Advanced Pleuromutilin Derivatives: Scalable Synthesis of Thiazole-Pyridine Quaternary Ammonium Salts for Resistant Infections
The escalating crisis of antimicrobial resistance represents one of the most formidable challenges facing the global pharmaceutical industry today, necessitating the urgent development of novel chemical entities with enhanced efficacy profiles. Patent CN114671865A discloses a groundbreaking class of pleuromutilin derivatives featuring a thiazole-pyridine alkyl quaternary ammonium salt side chain, which demonstrates superior antibacterial activity against multidrug-resistant pathogens. This technical insight report analyzes the synthetic methodology and commercial viability of these compounds, highlighting their potential as next-generation therapeutic agents for treating infectious diseases. For R&D directors and procurement specialists, understanding the structural advantages and process robustness of this patent is critical for securing a reliable supply chain of high-purity pharmaceutical intermediates. The integration of a quaternary ammonium moiety onto the pleuromutilin core not only modifies the pharmacokinetic properties but also significantly lowers the minimum inhibitory concentration (MIC) against resistant strains such as MRSA and VRE. As a leading fine chemical manufacturer, we recognize the strategic value of this technology in diversifying antibiotic portfolios and mitigating the risks associated with traditional treatment failures.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional pleuromutilin antibiotics, while effective against certain Gram-positive bacteria, often suffer from limited spectrum activity and the rapid emergence of bacterial resistance mechanisms that render them less effective over time. Conventional semi-synthetic modifications frequently involve complex multi-step sequences that require harsh reaction conditions, expensive catalysts, or difficult-to-remove impurities, which can drastically increase manufacturing costs and extend lead times. Furthermore, many existing derivatives fail to adequately penetrate the cell walls of Gram-negative bacteria or specific resistant strains like methicillin-resistant Staphylococcus aureus, limiting their clinical utility in severe infection scenarios. The reliance on older chemical scaffolds without significant side-chain engineering often results in compounds that are easily effluxed by bacterial pumps or degraded by metabolic enzymes, leading to suboptimal therapeutic outcomes. Supply chain managers often face challenges in sourcing these older intermediates due to fluctuating raw material availability and stringent environmental regulations surrounding legacy synthesis routes. Consequently, there is a pressing need for innovative chemical modifications that can overcome these biological and process-related bottlenecks while maintaining economic feasibility for large-scale production.
The Novel Approach
The novel approach detailed in the patent data introduces a strategic structural modification at the C-14 position of the pleuromutilin core, incorporating a thiazole-pyridine alkyl quaternary ammonium salt side chain that fundamentally alters the compound's interaction with bacterial targets. This specific chemical architecture leverages the cationic nature of the quaternary ammonium group to enhance electrostatic attraction to the negatively charged bacterial cell surface, thereby improving membrane permeability and intracellular accumulation. Unlike conventional methods that may rely on simple esterification or etherification, this route utilizes a robust three-step synthesis that ensures high regioselectivity and minimizes the formation of difficult-to-separate isomers. The use of readily available reagents such as 2-mercapto-4-(4-pyridyl)thiazole and various bromoalkanes allows for significant flexibility in tuning the lipophilicity and biological activity of the final product. From a commercial perspective, this methodology eliminates the need for precious metal catalysts and operates under mild conditions, which translates to substantial cost reduction in pharmaceutical intermediate manufacturing and a reduced environmental footprint. The result is a versatile platform technology capable of generating a library of derivatives with optimized potency against a broad spectrum of drug-resistant pathogens.
Mechanistic Insights into Thiazole-Pyridine Quaternization
The synthetic pathway begins with the activation of the C-14 hydroxyl group of pleuromutilin, a critical step that sets the stage for subsequent nucleophilic substitutions. In this initial phase, pleuromutilin is reacted with p-toluenesulfonyl chloride in a dichloromethane solvent system at room temperature, converting the hydroxyl group into a tosylate leaving group with high efficiency. This activation is essential because the native hydroxyl group is a poor leaving group, and direct substitution would require prohibitively harsh conditions that could degrade the sensitive macrocyclic lactone structure. The reaction proceeds with a molar ratio of 1:1.2 favoring the sulfonyl chloride to ensure complete conversion, and the use of dichloromethane provides an ideal medium for dissolving both the organic substrate and the reagent while facilitating easy workup. Following the reaction, the mixture is concentrated and washed with saturated sodium bicarbonate solution to remove acidic by-products, yielding Intermediate I with a high degree of purity. This step exemplifies the process's commitment to operational safety and yield optimization, avoiding the use of hazardous reagents that could complicate industrial scale-up.
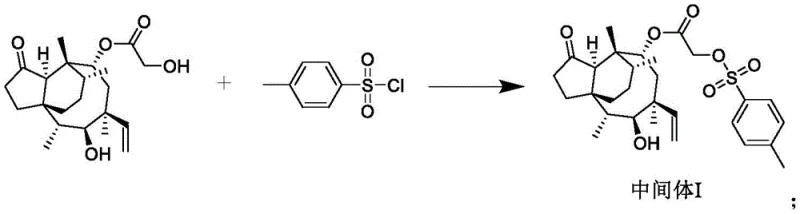
Following the activation step, the synthesis proceeds to the introduction of the heterocyclic moiety, which is pivotal for the compound's enhanced biological activity. Intermediate I is reacted with 2-mercapto-4-(4-pyridyl)thiazole in N,N-dimethylformamide (DMF) under basic conditions provided by potassium carbonate and potassium iodide. The reaction is heated to 60°C for 6 hours, allowing the thiol group of the thiazole derivative to displace the tosylate group via a nucleophilic substitution mechanism. The addition of potassium iodide acts as a catalyst to facilitate the displacement reaction, ensuring high conversion rates and minimizing the formation of elimination by-products. This step results in the formation of Intermediate II, which now possesses the core thiazole-pyridine structure necessary for the final quaternization. The choice of DMF as a solvent is strategic, as it effectively solubilizes the polar intermediates and inorganic bases, promoting homogeneous reaction conditions. After the reaction is complete, the product is isolated through extraction with ethyl acetate and purified via column chromatography, ensuring that the final intermediate meets stringent quality specifications before proceeding to the final step.
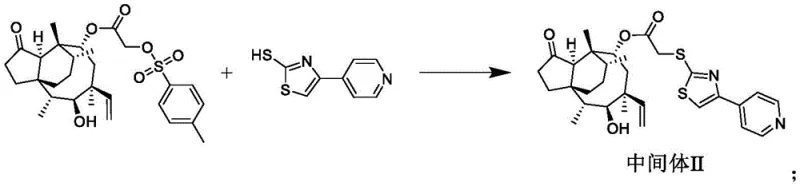
The final stage of the synthesis involves the quaternization of the pyridine nitrogen atom, which imparts the cationic charge responsible for the compound's superior antibacterial properties. Intermediate II is reacted with various substituted bromoalkanes in solvents such as acetonitrile, toluene, or acetone at room temperature for 8 to 12 hours. This alkylation step converts the neutral pyridine ring into a positively charged quaternary ammonium salt, significantly enhancing the molecule's ability to interact with bacterial cell membranes. The flexibility in choosing the alkyl group (R) allows for the fine-tuning of the compound's physicochemical properties, enabling the optimization of potency against specific resistant strains. The reaction conditions are remarkably mild, occurring at room temperature, which reduces energy consumption and minimizes the risk of thermal degradation of the sensitive pleuromutilin scaffold. Purification is achieved through column chromatography using dichloromethane and methanol mixtures, yielding the final pleuromutilin derivatives with high purity. This robust and scalable three-step sequence demonstrates a clear advantage over more complex synthetic routes, offering a reliable pathway for the commercial production of these advanced antibiotic intermediates.
How to Synthesize Pleuromutilin Derivatives Efficiently
The synthesis of these high-value pleuromutilin derivatives is designed for operational efficiency and scalability, making it an ideal candidate for commercial manufacturing environments. The process utilizes standard organic synthesis techniques and readily available reagents, reducing the barrier to entry for production and ensuring a stable supply chain. Each step has been optimized to maximize yield and minimize waste, with specific attention paid to purification protocols that ensure the removal of trace impurities and residual solvents. The use of mild reaction temperatures and common solvents like dichloromethane and acetonitrile simplifies the engineering requirements for reactors and recovery systems. Detailed standardized synthesis steps are provided below to guide process development teams in replicating this high-performance route.
- Activate the C-14 hydroxyl group of pleuromutilin using p-toluenesulfonyl chloride in dichloromethane at room temperature to form Intermediate I.
- React Intermediate I with 2-mercapto-4-(4-pyridyl)thiazole in DMF with potassium carbonate and potassium iodide at 60°C to yield Intermediate II.
- Perform quaternization by reacting Intermediate II with substituted bromoalkanes in acetonitrile or toluene at room temperature to obtain the final derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers significant strategic advantages in terms of cost stability and supply reliability. The elimination of expensive transition metal catalysts and the use of commodity chemicals for reagents drastically reduce the raw material cost base, insulating the production process from volatile market prices associated with precious metals. Furthermore, the mild reaction conditions reduce energy consumption and equipment wear, contributing to lower overall operational expenditures and a more sustainable manufacturing footprint. The robustness of the synthesis ensures consistent batch-to-batch quality, reducing the risk of production delays caused by failed batches or complex purification challenges. This reliability is crucial for maintaining continuous supply to pharmaceutical partners who require strict adherence to quality timelines for drug development programs.
- Cost Reduction in Manufacturing: The synthetic pathway avoids the use of costly palladium or platinum catalysts often found in cross-coupling reactions, relying instead on inexpensive inorganic bases and common alkylating agents. This shift in reagent profile leads to substantial cost savings in the bill of materials, allowing for more competitive pricing of the final active pharmaceutical ingredient. Additionally, the high yields reported in the patent examples indicate efficient atom economy, minimizing the waste of valuable starting materials and reducing disposal costs. The simplified workup procedures, involving standard extractions and washes, further reduce labor and solvent recovery costs compared to more intricate purification methods. These factors combine to create a highly cost-effective manufacturing process that enhances the commercial viability of the final drug product.
- Enhanced Supply Chain Reliability: The starting materials, including pleuromutilin and p-toluenesulfonyl chloride, are commercially available from multiple global suppliers, reducing the risk of single-source dependency. The use of standard solvents and reagents ensures that procurement teams can easily source materials without facing long lead times or geopolitical supply constraints. The scalability of the process from gram to kilogram scale without significant re-optimization allows for rapid ramp-up of production capacity to meet sudden increases in demand. This flexibility is essential for responding to public health emergencies or unexpected surges in antibiotic requirements. By securing a supply chain based on robust and widely available chemistry, organizations can mitigate the risks of disruption and ensure continuous availability of critical medical intermediates.
- Scalability and Environmental Compliance: The process operates under mild conditions with minimal generation of hazardous by-products, aligning with modern green chemistry principles and environmental regulations. The solvents used are common and can be efficiently recovered and recycled, reducing the overall environmental impact of the manufacturing process. The absence of heavy metal residues simplifies the purification process and ensures that the final product meets stringent regulatory limits for impurities. This compliance reduces the burden on quality control laboratories and accelerates the regulatory approval process for new drug applications. The scalable nature of the reaction allows for seamless transition from pilot plant to full commercial production, ensuring that supply can grow in tandem with market demand without compromising quality or safety standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of these novel pleuromutilin derivatives. The answers are derived directly from the patent data and reflect the practical considerations for implementing this technology in a commercial setting. Understanding these details is essential for stakeholders evaluating the feasibility of integrating these intermediates into their development pipelines.
Q: How does the thiazole-pyridine side chain improve antibacterial activity?
A: The introduction of the thiazole-pyridine alkyl quaternary ammonium salt side chain enhances the compound's ability to interact with bacterial cell walls, significantly improving efficacy against drug-resistant strains like MRSA and VRE compared to traditional pleuromutilins.
Q: Is the synthesis process suitable for large-scale manufacturing?
A: Yes, the patented method utilizes mild reaction conditions, common organic solvents, and readily available reagents, avoiding expensive transition metal catalysts, which makes it highly adaptable for commercial scale-up from kilograms to metric tons.
Q: What are the primary impurities controlled during synthesis?
A: The process includes specific purification steps such as column chromatography and washing with saturated solutions to remove unreacted starting materials and by-products, ensuring high purity specifications required for pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pleuromutilin Derivative Supplier
At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and reliability. Our technical team is well-versed in the complexities of semi-synthetic antibiotic manufacturing and is equipped to handle the specific purification requirements of pleuromutilin derivatives. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch meets the highest industry standards. Our commitment to quality and consistency makes us the ideal partner for pharmaceutical companies seeking to develop next-generation anti-infective therapies based on this innovative patent technology.
We invite you to contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific production volumes and quality requirements. Our experts are ready to provide specific COA data and route feasibility assessments to help you accelerate your development timeline. By partnering with us, you gain access to a reliable supply chain and deep technical expertise that can drive your project from concept to commercial success. Reach out today to discuss how we can support your needs for high-purity pharmaceutical intermediates.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
