Revolutionizing Amide Bond Formation: A Technical Analysis of NHC-Catalyzed Ester Aminolysis for Commercial Scale-Up
Revolutionizing Amide Bond Formation: A Technical Analysis of NHC-Catalyzed Ester Aminolysis for Commercial Scale-Up
The landscape of amide bond construction, a cornerstone reaction in the synthesis of active pharmaceutical ingredients (APIs) and fine chemicals, is undergoing a significant paradigm shift driven by the need for greener and more efficient methodologies. Patent CN108558692B, published in late 2020, introduces a groundbreaking preparation method for amide compounds that leverages the unique reactivity of N-heterocyclic carbenes (NHCs) to facilitate the direct aminolysis of organic acid esters. This technology represents a critical advancement for the industry, moving away from the reliance on stoichiometric activating agents and transition metal catalysts towards a more sustainable organocatalytic approach. For R&D directors and process chemists, this patent offers a compelling alternative that promises to streamline synthetic routes, reduce impurity profiles, and enhance overall process safety. The core innovation lies in the ability of the NHC catalyst to activate the relatively inert ester carbonyl group, rendering it sufficiently electrophilic to react with amines under remarkably mild conditions, thereby addressing long-standing challenges in amidation chemistry.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of amides has been dominated by two primary strategies, both of which carry significant operational burdens and environmental liabilities that impact the total cost of ownership for chemical manufacturers. The first conventional pathway involves the activation of carboxylic acids using coupling reagents such as phosphines and iodine (Ph3P/I2) or uronium salts like HATU, followed by reaction with amines. While effective, this approach is plagued by the generation of substantial stoichiometric waste, difficult-to-remove phosphine oxide byproducts, and the high cost of the activating agents themselves. Furthermore, these reactions often require prolonged reaction times and strict anhydrous conditions to prevent hydrolysis of the activated intermediate. The second traditional method relies on the oxidative aminolysis of aldehydes, typically mediated by transition metals. Although this avoids pre-activation, it suffers from narrow substrate scope, potential heavy metal contamination which is unacceptable in pharmaceutical grades, and the inherent instability of aldehyde starting materials. These legacy methods create bottlenecks in supply chains due to complex purification requirements and regulatory hurdles associated with residual metal limits.
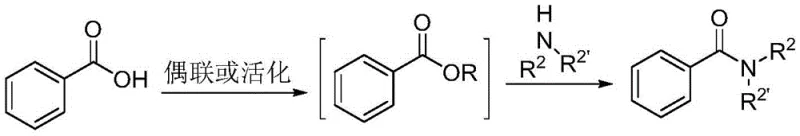
The Novel Approach
In stark contrast to these established but flawed methodologies, the novel approach detailed in the patent utilizes a catalytic amount of an N-heterocyclic carbene to drive the reaction between readily available organic acid esters and amines. This strategy fundamentally alters the economic and technical equation by replacing expensive, waste-generating activators with a reusable organic catalyst. The reaction proceeds efficiently at temperatures ranging from 0°C to 40°C, often reaching completion within just 0.1 to 1.5 hours, which drastically reduces energy consumption compared to thermal activation methods. By employing esters as the acyl source, the process benefits from the stability and low cost of these feedstocks, while the NHC catalyst ensures high conversion rates without the need for harsh conditions. This shift not only simplifies the operational workflow but also significantly enhances the purity of the final product by eliminating metal residues and complex coupling byproducts, making it an ideal candidate for the production of high-purity pharmaceutical intermediates.
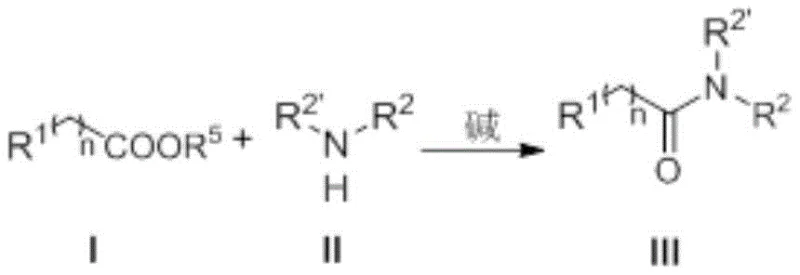
Mechanistic Insights into NHC-Catalyzed Ester Aminolysis
The efficacy of this transformation is rooted in the unique electronic properties of the N-heterocyclic carbene catalyst, which acts as a potent nucleophilic activator. Mechanistically, the carbene species, generated in situ from its precursor salt upon treatment with a base such as DBU or potassium carbonate, attacks the carbonyl carbon of the ester substrate. This nucleophilic addition forms a highly reactive tetrahedral intermediate or an acyl azolium species, effectively increasing the electrophilicity of the carbonyl center far beyond that of the parent ester. This activation lowers the energy barrier for the subsequent nucleophilic attack by the amine, facilitating the displacement of the alkoxide leaving group. The catalyst is then regenerated, allowing the cycle to continue with high turnover numbers. This mechanism bypasses the need for external activating groups, relying instead on the intrinsic reactivity of the carbene-ester adduct to drive the formation of the amide bond with exceptional selectivity and speed.
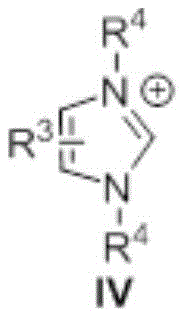
From an impurity control perspective, this mechanistic pathway offers distinct advantages for process chemistry teams tasked with defining critical quality attributes (CQAs). Because the reaction does not involve radical intermediates or redox-active metal centers, the formation of side products such as over-oxidized species or metal-complexed impurities is virtually eliminated. The primary byproduct is the corresponding alcohol derived from the ester leaving group, which is typically volatile or easily separated during the aqueous workup and extraction phases described in the patent examples. The use of mild bases like DBU or inorganic carbonates further minimizes the risk of base-sensitive functional group degradation, ensuring that complex molecular architectures containing sensitive moieties remain intact. This clean reaction profile translates directly to simplified downstream processing, reducing the burden on purification units and increasing the overall yield of the desired high-purity amide compound.
How to Synthesize Amide Compounds Efficiently
The practical implementation of this NHC-catalyzed protocol is designed for ease of execution in both laboratory and pilot plant settings, requiring standard equipment and commonly available reagents. The procedure begins with the preparation of the catalytic system under an inert atmosphere to prevent carbene deactivation by moisture or oxygen, followed by the sequential addition of substrates to manage exotherms and ensure optimal mixing. The robustness of the method allows for flexibility in solvent choice, with tetrahydrofuran being preferred for its solubilizing properties, though other polar aprotic solvents are compatible. Following the rapid reaction period, the workup involves a straightforward aqueous quench and organic extraction, avoiding the need for specialized scavengers or filtration steps often required in metal-catalyzed processes. For detailed operational parameters and specific stoichiometric ratios optimized for different substrate classes, please refer to the standardized synthesis guide below.
- Under an inert atmosphere, dissolve the N-heterocyclic carbene precursor and a suitable organic base in a dry organic solvent such as tetrahydrofuran.
- Sequentially add the organic acid ester substrate and the organic amine nucleophile to the reaction mixture while maintaining controlled temperatures between 0°C and 40°C.
- Upon completion, quench the reaction with water, extract the product using an organic solvent like dichloromethane, and purify the crude residue via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this NHC-catalyzed amidation technology presents a compelling value proposition centered on cost efficiency, supply security, and regulatory compliance. The shift from stoichiometric coupling reagents to a catalytic organic system fundamentally changes the cost structure of amide manufacturing by removing the dependency on high-priced activators like HATU or EDC, which are subject to volatile market pricing and supply constraints. Furthermore, the elimination of transition metals removes the necessity for expensive metal scavenging resins and the associated validation testing for residual metals, which is a significant hidden cost in API production. The mild reaction conditions also imply lower energy costs for heating or cooling, contributing to a reduced carbon footprint and aligning with corporate sustainability goals. These factors combine to create a more resilient and cost-effective supply chain for critical amide intermediates.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the replacement of expensive stoichiometric reagents with a low-loading organic catalyst and the use of cheap, stable ester starting materials. By avoiding the purchase of premium coupling agents and the disposal costs associated with their phosphine or urea byproducts, manufacturers can achieve substantial savings in raw material expenditures. Additionally, the simplified workup procedure reduces solvent consumption and labor hours required for purification, further driving down the cost of goods sold (COGS) without compromising on product quality or yield.
- Enhanced Supply Chain Reliability: Reliance on transition metal catalysts often introduces supply chain vulnerabilities due to the geopolitical concentration of metal mining and refining. This organocatalytic method utilizes nitrogen-based heterocycles which are synthesized from abundant commodity chemicals, ensuring a stable and diversified supply base. The robustness of the reaction also means that production schedules are less likely to be disrupted by batch failures or the need for re-processing due to metal contamination, thereby improving on-time delivery performance for downstream customers relying on these key pharmaceutical intermediates.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies safety and environmental risks, but this methodology mitigates those concerns through its inherent safety profile. The absence of pyrophoric reagents or heavy metals simplifies the handling of large volumes and reduces the regulatory burden associated with hazardous waste disposal. The high atom economy and minimal waste generation make this process highly attractive for meeting increasingly stringent environmental regulations, facilitating smoother permitting for commercial scale-up and ensuring long-term operational continuity in regulated markets.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented amidation technology, providing clarity on its applicability and benefits for industrial partners. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation, ensuring accuracy and relevance for decision-makers evaluating this route for their own manufacturing portfolios. Understanding these details is crucial for assessing the feasibility of integrating this method into existing production lines.
Q: What are the primary advantages of NHC-catalyzed amidation over traditional coupling reagents?
A: Unlike traditional methods requiring expensive coupling reagents like HATU or harsh acid chloride activation, this NHC-catalyzed method operates under mild conditions with high atom economy, eliminating toxic byproducts and simplifying downstream purification.
Q: Is this method suitable for large-scale pharmaceutical manufacturing?
A: Yes, the process utilizes inexpensive esters and amines, avoids transition metals, and proceeds rapidly at room temperature, making it highly scalable and compliant with stringent environmental regulations for commercial production.
Q: What is the typical substrate scope for this catalytic system?
A: The system demonstrates broad adaptability, successfully converting various substituted benzoates (including electron-deficient nitro-derivatives) and diverse amines into high-purity amide products with yields exceeding 90%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Amide Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the NHC-mediated amidation described in CN108558692B to redefine the standards of efficiency and purity in fine chemical synthesis. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory discoveries are seamlessly translated into robust industrial realities. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of amide intermediate we deliver meets the exacting demands of the global pharmaceutical industry. We are committed to leveraging such cutting-edge chemistry to provide our clients with superior products that accelerate their drug development timelines.
We invite you to collaborate with us to explore how this efficient synthesis route can be tailored to your specific project needs, offering a pathway to significant operational improvements. Please contact our technical procurement team to request a Customized Cost-Saving Analysis for your target molecules, along with specific COA data and route feasibility assessments. By partnering with NINGBO INNO PHARMCHEM, you gain access not just to a supplier, but to a strategic ally dedicated to optimizing your supply chain through scientific excellence and commercial integrity.
