Advanced Manufacturing of Amiodarone Hydrochloride via Green Lewis Acid Catalysis
Advanced Manufacturing of Amiodarone Hydrochloride via Green Lewis Acid Catalysis
The pharmaceutical industry is constantly seeking more efficient and environmentally sustainable pathways for the production of critical cardiovascular medications. A significant breakthrough in this domain is detailed in patent CN113527236B, which discloses a novel method for preparing Amiodarone Hydrochloride, a Class III antiarrhythmic agent widely used for treating severe ventricular and supraventricular arrhythmias. This innovative approach fundamentally reimagines the synthetic strategy by replacing the harsh, waste-generating Friedel-Crafts acylation traditionally used with a greener, Lewis acid-catalyzed aldol-type condensation followed by a streamlined one-pot iodination and oxidation sequence. By shifting away from stoichiometric aluminum chloride usage, this technology addresses major pain points regarding waste disposal, equipment corrosion, and product purification, offering a compelling value proposition for manufacturers aiming to optimize their supply chains.
For global procurement leaders and R&D directors, the implications of this patent extend beyond mere academic interest; it represents a tangible opportunity for cost reduction in API manufacturing and enhanced operational safety. The core of this technology lies in its ability to utilize cheap, easy-to-obtain reagents like 2-butyl benzofuran and p-acetoxy benzaldehyde under mild conditions, thereby lowering the barrier to entry for high-quality production. As a reliable amiodarone hydrochloride supplier, understanding these mechanistic shifts is crucial for evaluating the long-term viability and scalability of the supply source. The following analysis delves deep into the technical nuances, comparing this novel route against legacy methods to highlight its superiority in purity control and environmental compliance.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
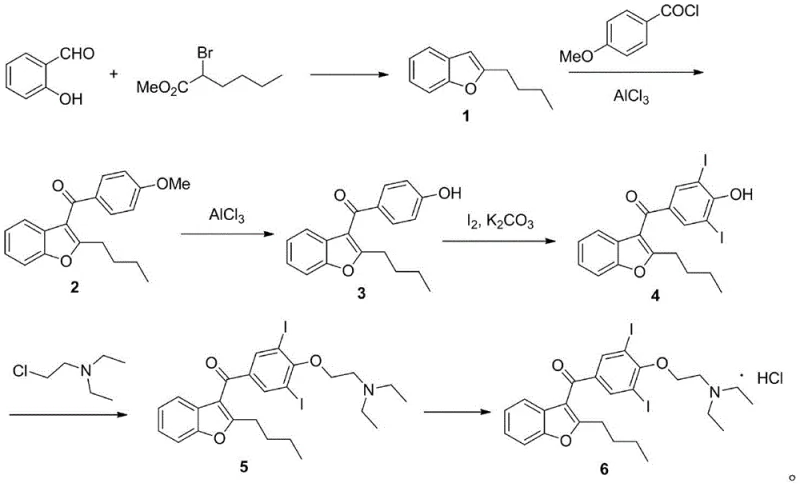
The classical industrial synthesis of Amiodarone Hydrochloride has long relied on a Friedel-Crafts acylation strategy, which, while effective, is fraught with significant chemical engineering challenges. The primary bottleneck in this traditional route is the heavy reliance on aluminum chloride (AlCl3) as a catalyst, which is required in stoichiometric or near-stoichiometric quantities to drive the reaction to completion. This excessive use of Lewis acid generates massive amounts of acidic wastewater and aluminum sludge upon quenching, creating a substantial burden on waste treatment facilities and inflating the overall environmental compliance costs. Furthermore, the reaction conditions are often harsh, requiring strict anhydrous environments and posing risks of equipment corrosion, which increases maintenance overheads for manufacturing plants.
Beyond the environmental drawbacks, the conventional method suffers from significant downstream processing difficulties, particularly regarding the purification of key intermediates. The separation of the mono-iodinated and di-iodinated species is notoriously difficult due to their similar physicochemical properties, often resulting in low isolation yields and impure final products that require extensive recrystallization or chromatography. These purification bottlenecks not only extend the production cycle time but also lead to substantial material loss, negatively impacting the overall atom economy of the process. For supply chain managers, these inefficiencies translate into higher volatility in production schedules and increased costs per kilogram of the final active pharmaceutical ingredient (API).
The Novel Approach
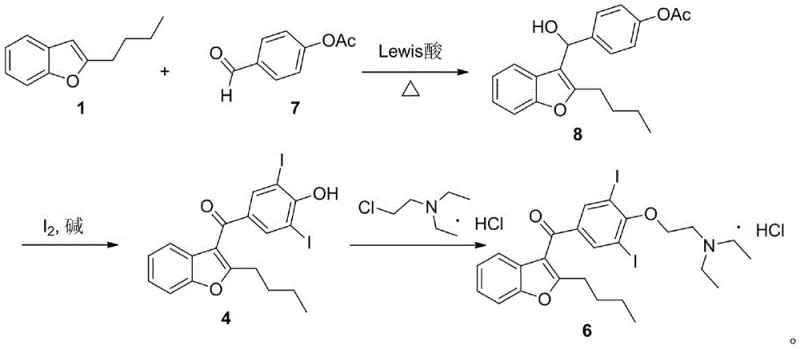
In stark contrast, the methodology outlined in patent CN113527236B introduces a paradigm shift by employing a Lewis acid-catalyzed condensation between 2-butyl benzofuran and p-acetoxy benzaldehyde. This initial step proceeds under much milder thermal conditions, typically between 60°C and 90°C, and critically, requires only a catalytic amount of the Lewis acid, such as ferric chloride or zinc chloride. This drastic reduction in catalyst loading eliminates the generation of bulk aluminum waste, simplifying the aqueous workup to a simple wash with sodium bicarbonate solution. The result is a cleaner reaction profile with fewer side products, setting a superior foundation for the subsequent steps in the synthesis.
The true ingenuity of this new route is revealed in the second step, where hydroxyl oxidation, deacetylation, and iodination occur simultaneously in a single pot using elemental iodine and a base. This telescoped operation not only saves time and solvent but also ensures that the di-iodination occurs selectively at the desired positions on the aromatic ring, minimizing the formation of regio-isomers that plague the traditional route. By integrating these transformations, the process achieves a level of operational simplicity that is rare in complex API synthesis, directly contributing to reduced labor costs and faster batch turnover times for commercial scale-up of complex antiarrhythmic drugs.
Mechanistic Insights into Lewis Acid-Catalyzed Condensation and Iodination
To fully appreciate the technical robustness of this synthesis, one must examine the mechanistic underpinnings of the Lewis acid catalysis employed in the first step. Unlike the brute-force electrophilic substitution driven by AlCl3 complexes, the interaction between the Lewis acid (e.g., FeCl3) and the carbonyl oxygen of p-acetoxy benzaldehyde activates the aldehyde towards nucleophilic attack by the electron-rich 2-position of the benzofuran ring. This activation lowers the energy barrier for the C-C bond formation, allowing the reaction to proceed efficiently at moderate temperatures without the need for aggressive reagents. The resulting intermediate, a benzylic alcohol derivative, is formed with high regioselectivity, avoiding the poly-acylation byproducts often seen in uncontrolled Friedel-Crafts reactions.
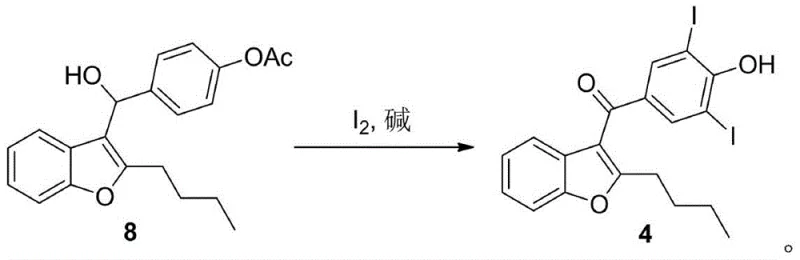
The subsequent transformation of this alcohol intermediate into the final di-iodo ketone precursor is a masterpiece of oxidative efficiency. In the presence of iodine and a base like sodium hydroxide, the benzylic alcohol is oxidized to a ketone, while the acetate protecting group is hydrolyzed to reveal the phenol. Simultaneously, the activated phenol ring undergoes electrophilic aromatic substitution with iodine. The base plays a dual role here: it facilitates the oxidation of the alcohol and activates the phenol ring towards iodination by forming the phenoxide ion, which is highly nucleophilic. This concerted mechanism ensures that the iodine atoms are installed precisely at the ortho-positions relative to the hydroxyl group, yielding the critical 3,5-diiodo structure required for Amiodarone's biological activity with exceptional purity.
From an impurity control perspective, this mechanism offers distinct advantages. The mild oxidative conditions prevent the over-oxidation of the butyl side chain or the degradation of the benzofuran ring, which are common degradation pathways in harsher acidic environments. Furthermore, the use of elemental iodine allows for precise stoichiometric control; any excess iodine can be easily quenched with sodium sulfite, leaving no toxic heavy metal residues in the product. This clean reaction profile is essential for meeting the stringent purity specifications required by regulatory bodies for cardiovascular drugs, where trace impurities can have significant safety implications.
How to Synthesize Amiodarone Hydrochloride Efficiently
Implementing this novel synthesis route requires careful attention to reaction parameters, particularly the choice of solvent and the stoichiometry of the iodination step. The patent demonstrates that solvents like 1,2-dichloroethane or dichloromethane are optimal for the initial condensation, providing a good balance of solubility and boiling point for the reflux conditions. For the iodination step, lower alcohols such as methanol or ethanol are preferred due to their ability to dissolve both the organic intermediate and the inorganic base, facilitating the homogeneous reaction necessary for high conversion rates. The detailed standardized synthesis steps below outline the precise operational window for achieving optimal yields and purity.
- React 2-butyl benzofuran with p-acetoxy benzaldehyde using a catalytic amount of Lewis acid (e.g., FeCl3) at 60-90°C to form the hydroxy-intermediate.
- Perform simultaneous hydroxyl oxidation, deacetylation, and iodination on the intermediate using iodine and alkali in alcohol solvent to obtain the di-iodo ketone.
- React the di-iodo ketone with N,N-diethyl chloroethylamine hydrochloride in the presence of base to form the final Amiodarone Hydrochloride salt.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this new synthetic route offers profound strategic benefits that go beyond simple chemical efficiency. The most immediate impact is seen in the drastic simplification of the waste management infrastructure required for production. By eliminating the need for handling and disposing of tons of aluminum chloride sludge, manufacturers can significantly reduce their environmental compliance costs and mitigate the risk of regulatory shutdowns due to waste discharge violations. This "green" credential is increasingly becoming a prerequisite for supplying to top-tier pharmaceutical companies who are under pressure to reduce the carbon footprint of their supply chains.
- Cost Reduction in Manufacturing: The economic argument for this process is compelling, primarily driven by the reduction in raw material consumption and waste treatment expenses. Since the Lewis acid is used in catalytic rather than stoichiometric quantities, the direct material cost for the catalyst is slashed. Moreover, the elimination of complex purification steps for intermediates means less solvent consumption and lower energy usage for distillation and drying. These cumulative savings translate into a more competitive cost structure for the final API, allowing suppliers to offer better pricing without compromising on quality margins.
- Enhanced Supply Chain Reliability: The robustness of this synthesis enhances supply continuity by reducing the dependency on specialized waste disposal services and corrosion-resistant equipment. The reagents involved, such as p-acetoxy benzaldehyde and elemental iodine, are commodity chemicals with stable global supply lines, reducing the risk of raw material shortages. Additionally, the milder reaction conditions place less stress on reactor vessels and piping, extending the lifespan of capital equipment and reducing unplanned maintenance downtime, which is critical for maintaining consistent delivery schedules to clients.
- Scalability and Environmental Compliance: Scaling this process from pilot plant to commercial tonnage is straightforward due to the absence of exothermic hazards associated with large-scale AlCl3 additions. The one-pot nature of the iodination step reduces the number of unit operations, minimizing the potential for human error and batch-to-batch variability. From an environmental standpoint, the process aligns perfectly with modern green chemistry principles, generating significantly less hazardous waste. This makes it easier for manufacturers to obtain necessary environmental permits and maintain a positive corporate social responsibility profile in the eyes of stakeholders.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a clear picture of what partners can expect when adopting this technology. Understanding these details is vital for making informed decisions about process validation and vendor qualification.
Q: How does this new synthesis route reduce environmental waste compared to traditional methods?
A: The traditional route relies on stoichiometric amounts of aluminum chloride (AlCl3) for Friedel-Crafts acylation, which generates significant acidic waste and requires complex hydrolysis. The new method utilizes only a catalytic amount of Lewis acid (such as ferric chloride), drastically reducing the volume of hazardous waste and simplifying the post-reaction workup.
Q: What are the purity advantages of the new intermediate crystallization process?
A: In conventional processes, separating the mono-iodo and di-iodo intermediates is challenging due to similar solubilities, often leading to low purification yields. The new route produces intermediates that are easier to crystallize directly from the reaction mixture, ensuring higher purity specifications and reducing the need for extensive chromatographic purification.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the method is highly scalable. It avoids the use of highly corrosive reagents in stoichiometric quantities and operates under milder thermal conditions (60-90°C). The reagents used, such as p-acetoxy benzaldehyde and elemental iodine, are commercially available and cost-effective, making the process robust for multi-ton manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Amiodarone Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize that the adoption of advanced synthetic routes like the one described in CN113527236B is key to staying competitive in the global pharmaceutical market. Our team of expert chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this green chemistry are fully realized in a manufacturing setting. We operate stringent purity specifications and maintain rigorous QC labs to guarantee that every batch of Amiodarone Hydrochloride meets the highest international standards for safety and efficacy, regardless of the synthetic route employed.
We invite potential partners to engage with our technical procurement team to discuss how this innovative manufacturing process can be tailored to your specific supply needs. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic advantages specific to your volume requirements. We encourage you to contact us today to索取 specific COA data and route feasibility assessments, and let us demonstrate how our commitment to technological excellence can drive value for your organization.
