Advanced Synthetic Route for High-Purity Edoxaban Tosylate and Critical Stereoisomers
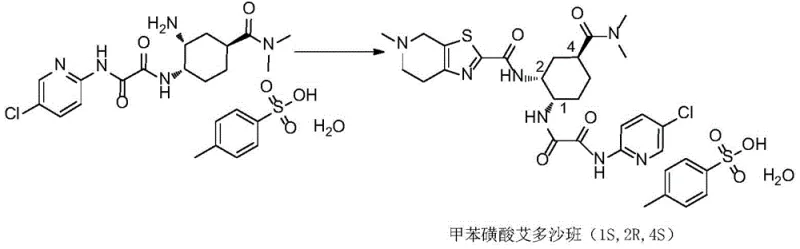
The pharmaceutical industry continuously demands higher purity standards for active pharmaceutical ingredients (APIs), particularly for complex small molecules like Edoxaban Tosylate, a potent Factor Xa inhibitor. Patent CN112321613B introduces a groundbreaking methodology for the preparation of Edoxaban Tosylate and, crucially, its specific stereoisomers. This innovation addresses a significant gap in the market where controlling trace impurities, specifically enantiomers and diastereomers, is paramount for regulatory compliance and patient safety. The disclosed technique utilizes a strategic chiral resolution approach starting from 3-cyclohexene-1-carboxylic acid, enabling the precise synthesis of the target API (1S,2R,4S) alongside its critical isomeric impurities (1R,2S,4R, 1R,2R,4S, and 1S,2S,4R). By establishing a reliable pathway to generate these reference standards, manufacturers can implement far more rigorous quality control protocols, ensuring that the final bulk drug substance meets the stringent purity specifications required by global health authorities.
This technical advancement is not merely an academic exercise but a practical solution for industrial-scale pharmaceutical intermediates production. The ability to synthesize specific isomers allows for better understanding of degradation pathways and ensures that the manufacturing process remains robust against stereochemical drift. For procurement and supply chain leaders, this translates to a more stable supply of high-quality raw materials, reducing the risk of batch failures due to impurity profiles exceeding limits. The patent details a sequence of reactions that are chemically elegant yet operationally simple, avoiding the need for exotic catalysts or extreme conditions that often plague complex asymmetric syntheses.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for Edoxaban often struggle with the precise control of stereochemistry at the cyclohexyl ring positions. Conventional methods may rely on asymmetric hydrogenation or enzymatic resolution, which can be cost-prohibitive due to the expense of chiral ligands or enzymes. Furthermore, these methods sometimes lack the flexibility to intentionally produce specific isomers needed for impurity profiling. Without access to authentic standards of the (1R,2S,4R) enantiomer or the (1R,2R,4S) epimer, quality control laboratories are forced to rely on relative retention times, which is insufficient for definitive identification and quantification. This limitation creates a bottleneck in the development of generic versions of the drug, as regulators require absolute confirmation of impurity structures. Additionally, older processes might involve multiple protection and deprotection steps that lower overall yield and increase waste generation, negatively impacting the environmental footprint and cost structure of the manufacturing process.
The Novel Approach
The methodology described in CN112321613B overcomes these hurdles by employing a classical yet highly effective chiral resolution strategy using (R)-1-phenylethylamine. This approach allows for the separation of enantiomers at an early stage, setting the stereochemical foundation for the entire synthesis. By manipulating the reaction conditions during the epoxide ring-opening and azide substitution steps, the process can be tuned to favor different stereochemical outcomes, thereby enabling the selective production of the desired API or its isomers. This versatility is a game-changer for cost reduction in pharmaceutical manufacturing, as it consolidates the production of both the drug substance and its critical quality control standards into a single, coherent platform. The use of common reagents like iodine, sodium azide, and standard coupling agents ensures that the process is easily transferable to existing multipurpose reactors without requiring specialized equipment.
Mechanistic Insights into Chiral Resolution and Stereoinversion
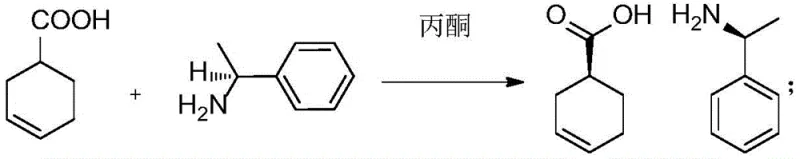
The core of this synthetic strategy lies in the initial resolution of racemic 3-cyclohexene-1-carboxylic acid. By reacting the racemate with (R)-1-phenylethylamine in acetone, a diastereomeric salt is formed. Due to the differences in solubility between the diastereomers, one specific configuration crystallizes out preferentially, allowing for physical separation. This resolved acid is then subjected to iodolactonization, where iodine and potassium carbonate facilitate the formation of an iodolactone with defined stereochemistry. The subsequent hydrolysis and epoxidation steps convert this intermediate into a key epoxide species. The stereochemical fate of the molecule is largely determined in the next step: the nucleophilic attack by azide ion. Depending on the specific isomer target, the reaction conditions and the starting chirality guide whether the stereochemistry is inverted or retained at the C-1 position. This mechanistic precision ensures that the final cyclohexyl amine scaffold possesses the exact spatial arrangement required for the subsequent amide couplings.
Impurity control is inherently built into this mechanism. Because the pathway to each isomer is distinct and well-understood, manufacturers can identify exactly where potential stereochemical leaks might occur. For instance, if the resolution step is incomplete, traces of the wrong enantiomer will carry through, appearing as the (1R,2S,4R) impurity in the final API. By monitoring the optical purity at the salt formation stage, producers can prevent downstream contamination. Furthermore, the final coupling with the 5-chloropyridine and thiazolopyridine fragments is performed under mild conditions that minimize racemization. The use of coupling reagents like EDCI and HOBT ensures efficient amide bond formation without compromising the delicate stereocenters established earlier in the sequence. This level of mechanistic control is essential for producing high-purity pharmaceutical intermediates that meet the rigorous standards of modern drug regulation.
How to Synthesize Edoxaban Tosylate Efficiently
The synthesis of Edoxaban Tosylate involves a sequential series of transformations starting from simple commodity chemicals. The process begins with the chiral resolution of the cyclohexene acid, followed by functional group manipulations to install the necessary amine and amide functionalities. Key steps include the formation of the iodolactone, conversion to an epoxide, and regioselective ring opening with azide. The resulting amine is then protected, coupled with the pyridine fragment, deprotected, and finally coupled with the thiazolopyridine acid. The detailed standardized synthesis steps see the guide below.
- Perform chiral resolution of 3-cyclohexene-1-carboxylic acid using (R)-1-phenylethylamine to establish the initial stereocenter.
- Execute iodolactonization followed by epoxide formation and ring-opening with azide to invert or retain stereochemistry as needed for specific isomers.
- Complete the synthesis via amide coupling with 5-chloropyridine and thiazolopyridine fragments, followed by tosylate salt formation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route offers substantial strategic benefits beyond mere technical feasibility. The primary advantage lies in the significant optimization of the cost structure associated with producing complex chiral molecules. By eliminating the reliance on expensive chiral catalysts or biocatalysts, the raw material costs are drastically reduced. The use of resolution, while theoretically yielding a maximum of 50% of the desired enantiomer in a single pass, is often more economically viable at scale when the resolving agent can be recovered and recycled. This leads to a leaner bill of materials and improved margin potential for the final API. Moreover, the process avoids the use of heavy metals, which simplifies the purification workflow and eliminates the costly and time-consuming step of metal scavenging, further contributing to overall cost reduction in pharmaceutical manufacturing.
- Cost Reduction in Manufacturing: The elimination of precious metal catalysts and the use of commodity reagents like iodine and sodium azide significantly lowers the direct material costs. Additionally, the streamlined purification steps reduce solvent consumption and waste disposal fees, creating a more sustainable and economical production model that enhances competitiveness in the generic drug market.
- Enhanced Supply Chain Reliability: The reliance on widely available starting materials such as 3-cyclohexene-1-carboxylic acid and (R)-1-phenylethylamine mitigates the risk of supply disruptions often associated with specialized chiral building blocks. This robustness ensures a consistent flow of intermediates, allowing manufacturers to maintain steady production schedules and meet delivery commitments even during periods of raw material volatility.
- Scalability and Environmental Compliance: The reaction conditions described, such as refluxing in acetone or stirring at ambient temperatures, are inherently scalable and safe for large-scale operations. The absence of cryogenic requirements or high-pressure hydrogenation simplifies the engineering controls needed for commercialization. Furthermore, the process generates less hazardous waste compared to transition-metal catalyzed routes, aligning with increasingly strict environmental regulations and corporate sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Edoxaban Tosylate and its isomers based on the patented technology. These insights are derived directly from the experimental data and process descriptions provided in the patent documentation, offering clarity on the feasibility and advantages of this specific synthetic route for industry stakeholders.
Q: Why is controlling Edoxaban Tosylate isomers critical for API quality?
A: Isomers such as the enantiomer (1R,2S,4R) and epimers (1R,2R,4S, 1S,2S,4R) are major process-related impurities that can affect drug safety and efficacy. High-purity reference standards are essential for rigorous QC.
Q: How does the patented resolution method improve cost efficiency?
A: By utilizing classical chiral resolution with readily available amines rather than expensive transition metal catalysts, the process significantly reduces raw material costs and simplifies downstream purification.
Q: Can this synthetic route be scaled for commercial production?
A: Yes, the patent describes robust conditions using common solvents like acetone and ethanol, avoiding cryogenic temperatures or hazardous reagents, which facilitates safe scale-up from kilograms to metric tons.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Edoxaban Tosylate Supplier
At NINGBO INNO PHARMCHEM, we understand the critical importance of purity and consistency in the production of anticoagulant APIs. Our team of expert chemists has extensively evaluated the synthetic route disclosed in CN112321613B and confirmed its viability for large-scale manufacturing. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and reliability. Our state-of-the-art facilities are equipped with rigorous QC labs capable of detecting and quantifying trace isomers, guaranteeing that every batch of Edoxaban Tosylate we deliver meets stringent purity specifications and complies with international pharmacopoeia standards.
We invite you to collaborate with us to optimize your supply chain for this vital medication. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. By leveraging our expertise in chiral resolution and process optimization, we can help you secure a stable source of high-quality intermediates and API. Contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our advanced manufacturing capabilities can support your long-term business goals.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
