Revolutionizing DHEA Production: A Direct Microbial Fermentation Route from Plant Sterols
The pharmaceutical industry continuously seeks more efficient and sustainable pathways for producing critical hormonal intermediates, and the technology disclosed in patent CN102816825A represents a significant leap forward in the manufacturing of Dehydroepiandrosterone (DHEA). Traditionally, DHEA production has relied heavily on complex chemical synthesis starting from expensive precursors or multi-step bioconversion processes that first generate 4-androstene-3,17-dione (4-AD) before further chemical modification. This new method disrupts that status quo by enabling the direct preparation of DHEA from readily available plant sterols through a specialized microbial fermentation process. By integrating a strategic hydroxyl protection step prior to fermentation, the process achieves high specificity and yield, effectively bypassing the need for the intermediate 4-AD stage. This innovation not only simplifies the production workflow but also aligns with modern green chemistry principles by reducing the reliance on harsh organic solvents and minimizing waste generation.
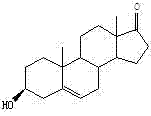
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of DHEA has been plagued by high operational costs and environmental burdens associated with traditional chemical methodologies. Conventional routes often utilize raw materials such as acetic acid gestation diene alcohol ketone, which command high market prices and are subject to volatile supply chain fluctuations. Furthermore, these chemical processes typically involve three or more distinct reaction steps, each requiring rigorous purification and the use of substantial quantities of organic solvents. This multi-step nature inherently leads to cumulative yield losses, where the efficiency of the final product is compromised by the inefficiencies of each preceding stage. Additionally, the heavy reliance on organic solvents creates significant environmental pressure regarding waste disposal and solvent recovery, increasing the overall carbon footprint of the manufacturing process. The complexity of these routes also poses challenges for scale-up, as maintaining consistent quality and purity across multiple chemical transformations requires sophisticated process control and expensive equipment.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a streamlined bio-engineering strategy that leverages the natural catalytic power of specific Mycobacterium strains to transform low-cost plant sterols directly into DHEA. The core innovation lies in the pre-fermentation protection of the 3-hydroxyl group on the sterol molecule, which fundamentally alters the substrate's interaction with the microorganism. By masking this reactive site, the process forces the bacterial enzymes to focus exclusively on cleaving the side chain at the C17 position, thereby directly generating the desired 17-ketone structure without forming the 4-AD intermediate. This biological shortcut drastically reduces the number of processing steps, eliminating the need for separate chemical oxidation or rearrangement reactions that are typical in older methods. The use of plant sterols, which are abundant byproducts of the vegetable oil industry, provides a robust and economically advantageous feedstock that decouples production costs from the volatility of synthetic chemical markets. Consequently, this method offers a pathway to significantly lower production costs while simultaneously improving the overall mass yield and environmental profile of DHEA manufacturing.
Mechanistic Insights into Hydroxyl-Protection Assisted Microbial Transformation
The success of this fermentation process hinges on a sophisticated understanding of substrate-enzyme interactions and the strategic manipulation of molecular polarity. In standard microbial transformations of plant sterols, Mycobacterium species typically degrade the side chain to produce 4-AD or ADD (Androst-1,4-diene-3,17-dione), often oxidizing the 3-hydroxyl group in the process. To circumvent this, the patent introduces a protection step using methylal (dimethoxymethane) in the presence of a vanadium pentoxide catalyst. This reaction converts the polar 3-hydroxyl group into a less polar ether derivative, which dramatically increases the substrate's solubility in the vegetable oil-based fermentation medium. Enhanced solubility is critical because it improves the mass transfer of the sterol into the bacterial cells, thereby accelerating the conversion rate and boosting overall efficiency. The protection group acts as a steric and electronic shield, preventing the microbial enzymes from attacking the A-ring of the steroid nucleus and ensuring that the biocatalytic activity is directed solely toward the oxidative cleavage of the aliphatic side chain.
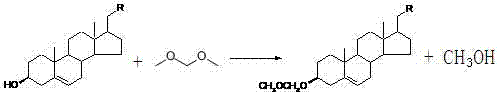
Following the fermentation, the process employs a mild acid hydrolysis step to remove the protecting group, regenerating the free 3-hydroxyl functionality essential for DHEA's biological activity. This deprotection is highly selective and occurs under conditions that do not degrade the sensitive steroid backbone. The entire sequence—from protection to fermentation to hydrolysis—is designed to minimize the formation of impurities such as residual plant sterols or over-oxidized byproducts like 4-AD. By controlling the pH of the fermentation broth between 6.8 and 7.2 using phosphate buffers, the process further stabilizes the protecting group against premature hydrolysis during the biological phase. This precise control over reaction conditions ensures that the final crude product contains a high concentration of the target molecule, simplifying the downstream purification requirements. The result is a highly efficient catalytic cycle that maximizes atom economy and minimizes the generation of hazardous waste streams.
How to Synthesize Dehydroepiandrosterone Efficiently
The synthesis of DHEA via this patented method involves a logical sequence of four primary operations that can be seamlessly integrated into existing fermentation infrastructure. The process begins with the chemical protection of the sterol feedstock, followed by the biological transformation in a controlled bioreactor environment, and concludes with chemical hydrolysis and crystallization. Each step is optimized to ensure maximum throughput and minimal resource consumption, making it an ideal candidate for large-scale commercial production. The detailed standardized synthesis steps are outlined below to guide technical implementation.
- Protect the 3-hydroxyl group of plant sterols using methylal and a vanadium pentoxide catalyst to enhance solubility and prevent side reactions.
- Ferment the protected sterol using Mycobacterium sp. NRRL B-3683 or 3805 in a vegetable oil-based medium to cleave the side chain.
- Hydrolyze the fermented product using dilute acid to remove the protecting group, followed by recrystallization to achieve high purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this microbial fermentation technology presents a compelling value proposition centered on cost stability and operational resilience. The shift from expensive, synthetically derived raw materials to plant sterols fundamentally alters the cost structure of DHEA production. Plant sterols are widely available as byproducts of soybean and tall oil processing, creating a supply base that is both vast and geographically diverse. This abundance mitigates the risk of raw material shortages and price spikes that often plague the fine chemical sector. Furthermore, the reduction in the number of synthesis steps translates directly into lower operational expenditures, as fewer unit operations mean reduced energy consumption, lower labor requirements, and decreased capital investment in reactor capacity. The elimination of transition metal catalysts and harsh organic solvents also simplifies waste management protocols, leading to substantial savings in environmental compliance and disposal costs.
- Cost Reduction in Manufacturing: The primary driver for cost reduction in this process is the substitution of high-value chemical precursors with low-cost plant sterols. By removing the need for the intermediate 4-AD production step and subsequent chemical conversion, the manufacturer eliminates an entire stage of processing, along with its associated reagents, solvents, and purification costs. The use of methylal as a protecting agent is also economically favorable due to its low cost and ease of recovery. Additionally, the improved solubility of the protected substrate in vegetable oil enhances the volumetric productivity of the fermentation tanks, allowing for higher output per batch without increasing facility footprint. These factors combine to create a significantly leaner manufacturing process that offers a competitive edge in pricing.
- Enhanced Supply Chain Reliability: Relying on plant sterols diversifies the supply chain away from the petrochemical-dependent routes used for traditional steroid synthesis. The agricultural origin of the raw material ensures a renewable and sustainable source that is less susceptible to the geopolitical and logistical disruptions common in the synthetic chemical trade. The robustness of the Mycobacterium strains used, specifically NRRL B-3683 and 3805, further guarantees consistent biological performance across different batches and scales. This biological consistency reduces the variability in production timelines, enabling more accurate forecasting and inventory planning. The simplified process flow also reduces the number of potential failure points, enhancing the overall reliability of supply for downstream pharmaceutical customers.
- Scalability and Environmental Compliance: The fermentation-based nature of this process is inherently scalable, leveraging well-established bioreactor technologies that can be expanded from pilot scale to multi-ton production with minimal engineering risk. The use of aqueous and vegetable oil-based media reduces the fire hazard and toxicity risks associated with large volumes of flammable organic solvents. Moreover, the biological degradation of the sterol side chain is a highly specific reaction that generates fewer toxic byproducts compared to chemical oxidation methods. This cleaner profile facilitates easier regulatory approval and reduces the burden on wastewater treatment facilities. The ability to recycle the vegetable oil medium and recover the protecting agent further contributes to a circular economy model, aligning with increasingly stringent global environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this DHEA production technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a clear understanding of the process capabilities and limitations. Understanding these details is crucial for evaluating the feasibility of adopting this method for commercial manufacturing.
Q: Why is the 3-hydroxyl group protected before fermentation?
A: Protecting the 3-hydroxyl group prevents the microorganism from oxidizing it or performing unwanted side reactions, ensuring specificity for side-chain cleavage to form the 17-ketone structure directly.
Q: What are the advantages of using plant sterols over traditional chemical precursors?
A: Plant sterols are abundant, low-cost byproducts of vegetable oil refining, significantly reducing raw material costs compared to expensive chemical intermediates like acetic acid gestation diene alcohol ketone.
Q: Which bacterial strains are suitable for this transformation?
A: The patent specifies Mycobacterium sp. NRRL B-3683 and NRRL B-3805 as highly effective strains for converting protected plant sterols into the target steroid skeleton.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dehydroepiandrosterone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the microbial fermentation route for Dehydroepiandrosterone and are fully equipped to bring this technology to commercial reality. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our state-of-the-art facilities are designed to handle complex biocatalytic processes, featuring rigorous QC labs and stringent purity specifications that guarantee the highest quality intermediates for your pharmaceutical applications. We understand that transitioning to a new production method requires confidence in both the chemistry and the partner executing it.
We invite you to collaborate with us to leverage this cost-effective and sustainable manufacturing route for your DHEA requirements. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume needs, demonstrating exactly how this bio-route can improve your margins. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a reliable, high-quality supply of this critical hormonal intermediate.
