Strategic Manufacturing of High-Purity Silodosin Intermediate via Novel Friedel-Crafts Route
Introduction to Advanced Silodosin Intermediate Manufacturing
The pharmaceutical landscape for Benign Prostatic Hyperplasia (BPH) treatment demands intermediates of exceptional purity and consistent stereochemical integrity. Patent CN108033906B introduces a transformative preparation method for the critical Silodosin intermediate, 1-(3-hydroxypropyl)-5-[(2R)-2-[2-[2-(2,2,2-trifluoroethoxy)phenoxy]ethylamino]propyl]-7-nitrile-1H-indoline. This technology represents a significant departure from legacy synthetic pathways, addressing long-standing industry pain points regarding toxicity, cost, and scalability. By leveraging a strategic Friedel-Crafts acylation followed by a highly selective reduction sequence, this methodology achieves high optical purity (98.1% ee) while utilizing entirely commercially available raw materials. For global procurement teams and R&D directors, this patent outlines a pathway that not only simplifies the supply chain but also drastically reduces the environmental footprint associated with traditional nitro-based syntheses.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of this complex indoline derivative has been plagued by inefficient multi-step sequences and hazardous reagent usage. As illustrated in prior art such as JP2001199956, traditional routes often rely on asymmetric reductive amination using expensive chiral auxiliaries like L-phenylglycinol, which significantly inflates raw material costs. Furthermore, these legacy processes frequently employ nitroethane, a reagent known for its safety hazards and regulatory scrutiny, alongside phosphorus oxychloride, which generates substantial volumes of difficult-to-treat acidic wastewater. The necessity for column chromatography purification in these older methods further impedes industrial scalability, creating bottlenecks in production throughput and increasing the overall cost of goods sold (COGS).
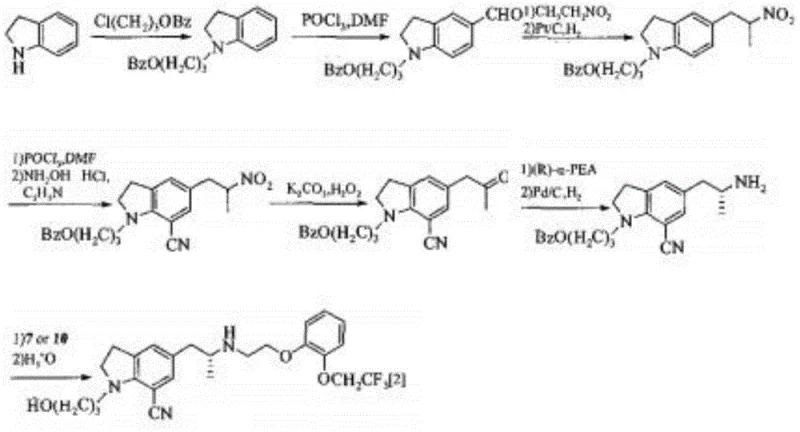
The Novel Approach
In stark contrast, the methodology disclosed in CN108033906B streamlines the synthesis into a robust seven-step sequence that prioritizes operational simplicity and safety. The core innovation lies in the early introduction of chirality via S-3-chloropropionyl chloride, eliminating the need for costly resolution steps later in the synthesis. By replacing hazardous nitro-compounds with stable acylation and reduction chemistries, the new route ensures a safer working environment and simpler waste management protocols. The elimination of column chromatography in favor of crystallization and extraction techniques demonstrates a clear design-for-manufacturing philosophy, making this approach ideally suited for reliable pharmaceutical intermediate supplier operations aiming for metric-ton scale production.
Mechanistic Insights into Friedel-Crafts Acylation and Stereoselective Reduction
The cornerstone of this synthetic strategy is the initial Friedel-Crafts reaction between 7-cyano indoline and S-3-chloropropionyl chloride, catalyzed by anhydrous aluminum trichloride. This step is critical as it establishes the carbon framework and the stereocenter simultaneously. The use of a Lewis acid catalyst ensures high regioselectivity at the 5-position of the indoline ring, minimizing the formation of regioisomeric impurities that are notoriously difficult to separate. Following acylation, the carbonyl group is reduced to a methylene group using triethylsilane in trifluoroacetic acid. This reduction protocol is particularly advantageous because it proceeds under mild conditions (5-10°C), which is essential for preserving the optical integrity of the chiral center established in the previous step, thereby preventing racemization.
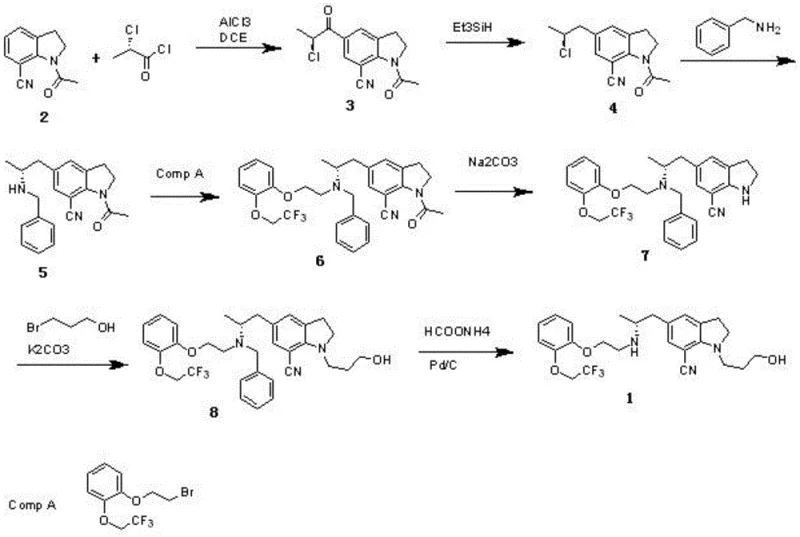
Impurity control is further enhanced in the subsequent alkylation and deprotection stages. The reaction of the intermediate amine with 2-(2,2,2-trifluoroethoxy)phenyl bromoethyl ether is conducted under basic conditions using potassium carbonate, which effectively suppresses side reactions such as over-alkylation or elimination. The final deprotection step utilizes a transfer hydrogenation system with ammonium formate and Pd/C. This choice of reducing agent is superior to high-pressure hydrogen gas for certain facility configurations, offering a safer alternative for removing the benzyl protecting group without affecting the sensitive nitrile functionality or the trifluoroethoxy ether linkage, ensuring the final product meets stringent purity specifications required for API synthesis.
How to Synthesize Silodosin Intermediate Efficiently
The synthesis of this high-value intermediate requires precise control over reaction parameters to maximize yield and optical purity. The process begins with the careful addition of S-3-chloropropionyl chloride to a cooled mixture of 7-cyano indoline and aluminum trichloride, maintaining strict temperature control to manage the exotherm. Subsequent steps involve sequential alkylation and functional group transformations that build molecular complexity while maintaining the structural integrity of the indoline core. For detailed operational parameters, including specific solvent choices like dichloroethane and DMF, and precise molar ratios optimized for scale-up, please refer to the standardized synthesis guide below.
- Perform Friedel-Crafts reaction of 7-cyano indoline with S-3-chloropropionyl chloride using AlCl3 catalyst to obtain Compound 3.
- Reduce the carbonyl group of Compound 3 to methylene using triethylsilane and trifluoroacetic acid to yield Compound 4.
- React Compound 4 with benzylamine, followed by alkylation with trifluoroethoxy phenyl bromoethyl ether to form the core amine structure.
- Execute deacetylation of the indoline nitrogen using sodium carbonate, followed by alkylation with 3-bromopropanol.
- Finalize the synthesis by removing the benzyl protecting group using ammonium formate and Pd/C catalyst to obtain the target intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented process offers compelling advantages that directly address the priorities of procurement managers and supply chain heads. The shift away from proprietary or hard-to-source chiral resolving agents towards commodity chemicals like S-3-chloropropionyl chloride and benzylamine creates a more resilient supply chain less susceptible to market volatility. The simplification of downstream processing, specifically the removal of chromatographic purification, translates to significantly reduced processing times and lower solvent consumption, which are key drivers in reducing the overall manufacturing cost.
- Cost Reduction in Manufacturing: The elimination of expensive chiral pool reagents like L-phenylglycinol and the avoidance of complex resolution steps result in substantial raw material savings. Furthermore, the high yield of each individual step cumulatively leads to a much higher overall process yield compared to prior art, effectively lowering the cost per kilogram of the final intermediate. The use of standard industrial solvents and catalysts also ensures that the process remains economically viable even at fluctuating market prices.
- Enhanced Supply Chain Reliability: By utilizing raw materials that are all commercially available off-the-shelf products, the risk of supply disruption is minimized. The process does not rely on custom-synthesized starting materials or specialized reagents that might have long lead times. This availability ensures that production schedules can be maintained consistently, providing a reliable flow of high-purity pharmaceutical intermediates to downstream API manufacturers without unexpected delays.
- Scalability and Environmental Compliance: The absence of toxic nitroethane and the reduction of phosphorus-containing waste streams significantly lower the environmental compliance burden. The process is designed with green chemistry principles in mind, utilizing safer reagents and generating less hazardous waste, which simplifies the permitting process for large-scale manufacturing facilities. The robust nature of the reaction conditions allows for seamless scale-up from pilot plant to commercial production volumes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on why this method represents a superior choice for modern pharmaceutical manufacturing.
Q: How does this new route improve optical purity compared to traditional methods?
A: The novel route utilizes S-3-chloropropionyl chloride as a chiral source in the initial Friedel-Crafts step, establishing stereochemistry early. Subsequent mild reduction conditions (5-10°C) prevent racemization, achieving up to 98.1% ee without complex resolution steps.
Q: What are the safety advantages over the JP2001199956 process?
A: Unlike the prior art which relies on hazardous nitroethane and generates difficult-to-treat phosphorus oxychloride wastewater, this method employs commercially available, non-toxic reagents and avoids heavy metal catalysts until the final mild hydrogenolysis step.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process eliminates column chromatography purification, relying instead on simple crystallization and extraction workups. The use of robust reagents like aluminum trichloride and potassium carbonate ensures high yields and operational simplicity suitable for metric-ton scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Silodosin Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development of BPH therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of Silodosin intermediate adheres to the highest standards of quality and consistency required by global regulatory bodies.
We invite you to collaborate with us to leverage this advanced synthetic technology for your supply chain needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can drive value and efficiency for your organization.
