Scalable Production of High-Purity Silodosin Key Intermediate via Safe Friedel-Crafts Cyanation
Scalable Production of High-Purity Silodosin Key Intermediate via Safe Friedel-Crafts Cyanation
The pharmaceutical industry constantly seeks robust, safe, and scalable pathways for complex active pharmaceutical ingredient (API) intermediates. A significant breakthrough in this domain is detailed in patent CN114751852A, which discloses a novel preparation method for a key intermediate of Silodosin, a potent α1A receptor antagonist used for treating Benign Prostatic Hyperplasia (BPH). This technical disclosure outlines a sophisticated synthetic route that fundamentally reimagines the construction of the indoline core, specifically targeting the critical 7-cyano and 5-chiral aminopropyl functionalities. Unlike conventional methods that rely on hazardous reagents and harsh conditions, this innovation leverages a Friedel-Crafts reaction with trichloroacetonitrile and a strategic chiral epoxide opening. For global procurement teams and R&D directors, this represents a pivotal shift towards greener, safer, and more economically viable manufacturing of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Silodosin intermediates has been plagued by significant safety and environmental hurdles that complicate commercial scale-up. Traditional routes typically employ the Vilsmeier-Haack reaction to introduce formyl groups, followed by oximation and dehydration with acetic anhydride to generate the essential 7-cyano group. This sequence is notoriously dangerous on an industrial scale due to the potential generation of trace hydrocyanic acid (HCN), a highly toxic gas that requires specialized containment and scrubbing systems. Furthermore, the installation of the 5-amino group often involves nitro reduction or reductive amination, which frequently necessitates the use of explosive sodium azide or expensive heavy metal catalysts like Palladium or Platinum. These legacy processes not only pose severe safety risks regarding explosion and toxicity but also result in phosphorus-containing wastewater and heavy metal residues that are difficult and costly to remove to meet stringent regulatory standards for API intermediates.
The Novel Approach
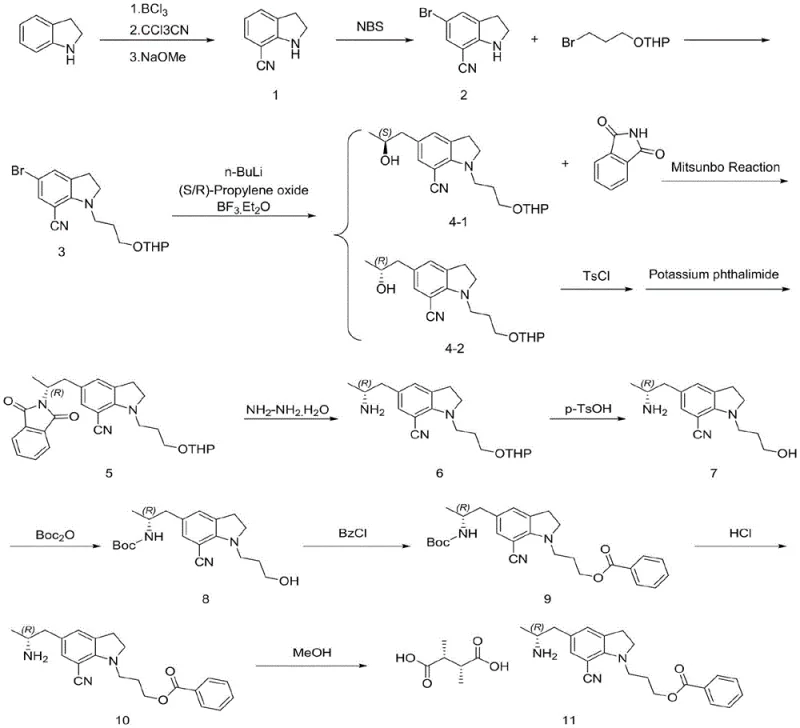
The methodology presented in patent CN114751852A offers a transformative solution by completely bypassing these hazardous steps. The new route initiates with a direct Friedel-Crafts reaction between indoline and trichloroacetonitrile, catalyzed by boron trichloride, to efficiently install the cyano group at the 7-position without generating HCN. Subsequently, the chiral center is established through a nucleophilic addition of a lithiated indoline species to chiral propylene oxide, followed by a stereochemical inversion via a Mitsunobu reaction or a tosylation-substitution sequence. This approach eliminates the need for explosive azides and heavy metal hydrogenation catalysts entirely. By integrating these safer chemical transformations, the process achieves a continuous operation capability that drastically simplifies the production workflow, reduces waste treatment costs, and facilitates the industrial scale production of Silodosin intermediates with superior purity profiles.
Mechanistic Insights into Friedel-Crafts Cyanation and Chiral Inversion
The core of this synthetic innovation lies in the mechanistic elegance of the Friedel-Crafts cyanation and the subsequent stereocontrol strategies. The reaction begins with the activation of indoline using boron trichloride (BCl3) in toluene, which likely forms a Lewis acid-base complex that enhances the nucleophilicity of the aromatic ring or activates the trichloroacetonitrile electrophile. Upon addition of trichloroacetonitrile at elevated temperatures (50-60°C), the electrophilic aromatic substitution occurs selectively at the 7-position. The resulting intermediate is then treated with sodium methoxide to effect dissociation and crystallization, yielding 7-cyanoindoline. This step is critical as it avoids the multi-step oxidation/reduction sequences of the past, directly installing the nitrile functionality with high atom economy. The subsequent bromination with N-bromosuccinimide (NBS) prepares the molecule for lithiation, where n-butyllithium generates a reactive aryl lithium species at the 5-position.
Stereochemical control is achieved through a highly selective nucleophilic attack on chiral propylene oxide in the presence of boron trifluoride diethyl etherate (BF3·Et2O). This Lewis acid catalyzes the ring opening of the epoxide, ensuring the correct regiochemistry. However, since the initial addition might yield the (S)-configuration depending on the epoxide used, the process incorporates a deliberate inversion step. This is accomplished either via a classic Mitsunobu reaction using triphenylphosphine and DIAD with phthalimide, or alternatively through tosylation followed by nucleophilic substitution with potassium phthalimide. Both pathways effectively invert the stereocenter to the desired (R)-configuration required for Silodosin activity. The phthalimide group serves as a robust protecting group for the amine, which is later cleanly removed using hydrazine hydrate, ensuring that the final amino group is free from racemization and impurities commonly associated with reductive amination routes.
How to Synthesize Silodosin Key Intermediate Efficiently
The synthesis of this complex pharmaceutical intermediate requires precise control over reaction conditions, particularly temperature and stoichiometry, to ensure high yields and purity. The process is divided into distinct operational phases: core functionalization, side-chain installation, stereochemical inversion, and final deprotection/salt formation. Each step has been optimized in the patent examples to demonstrate feasibility on a multi-kilogram scale, utilizing common organic solvents like toluene, dichloromethane, and tetrahydrofuran. The following guide summarizes the critical operational parameters derived from the patent data, highlighting the transition from raw materials to the final tartrate salt.
- Perform Friedel-Crafts reaction on indoline with trichloroacetonitrile using BCl3 to obtain 7-cyanoindoline, followed by bromination with NBS.
- Substitute the bromo-indoline with 2-(3-bromopropoxy)tetrahydro-2H-pyran to install the side chain, then perform lithiation and nucleophilic addition with chiral propylene oxide.
- Execute stereochemical inversion via Mitsunobu reaction or Tosylation/Substitution with phthalimide, followed by hydrazine deprotection, THP removal, Boc protection, benzoylation, and final salt formation with L-tartaric acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthesis route translates into tangible strategic advantages beyond mere chemical curiosity. The elimination of hazardous reagents like sodium azide and the avoidance of HCN generation significantly lowers the barrier for regulatory approval and reduces the insurance and compliance costs associated with manufacturing. Traditional routes often require specialized facilities capable of handling high-pressure hydrogenation or toxic gas scrubbing, which limits the number of qualified suppliers and creates supply chain bottlenecks. By shifting to this ambient pressure, metal-free chemistry, manufacturers can utilize standard stainless steel reactors, thereby expanding the potential supplier base and enhancing supply chain resilience against disruptions.
- Cost Reduction in Manufacturing: The economic impact of this route is profound, primarily driven by the simplification of the purification process. Since the method avoids heavy metal catalysts such as Palladium or Platinum, there is no need for expensive metal scavenging resins or rigorous testing for residual metals, which are costly and time-consuming steps in API manufacturing. Furthermore, the avoidance of the Vilsmeier reaction eliminates the generation of phosphorus-containing wastewater, significantly reducing waste treatment expenses. The continuous nature of the optimized steps allows for better throughput and reduced cycle times, leading to substantial cost savings in labor and utility consumption without compromising on the quality of the silodosin intermediate.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the reliance on specialized or hazardous raw materials. This new methodology utilizes readily available starting materials like indoline, trichloroacetonitrile, and chiral propylene oxide, which are commodity chemicals with stable global supply chains. By removing the dependency on explosive sodium azide or scarce noble metal catalysts, the risk of production stoppages due to raw material shortages or safety incidents is drastically minimized. This stability ensures a consistent flow of high-purity intermediates to downstream API manufacturers, allowing for more accurate forecasting and inventory management for long-term contracts.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies safety and environmental issues, but this route is inherently designed for industrial expansion. The reactions proceed under mild conditions—often at room temperature or moderate reflux—without the need for extreme pressures or cryogenic temperatures beyond standard industrial capabilities. The absence of heavy metals and toxic byproducts aligns perfectly with modern Green Chemistry principles and increasingly strict environmental regulations in major pharmaceutical markets. This compliance facilitates faster technology transfer from lab to plant and reduces the likelihood of regulatory shutdowns, ensuring a sustainable and scalable supply of complex pharmaceutical intermediates for the global market.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Silodosin intermediates using this advanced methodology. These insights are derived directly from the experimental data and beneficial effects described in the patent literature, providing clarity on safety, purity, and scalability for potential partners and technical stakeholders.
Q: How does this synthesis route improve safety compared to traditional Vilsmeier methods?
A: This novel route eliminates the Vilsmeier reaction, hydroxylamine oximation, and acetic anhydride dehydration steps traditionally used to introduce the 7-cyano group. By avoiding these steps, the process prevents the generation of hazardous hydrocyanic acid (HCN) and phosphorus-containing wastewater, significantly enhancing operational safety and environmental compliance.
Q: Does this method involve heavy metal catalysts that could contaminate the final API?
A: No, the process is designed to be metal-free regarding catalytic hydrogenation or cross-coupling. It explicitly avoids the use of heavy metals such as Palladium (Pd), Platinum (Pt), or Zinc (Zn) which are often required in nitro reduction or reductive amination steps in older routes. This ensures the final product has negligible heavy metal residues, simplifying downstream purification.
Q: How is the chiral center at the 5-position controlled without resolution?
A: The chirality is introduced early in the synthesis using chiral propylene oxide ((S) or (R)) during the nucleophilic addition step. The configuration is subsequently inverted via a Mitsunobu reaction or a tosylation/substitution sequence with potassium phthalimide. This strategy avoids the low atom utilization associated with traditional L-tartaric acid resolution of racemic mixtures.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Silodosin Intermediate Supplier
The technical advancements detailed in patent CN114751852A underscore the immense potential for producing high-quality Silodosin intermediates through safer and more efficient chemistry. At NINGBO INNO PHARMCHEM, we pride ourselves on being at the forefront of adopting such innovative synthetic routes to deliver superior value to our global clients. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this metal-free, azide-free process are fully realized in a GMP-compliant manufacturing environment. Our rigorous QC labs and stringent purity specifications guarantee that every batch of silodosin intermediate meets the exacting standards required for final API synthesis.
We invite pharmaceutical companies and procurement leaders to explore how this optimized route can enhance their supply chain efficiency and reduce overall manufacturing costs. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data, route feasibility assessments, and samples, allowing you to validate the superior quality and reliability of our Silodosin intermediate supply for your next project.
