the facility's carbon footprint and utility costs. The use of water as a co-solvent not only enhances the solubility of the hydrazine salt but also facilitates the precipitation of the product upon cooling, simplifying the isolation procedure to a mere filtration and recrystallization. This "green chemistry" alignment not only satisfies increasingly rigorous environmental regulations but also translates directly into cost reduction in pharmaceutical intermediate manufacturing by minimizing waste treatment expenses and maximizing throughput efficiency.
Mechanistic Insights into Fischer Indole Cyclization
The core of this technological breakthrough is the Fischer indole synthesis, a classic organic transformation that has been ingeniously adapted here for industrial robustness. The mechanism initiates with the acid-catalyzed condensation of the hydrazine moiety of 4-cyanophenylhydrazine with the acetal group of 1,1-dimethoxy-6-chlorohexane, forming a hydrazone intermediate in situ. Under the thermal conditions provided (70-75°C), this hydrazone undergoes a concerted [3,3]-sigmatropic rearrangement, a pericyclic reaction that establishes the new carbon-carbon bond essential for the indole framework. Following the rearrangement, the loss of ammonia and subsequent aromatization yield the final 3-substituted indole structure. The presence of the electron-withdrawing cyano group on the phenyl ring is carefully managed by the solvent system to ensure it does not interfere with the nucleophilicity of the hydrazine nitrogen, a delicate balance achieved through the optimized alcohol-water ratio.
From an impurity control perspective, the choice of 1,1-dimethoxy-6-chlorohexane as the alkylating agent is strategic. Unlike the acyl chloride used in older methods, the acetal functionality is less prone to uncontrolled polymerization or side reactions with the solvent. The patent data indicates that maintaining the dropwise addition rate between 0.5 and 1 mL/min is critical; this kinetic control prevents the local accumulation of reactants that could lead to oligomerization or the formation of bis-indole by-products. Furthermore, the solubility characteristics of the product in ethanol are exploited for purification; since the target molecule is significantly less soluble in cold ethanol-water mixtures than at elevated temperatures, a simple temperature swing crystallization effectively rejects soluble impurities, delivering the high purity (>99%) required for downstream coupling with the benzofuran-piperazine fragment.
How to Synthesize 3-(4-Chlorobutyl)-5-cyanoindole Efficiently
The practical execution of this synthesis relies on precise adherence to the solvent ratios and thermal profiles outlined in the patent examples to ensure reproducibility at scale. The process begins with the dissolution of the acetal precursor in the alcohol-water mixture under an inert atmosphere to prevent oxidation, followed by the controlled introduction of the hydrazine salt solution. Detailed standard operating procedures regarding agitation speeds, heat transfer coefficients, and filtration parameters are essential for translating this laboratory success to multi-ton production.
- Dissolve 1,1-dimethoxy-6-chlorohexane in a mixture of ethanol and water under inert gas protection, heating to 65-75°C to ensure complete solubility.
- Prepare a solution of 4-cyanophenylhydrazine hydrochloride in ethanol and water, and slowly add this mixture to the heated reaction vessel.
- Maintain the reaction temperature at 70-75°C for 0.5 to 2 hours to facilitate ring closure, then cool to room temperature to precipitate the product for filtration and recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this Fischer indole route offers compelling strategic benefits that extend far beyond simple yield improvements. The elimination of sodium borohydride and anhydrous aluminum trichloride removes two major supply chain vulnerabilities; these reagents are often subject to strict transportation regulations and storage limitations due to their hazardous classification. By replacing them with commodity chemicals like ethanol, water, and commercially available hydrazine salts, the manufacturing process becomes significantly more resilient to logistical disruptions and regulatory changes. This simplification of the Bill of Materials (BOM) allows for more accurate forecasting and reduces the administrative burden associated with handling dangerous goods, thereby streamlining the entire procurement lifecycle.
- Cost Reduction in Manufacturing: The economic impact of this process innovation is driven primarily by the removal of expensive and hazardous reagents and the simplification of the reaction workflow. Eliminating the cryogenic reduction step drastically reduces energy consumption, as the facility no longer needs to maintain sub-zero temperatures for extended periods, leading to substantial utility savings. Additionally, the avoidance of chlorinated solvents lowers waste disposal costs, which are typically high for halogenated organic waste streams. The one-pot nature of the reaction also reduces labor hours and reactor occupancy time, increasing the overall asset utilization rate of the production plant and driving down the unit cost of the intermediate.
- Enhanced Supply Chain Reliability: The reliance on stable, non-hazardous raw materials significantly de-risks the supply chain. Precursors like cycloheptene (used to make the acetal) and phenylhydrazines are widely produced commodities with robust global supply networks, unlike specialized reducing agents that may have limited suppliers. This diversification of sourcing options ensures continuity of supply even during market fluctuations. Furthermore, the milder reaction conditions reduce the wear and tear on processing equipment, minimizing unplanned maintenance downtime and ensuring consistent delivery schedules to downstream API manufacturers who depend on just-in-time inventory models.
- Scalability and Environmental Compliance: Scaling this process from pilot to commercial production is inherently safer and more straightforward due to the absence of exothermic hazards associated with hydride reductions. The use of water as a primary solvent component aligns perfectly with modern sustainability goals and ESG (Environmental, Social, and Governance) mandates, making the final product more attractive to environmentally conscious pharmaceutical partners. The simplified workup procedure, which avoids complex extractions with halogenated solvents, facilitates easier scale-up and reduces the physical footprint required for solvent recovery systems, enabling faster time-to-market for new generic or branded formulations of Vilazodone.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route, derived directly from the experimental data and claims within the patent documentation. Understanding these nuances is crucial for technical teams evaluating the feasibility of adopting this method for their own production lines or for procurement officers negotiating supply contracts.
Q: Why is the Fischer Indole method superior to the traditional Friedel-Crafts acylation route for this intermediate?
A: The traditional route relies on hazardous reagents like sodium borohydride and anhydrous aluminum trichloride, which pose significant safety risks including explosion and corrosion. The Fischer Indole method described in patent CN102964287B utilizes mild alcohol-water solvents and avoids dangerous reducing agents, resulting in a safer process with higher yields and easier purification.
Q: What are the critical process parameters for ensuring high purity in this synthesis?
A: Critical parameters include maintaining the reaction temperature strictly between 65°C and 75°C and controlling the dropwise addition rate of the hydrazine salt to between 0.5 and 1 mL/min. Deviations can lead to incomplete reactions or the formation of by-products, whereas precise control ensures the formation of high-purity 3-(4-chlorobutyl)-5-cyanoindole suitable for pharmaceutical applications.
Q: Is the starting material 1,1-dimethoxy-6-chlorohexane commercially viable for large-scale production?
A: Yes, the starting material can be efficiently synthesized from cycloheptene via ozonolysis followed by treatment with ferric chloride, or it can be sourced commercially. This flexibility in raw material sourcing enhances supply chain reliability and allows manufacturers to optimize costs based on market availability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-(4-Chlorobutyl)-5-cyanoindole Supplier
At NINGBO INNO PHARMCHEM, we recognize that the synthesis of complex pharmaceutical intermediates like 3-(4-chlorobutyl)-5-cyanoindole requires not just chemical expertise but a deep commitment to quality and scalability. Our technical team has extensively analyzed the pathways described in CN102964287B and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We understand that transitioning from a lab-scale Fischer indole reaction to a multi-ton industrial process involves critical engineering challenges, from heat management during the exothermic cyclization to the optimization of crystallization kinetics for maximum purity. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, ensuring that every batch meets the exacting standards required for antidepressant API synthesis.
We invite global partners to leverage our manufacturing capabilities to secure a stable and cost-effective supply of this vital intermediate. By collaborating with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how our optimized process can improve your bottom line. We encourage you to contact our technical procurement team today to request specific COA data from our recent pilot runs and to discuss route feasibility assessments for your long-term supply needs. Let us be your partner in bringing safer, more affordable mental health treatments to the global market.
