Scalable Biosynthesis of Dehydroepiandrosterone via Tandem Enzymatic Catalysis
Introduction to Advanced Steroid Biosynthesis
The pharmaceutical industry is constantly seeking more efficient and sustainable pathways for producing critical hormonal intermediates, and the recent disclosure in patent CN113493814B represents a significant leap forward in the biosynthesis of Dehydroepiandrosterone (DHEA). This vital steroid hormone precursor, known for its extensive applications in anti-aging therapies, immune regulation, and as a key building block for sex hormones, has traditionally been manufactured through cumbersome chemical processes that suffer from low atom economy and environmental hazards. The new methodology described in this patent introduces a streamlined, one-step biological enzyme catalytic reaction that converts cheap and easily obtainable steroid androstenedione ester directly into the target product with an impressive yield of up to 94 percent. By leveraging a sophisticated tandem enzymatic system, this innovation not only simplifies the synthetic route but also fundamentally alters the impurity profile, ensuring a higher quality product that meets the stringent demands of modern pharmaceutical supply chains.
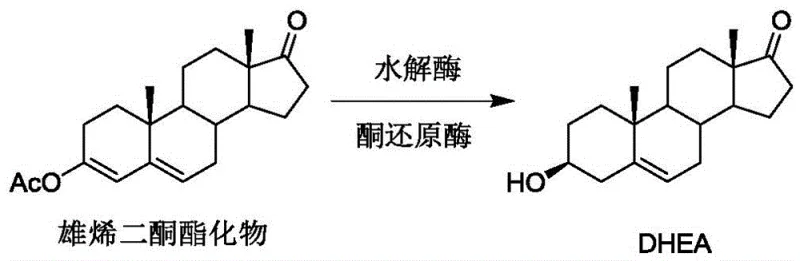
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of Dehydroepiandrosterone has relied heavily on multi-step chemical syntheses that are fraught with inefficiencies and safety concerns. For instance, earlier reported routes involved starting from dehydroepiandrosterone itself or related steroids and subjecting them to a sequence of oximation, Beckmann rearrangement, and hydrolysis reactions, which collectively resulted in a total yield of only about 70 percent while generating substantial wastewater and requiring toxic reagents like acetic anhydride. Other chemical approaches necessitated complex protection and deprotection strategies, such as ketal formation, to manage reactive carbonyl groups, adding unnecessary steps and cost to the manufacturing process. Furthermore, previous biocatalytic attempts, while an improvement, still relied on the generation of a double bond shifted Intermediate A using harsh bases like potassium tert-butoxide, a reagent known for its instability and explosive potential, which posed significant risks during scale-up and often led to the accumulation of oxidative impurities due to the inherent instability of the intermediate.
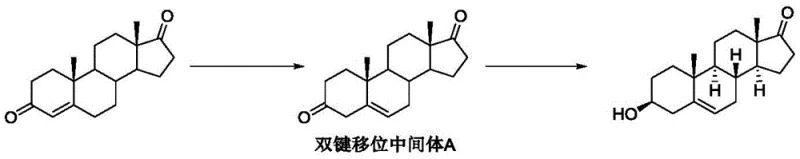
The Novel Approach
In stark contrast to these legacy methods, the novel approach disclosed in CN113493814B eliminates the need for hazardous chemical bases and complex protection groups by employing a dual-enzyme system that operates under mild, physiological conditions. This groundbreaking method utilizes a specific hydrolase to initiate the reaction on the androstenedione ester substrate, followed immediately by a selective ketoreductase that drives the transformation to completion. The brilliance of this design lies in its ability to bypass the isolation of unstable intermediates; instead of allowing the reaction to stall at the double bond shifted stage, the ketoreductase actively consumes the intermediate as it forms, pulling the equilibrium forward and preventing the degradation pathways that typically plague steroid synthesis. This results in a process that is not only environmentally friendly and safer for operators but also delivers a superior yield and purity profile that is difficult to achieve with traditional organic synthesis.
Mechanistic Insights into Tandem Enzymatic Catalysis
The core of this technological breakthrough is the synergistic interaction between the hydrolase and the ketoreductase within a single reaction vessel, creating a dynamic catalytic cycle that effectively manages thermodynamic equilibria. Upon introduction of the steroid androstenedione ester, the hydrolase catalyzes the hydrolysis of the ester group, generating an enol structure that exists in a reversible equilibrium with the desired double bond shifted Intermediate A. In a standard scenario, this equilibrium might favor the starting material or lead to side reactions, but the presence of the selective ketoreductase changes the landscape entirely by rapidly and asymmetrically reducing the 3-position ketocarbonyl of Intermediate A into the final DHEA product. This rapid consumption of Intermediate A breaks the reaction balance, continuously driving the conversion of the enol structure and ensuring that the concentration of the unstable intermediate remains negligible throughout the process.
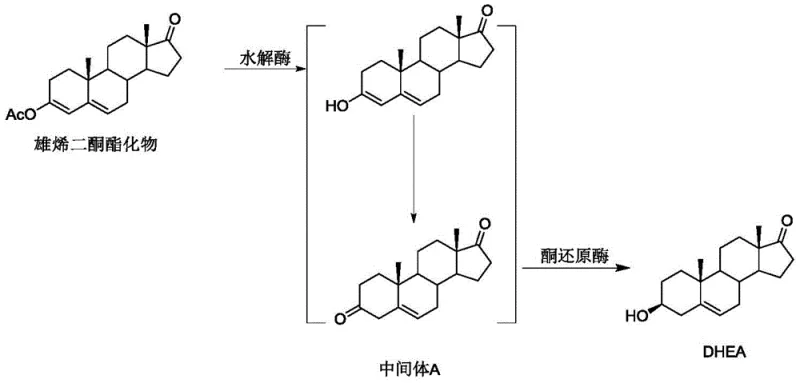
This mechanism provides a robust solution to the impurity challenges that have long hindered the commercial viability of biocatalytic steroid production. By maintaining the level of Intermediate A at a minimum, the process effectively shuts down the formation of oxidative impurities that arise from the exposure of the conjugated enone system to oxygen or other oxidants. The use of a coenzyme regeneration system, such as glucose coupled with glucose dehydrogenase, further enhances the efficiency of the ketoreductase, ensuring that the reducing equivalents are constantly replenished without the need for stoichiometric amounts of expensive cofactors. This intricate dance of enzymes ensures that the reaction proceeds with high specificity and minimal byproduct formation, offering a level of control over the stereochemistry and purity that is essential for high-value pharmaceutical intermediates.
How to Synthesize Dehydroepiandrosterone Efficiently
The implementation of this biosynthetic route requires careful optimization of the reaction medium and enzyme loading to maximize the synergistic effects of the catalytic system. The process typically involves dissolving the steroid substrate and the enzyme preparations in a biphasic or mixed solvent system containing tert-butyl alcohol and a phosphate buffer, which helps to solubilize the hydrophobic steroid while maintaining the aqueous environment necessary for enzyme activity. Detailed standardized synthesis steps for replicating this high-yield transformation are provided in the guide below.
- Dissolve steroid androstenedione ester, hydrolase, and ketoreductase in a solvent system comprising tert-butyl alcohol and phosphate buffer.
- Add nicotinamide adenine dinucleotide (NAD+) as a coenzyme along with a regeneration system such as glucose and glucose dehydrogenase.
- Maintain the reaction temperature between 30-35°C and pH 6-8 until conversion exceeds 99%, then isolate the product via crystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this enzymatic technology offers profound strategic benefits that extend far beyond simple yield improvements. The elimination of hazardous reagents like potassium tert-butoxide and the removal of multi-step protection chemistry drastically simplify the manufacturing workflow, reducing the burden on waste treatment facilities and lowering the overall cost of goods sold. By transitioning from a linear, step-wise chemical synthesis to a convergent, one-pot biological process, manufacturers can significantly reduce the capital expenditure required for reactor capacity and minimize the downtime associated with intermediate isolations and purifications.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the use of inexpensive starting materials, specifically steroid androstenedione esters, which are readily available in the bulk chemical market compared to more specialized precursors. Furthermore, the high conversion rate and the avoidance of complex downstream processing steps mean that the overall resource intensity per kilogram of product is substantially lowered, leading to meaningful cost savings in API manufacturing without compromising on quality standards.
- Enhanced Supply Chain Reliability: From a supply chain perspective, the robustness of this enzymatic route mitigates the risks associated with the sourcing of volatile or regulated chemical reagents. The reliance on stable enzyme preparations and common buffer salts ensures a more predictable and continuous production schedule, reducing the lead time for high-purity hormonal intermediates and shielding the supply chain from the fluctuations often seen in the petrochemical-derived reagent market.
- Scalability and Environmental Compliance: The mild reaction conditions, operating near neutral pH and moderate temperatures, make this process inherently scalable from laboratory benchtops to industrial fermenters without the need for exotic pressure vessels or cryogenic cooling systems. This ease of scale-up, combined with the generation of biodegradable byproducts and the absence of heavy metal catalysts, aligns perfectly with modern green chemistry initiatives and simplifies regulatory compliance for environmental discharge.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this biosynthetic pathway, derived directly from the patent specifications and experimental data. These insights are intended to clarify the operational parameters and quality outcomes expected from adopting this technology.
Q: Why does this new enzymatic route prevent the formation of oxidative impurities?
A: The process utilizes a tandem catalytic system where the ketoreductase immediately reduces the unstable Intermediate A as soon as it is formed by the hydrolase. This prevents the accumulation of Intermediate A, which is prone to oxidation and degradation in traditional chemical or single-enzyme processes.
Q: What are the safety advantages of this biosynthetic method over previous chemical routes?
A: Unlike prior art methods that require hazardous reagents like potassium tert-butoxide (which is explosive) or large amounts of acetic anhydride, this biological method operates under mild aqueous conditions (pH 6-8, 30-35°C), significantly enhancing operational safety and environmental compliance.
Q: Can this process be scaled for industrial production of API intermediates?
A: Yes, the patent demonstrates high yields (up to 94%) and uses commercially available enzymes and cheap starting materials (androstenedione ester). The absence of complex protection/deprotection steps and the use of standard fermentation-like conditions make it highly suitable for large-scale commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dehydroepiandrosterone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this enzymatic route for the global supply of steroid hormones and are fully equipped to bring this technology to commercial reality. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from patent to plant is seamless and efficient. Our state-of-the-art facilities are designed to handle complex biocatalytic processes with precision, supported by rigorous QC labs that enforce stringent purity specifications to guarantee that every batch of Dehydroepiandrosterone meets the highest international pharmacopeial standards.
We invite forward-thinking pharmaceutical companies to collaborate with us to optimize their supply chains and reduce their manufacturing footprint. By leveraging our expertise in process development, we can provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this green synthesis route can improve your bottom line. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a sustainable and cost-effective source of this critical API intermediate.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
