Advanced Synthesis of Amide-Side Chain Pleuromutilin Derivatives for Commercial Scale-Up
The pharmaceutical industry faces a critical challenge in combating multidrug-resistant Gram-positive bacteria, particularly methicillin-resistant Staphylococcus aureus (MRSA). Patent CN111574395A introduces a groundbreaking class of pleuromutilin derivatives featuring a novel amide side chain at the C14 position. These compounds represent a significant evolution in antibiotic design, offering potent antibacterial activity alongside improved physicochemical properties. Unlike traditional pleuromutilins which may suffer from limited solubility or complex synthesis, this new series leverages a streamlined synthetic pathway to introduce diverse amide functionalities. For R&D directors and procurement specialists, this technology offers a reliable pleuromutilin derivative supplier opportunity that addresses both efficacy and manufacturability concerns in the development of next-generation anti-infectives.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the modification of the pleuromutilin core has been constrained by the chemical stability of the tricyclic diterpene skeleton and the reactivity of the C14 hydroxyl group. Conventional methods often rely on ester linkages which can be metabolically unstable or require harsh conditions that degrade the sensitive 5-6-8 ring system. Furthermore, many existing semi-synthetic routes involve multiple protection and deprotection steps that drastically increase the cost of goods sold (COGS) and reduce overall yield. The reliance on expensive coupling reagents or difficult-to-remove catalysts in older methodologies creates significant bottlenecks for supply chain heads looking to secure cost reduction in antibiotic manufacturing. Additionally, the poor water solubility of many first-generation pleuromutilins limits their formulation options, often requiring complex salt formation or solubilizing excipients that complicate the final drug product development.
The Novel Approach
The methodology described in the patent overcomes these hurdles by introducing a robust amide linkage strategy that enhances metabolic stability while maintaining high affinity for the bacterial ribosome. By converting the C14 hydroxyl into a leaving group and subsequently displacing it with an amine precursor, the process allows for the modular attachment of various benzamide or piperazine-amide moieties. This approach not only simplifies the synthetic sequence but also enables rapid structure-activity relationship (SAR) studies through the use of diverse acyl chlorides. The resulting compounds demonstrate superior water solubility, particularly when formulated as sulfate salts, which is a critical parameter for parenteral or oral bioavailability. This novel chemistry provides a scalable platform for generating high-purity pharmaceutical intermediates that meet stringent regulatory standards for impurity profiles.
Mechanistic Insights into C14 Side Chain Functionalization
The core of this innovation lies in the precise functionalization of the C14 position through a four-step cascade involving activation, substitution, reduction, and acylation. Initially, the secondary hydroxyl group at C14 is activated via tosylation using p-toluenesulfonyl chloride, creating a highly reactive sulfonate ester capable of undergoing nucleophilic substitution. This intermediate is then treated with sodium azide in an aprotic solvent to install the nitrogen functionality, followed by a Staudinger reduction using triphenylphosphine to generate the primary amine. This amine serves as a versatile handle for the final diversification step, where it reacts with activated carboxylic acid derivatives to form the stable amide bond. The mechanistic elegance of this route ensures that the stereochemistry of the rigid pleuromutilin core is preserved throughout the transformation, minimizing the formation of diastereomeric impurities.
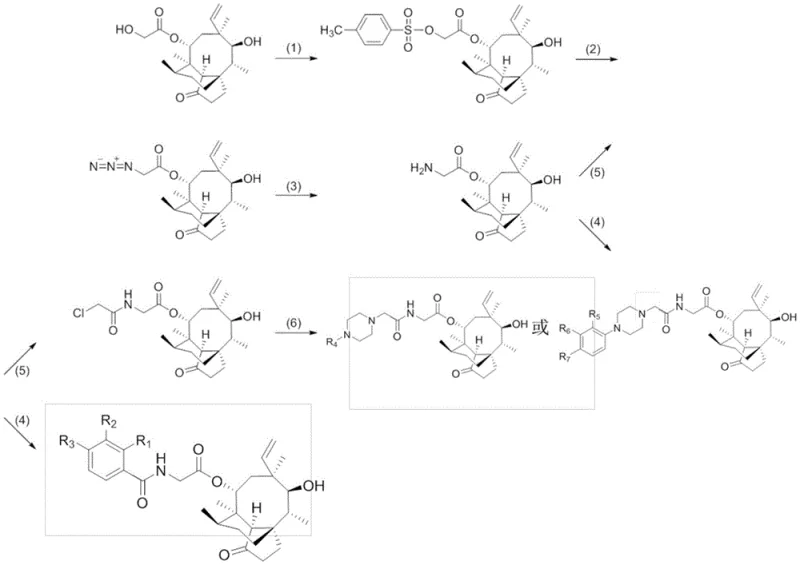
Impurity control is meticulously managed through the selection of mild reaction conditions and efficient purification techniques. For instance, the azidation step is conducted under reflux in acetone or DMF, which maximizes conversion while minimizing side reactions on the sensitive enone system. The subsequent acylation is performed under ice-bath conditions to prevent over-reaction or degradation of the thermally labile intermediates. The use of column chromatography with specific solvent systems, such as dichloromethane and methanol gradients, allows for the effective separation of the target amide from unreacted starting materials and byproducts. This rigorous control over the reaction environment ensures that the final active pharmaceutical ingredient (API) intermediate meets the high purity specifications required for clinical development, reducing the burden on downstream processing teams.
How to Synthesize Pleuromutilin Amide Derivatives Efficiently
The synthesis of these high-value intermediates follows a logical progression designed for reproducibility and scalability. The process begins with the activation of the natural product feedstock, followed by sequential functional group interconversions that build complexity in a controlled manner. Each step has been optimized to balance reaction rate with selectivity, ensuring that the delicate tricyclic framework remains intact. Detailed standard operating procedures for each transformation, including specific molar ratios, solvent volumes, and temperature profiles, are essential for achieving consistent results across different batch sizes. The following guide outlines the critical operational parameters necessary for successful execution of this chemistry in a GMP-compliant environment.
- React pleuromutilin with p-toluenesulfonyl chloride to form Intermediate I.
- Convert Intermediate I to azide Intermediate II using sodium azide in aprotic solvent.
- Reduce the azide to an amine (Intermediate III) using triphenylphosphine.
- Perform final acylation with benzoyl chloride derivatives or activated piperazine precursors to yield the target compound.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthetic route offers substantial advantages that directly impact the bottom line and supply security. The reliance on commodity chemicals such as p-toluenesulfonyl chloride, sodium azide, and various benzoyl chlorides means that raw material sourcing is straightforward and less susceptible to geopolitical supply disruptions. The elimination of precious metal catalysts or exotic reagents significantly lowers the input costs, facilitating cost reduction in antibiotic manufacturing without compromising quality. Furthermore, the robustness of the reaction conditions allows for flexibility in equipment selection, meaning that existing multipurpose reactors can be utilized without the need for specialized infrastructure investments. This adaptability shortens the timeline for technology transfer from pilot plant to commercial production, enabling faster time-to-market for new drug candidates.
- Cost Reduction in Manufacturing: The synthetic pathway eliminates the need for expensive chiral catalysts or complex enzymatic resolutions by leveraging the inherent chirality of the natural pleuromutilin starting material. This direct utilization of the chiral pool significantly reduces material costs and waste generation associated with racemic synthesis. Additionally, the high yields reported in the experimental examples indicate efficient atom economy, which translates to lower solvent consumption and reduced waste disposal costs. By streamlining the number of isolation steps and utilizing common purification methods, the overall processing time is minimized, leading to substantial operational savings.
- Enhanced Supply Chain Reliability: The starting materials and reagents required for this synthesis are widely available from multiple global suppliers, mitigating the risk of single-source dependency. The chemical stability of the key intermediates allows for potential stockpiling or campaign manufacturing, ensuring a continuous supply of critical intermediates even during market fluctuations. The scalability of the process from gram to kilogram scales has been demonstrated, providing confidence that supply can be ramped up rapidly to meet clinical or commercial demand. This reliability is crucial for maintaining uninterrupted production schedules for downstream API manufacturers.
- Scalability and Environmental Compliance: The process utilizes standard organic solvents that can be easily recovered and recycled, aligning with green chemistry principles and reducing the environmental footprint of the manufacturing operation. The absence of heavy metals in the catalytic steps simplifies the purification process and ensures that the final product meets strict residual metal limits set by regulatory agencies. The robust nature of the reaction conditions allows for safe operation in large-scale reactors, minimizing the risk of thermal runaways or hazardous incidents. This combination of safety, efficiency, and environmental stewardship makes the process highly attractive for long-term commercial partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these novel pleuromutilin derivatives. Understanding these aspects is vital for stakeholders evaluating the feasibility of integrating this technology into their development pipelines. The answers are derived directly from the technical specifications and experimental data provided in the patent documentation, ensuring accuracy and relevance for decision-makers.
Q: What is the primary advantage of these new pleuromutilin derivatives?
A: These derivatives exhibit excellent in vitro antibacterial activity against MRSA and improved water solubility compared to existing drugs like Retapamulin, while offering a lower cost of preparation.
Q: How does the C14 side chain modification affect efficacy?
A: Modifying the C14 side chain allows the molecule to penetrate deeper into the hydrophobic pocket of the bacterial ribosome 50S subunit, enhancing inhibition of protein synthesis.
Q: Is this synthesis route suitable for large-scale production?
A: Yes, the route utilizes standard organic transformations such as tosylation and acylation with readily available reagents, making it highly adaptable for commercial scale-up from kilograms to metric tons.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pleuromutilin Derivative Supplier
NINGBO INNO PHARMCHEM stands at the forefront of custom synthesis, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the specific requirements of pleuromutilin chemistry, ensuring stringent purity specifications and rigorous QC labs validate every batch. We understand the critical nature of antibiotic supply chains and are committed to delivering high-quality intermediates that accelerate your drug development timelines. Our team of expert chemists is ready to optimize this patented route for your specific needs, ensuring a seamless transition from laboratory discovery to industrial reality.
We invite you to contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your project volume. By partnering with us, you gain access to specific COA data and route feasibility assessments that demonstrate the viability of this advanced synthetic approach. Let us help you secure a competitive edge in the antibacterial market with our reliable supply of high-performance pleuromutilin derivatives.
