Advanced Synthesis of N-Alkyl/N-Aryl Thioamide Derivatives for Pharmaceutical Applications
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies for constructing sulfur-containing heterocycles and functional groups, particularly thioamides, which serve as pivotal scaffolds in drug discovery and agrochemical development. Patent CN111056889A, published in April 2020, introduces a groundbreaking synthetic strategy for a broad class of N-alkyl/N-aryl thioamide derivatives. This technology addresses the longstanding challenges associated with thioamide synthesis by leveraging a highly efficient nucleophilic substitution reaction between amine thiocarbonyl fluorides and malonates. The core innovation lies in the utilization of the thiocarbonyl fluoride moiety as a highly reactive electrophile, which undergoes smooth displacement by malonate anions under mild basic conditions. As illustrated in the general structural formula below, the resulting derivatives possess significant structural diversity, allowing for extensive optimization of biological activity.
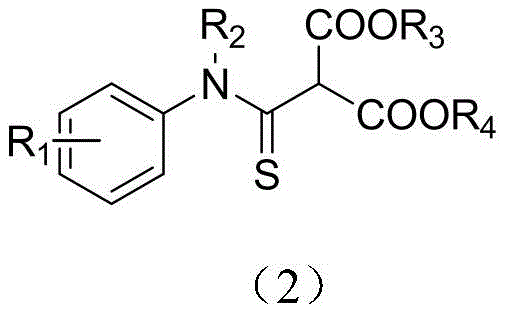
These compounds are not merely academic curiosities; they represent high-value pharmaceutical intermediates with potential applications in anti-inflammatory, antibacterial, and anti-tuberculosis therapies. The ability to introduce diverse ester groups and aryl substitutions at the alpha-position of the thioamide carbonyl opens up vast chemical space for medicinal chemists. For procurement and supply chain leaders, understanding the underlying chemistry of this patent is crucial, as it signals a shift towards more sustainable and cost-effective manufacturing processes for sulfur-containing building blocks. The method's reliance on stable starting materials and its operation at moderate temperatures suggest a high degree of process safety and scalability, which are critical factors for reliable fine chemical intermediates supplier partnerships.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of thioamide derivatives has been plagued by significant operational and environmental drawbacks that hinder their widespread adoption in large-scale pharma intermediates manufacturing. Traditional routes often rely on the thionation of corresponding amides using reagents such as Lawesson's reagent or phosphorus pentasulfide. These reagents are notoriously difficult to handle due to their unpleasant odor, high toxicity, and sensitivity to moisture, posing severe safety risks in a production environment. Furthermore, these classical methods frequently suffer from poor atom economy, generating substantial amounts of phosphorus-containing waste that requires complex and costly disposal procedures. Another common approach involves the reaction of amines with dithioesters or acid chlorides followed by sulfurization, which typically requires multiple steps, harsh reaction conditions, and the use of expensive transition metal catalysts that leave behind trace metal impurities. These impurities necessitate additional purification steps, such as scavenging or recrystallization, which drastically reduce overall yield and increase the cost of goods sold (COGS). Consequently, there has been an urgent demand within the industry for a greener, more direct synthetic pathway that eliminates these bottlenecks.
The Novel Approach
The methodology disclosed in CN111056889A represents a paradigm shift by employing amine thiocarbonyl fluorides as key precursors, which react directly with malonates in a single pot. This novel approach bypasses the need for hazardous thionating agents and multi-step sequences, thereby streamlining the production workflow significantly. The reaction proceeds via a nucleophilic substitution mechanism where the fluoride ion acts as an excellent leaving group, driven by the formation of stable inorganic fluoride salts. By utilizing inexpensive inorganic bases such as cesium carbonate or potassium carbonate, the process avoids the use of costly organometallic reagents. The operational simplicity is further enhanced by the use of common organic solvents like acetonitrile or tetrahydrofuran, which are easily recovered and recycled. This direct construction of the C-C bond adjacent to the thiocarbonyl group allows for the rapid assembly of complex molecular architectures with high precision. For R&D teams, this means faster iteration cycles in lead optimization, while for production teams, it translates to a simplified unit operation that reduces equipment downtime and maintenance costs associated with corrosive reagents.
Mechanistic Insights into Nucleophilic Substitution on Thiocarbonyl Fluorides
The success of this synthetic route hinges on the unique reactivity profile of the amine thiocarbonyl fluoride functionality. Unlike traditional acid fluorides which are prone to rapid hydrolysis, thiocarbonyl fluorides exhibit remarkable stability towards water while remaining highly susceptible to nucleophilic attack by carbon nucleophiles under basic conditions. The reaction mechanism initiates with the deprotonation of the active methylene group in the malonate ester by the inorganic base, generating a resonance-stabilized enolate anion. This nucleophile then attacks the electrophilic carbon of the thiocarbonyl group in the amine thiocarbonyl fluoride. The presence of the sulfur atom enhances the electrophilicity of the carbonyl carbon compared to its oxygen analogue, facilitating the addition step. Subsequently, the expulsion of the fluoride ion restores the double bond character of the thiocarbonyl group, yielding the final thioamide product. The general reaction scheme below clearly depicts this transformation, highlighting the preservation of the ester functionalities which are vital for downstream derivatization.
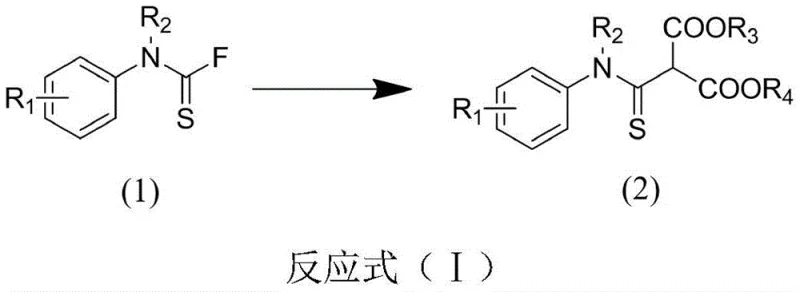
From an impurity control perspective, this mechanism offers distinct advantages over radical-based or transition-metal catalyzed pathways. Since the reaction does not involve free radicals or metal complexes, the formation of side products such as homocoupling dimers or metal-complexed impurities is virtually eliminated. The primary byproduct is the inorganic fluoride salt, which is insoluble in the organic reaction medium and can be easily removed by simple filtration. This inherent cleanliness of the reaction profile ensures that the crude product possesses high purity, reducing the burden on downstream purification units. For quality assurance teams, this translates to a more consistent impurity profile and easier validation of the cleaning procedures. Furthermore, the tolerance of the reaction conditions to various functional groups—such as halogens, esters, and ethers—means that protecting group strategies can often be minimized, further enhancing the step economy and overall efficiency of the synthesis.
How to Synthesize N-Alkyl/N-Aryl Thioamide Derivatives Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to stoichiometry and reaction monitoring to maximize yield and purity. The patent specifies a molar ratio of amine thiocarbonyl fluoride to malonate of approximately 1:1.5, ensuring that the electrophile is fully consumed while minimizing excess reagent waste. The choice of base is critical, with cesium carbonate showing superior performance in terms of reaction rate and conversion, although potassium carbonate serves as a cost-effective alternative for less demanding substrates. The reaction is typically conducted at 80°C, a temperature that balances reaction kinetics with thermal safety, preventing the decomposition of sensitive functional groups. Monitoring the reaction progress via Thin Layer Chromatography (TLC) is recommended to determine the optimal endpoint, usually achieved within 8 to 12 hours. Upon completion, the workup procedure is straightforward: the reaction mixture is filtered to remove the precipitated inorganic salts, and the filtrate is concentrated under reduced pressure. The resulting residue is then purified by silica gel column chromatography using a gradient of ethyl acetate and petroleum ether to isolate the pure thioamide derivative.
- Charge amine thiocarbonyl fluoride, malonate, and inorganic base (e.g., cesium carbonate) into a reaction flask with organic solvent.
- Heat the reaction mixture to 80°C and stir for 8-12 hours while monitoring progress via TLC.
- Filter off insoluble solids, concentrate the filtrate, and purify the residue using silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic methodology offers tangible strategic benefits that extend beyond mere technical feasibility. The shift towards this nucleophilic substitution route fundamentally alters the cost structure and risk profile of producing thioamide intermediates. By eliminating the need for specialized thionating reagents and transition metal catalysts, the process significantly reduces the dependency on volatile raw material markets and expensive supply chains. The use of commodity chemicals like malonates and inorganic bases ensures a stable and predictable supply of inputs, mitigating the risk of production delays caused by raw material shortages. Moreover, the simplified workup and purification steps reduce the consumption of solvents and stationary phases, leading to substantial cost savings in waste management and consumables. This efficiency gain is particularly relevant for companies aiming to optimize their cost reduction in pharmaceutical intermediates manufacturing initiatives without compromising on product quality.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts is a major driver for cost optimization in this process. Traditional methods often require palladium or copper catalysts, which are not only expensive to purchase but also necessitate rigorous removal steps to meet strict regulatory limits on residual metals in pharmaceutical ingredients. By avoiding these metals entirely, manufacturers save on both the initial catalyst cost and the downstream processing costs associated with metal scavenging resins and additional filtration steps. Additionally, the high atom economy of the reaction means that a larger proportion of the starting material mass is incorporated into the final product, reducing the effective cost per kilogram of the active intermediate. The use of recyclable solvents like acetonitrile further contributes to lower operating expenses, making the overall process economically superior to legacy methods.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route enhances supply chain resilience by simplifying the manufacturing workflow. Since the reaction tolerates a wide range of substrates and functional groups, the same production line can be easily adapted to synthesize various analogues within the N-alkyl/N-aryl thioamide family without requiring significant retooling or process re-validation. This flexibility allows suppliers to respond quickly to changing customer demands and scale up production of specific derivatives as needed. The moderate reaction temperature of 80°C is easily achievable with standard heating systems found in most multipurpose chemical plants, removing the need for specialized cryogenic or high-pressure equipment. This accessibility ensures that production can be distributed across multiple manufacturing sites, reducing the risk of supply disruption due to localized issues.
- Scalability and Environmental Compliance: From an environmental and regulatory standpoint, this method aligns perfectly with modern green chemistry principles. The generation of benign inorganic fluoride salts as the primary byproduct simplifies waste treatment compared to the phosphorus or sulfur waste generated by traditional thionation methods. This reduction in hazardous waste volume lowers disposal costs and minimizes the environmental footprint of the manufacturing process. The absence of toxic reagents also improves workplace safety, reducing the need for extensive personal protective equipment and specialized containment facilities. These factors collectively facilitate smoother regulatory approvals and audits, ensuring uninterrupted supply continuity. The process is inherently scalable, having been demonstrated to work efficiently from gram to multi-kilogram scales, providing a clear path for commercial scale-up of complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of these thioamide derivatives. The answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing a reliable reference for stakeholders evaluating this technology. Understanding these details is essential for assessing the feasibility of integrating this route into existing production pipelines or R&D programs.
Q: What are the key advantages of this thioamide synthesis method?
A: The method utilizes easily prepared raw materials and green inorganic bases, achieving good to excellent yields through a simple nucleophilic substitution reaction that is atom-economic and step-economic.
Q: What substrates are compatible with this reaction?
A: The process demonstrates a wide substrate scope, accommodating various R1 groups including hydrogen, alkyl, phenyl, halogens, and esters, as well as diverse N-alkyl and N-aryl substitutions.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the reaction operates at a moderate temperature of 80°C using common solvents like acetonitrile, making it highly scalable and safe for commercial production without requiring extreme conditions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Alkyl/N-Aryl Thioamide Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthetic methods described in CN111056889A for the next generation of therapeutic agents. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate these innovative laboratory protocols into robust, GMP-compliant manufacturing processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can move seamlessly from clinical trials to market launch. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of N-alkyl/N-aryl thioamide derivatives meets the highest quality standards required by global regulatory agencies.
We invite you to collaborate with us to leverage this advanced chemistry for your drug development programs. Our technical sales team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. Please contact our technical procurement team today to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your custom synthesis projects. Let us help you accelerate your pipeline with reliable, high-quality chemical solutions.
