Advanced Synthesis of N-Alkyl/N-Aryl Thioamide Derivatives for Commercial Pharmaceutical Manufacturing
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies for constructing sulfur-containing heterocycles and functional groups, particularly thioamides, due to their profound biological relevance. Patent CN111056889B introduces a groundbreaking synthetic strategy for N-alkyl/N-aryl thioamide derivatives, addressing critical gaps in efficiency and environmental sustainability. This technology leverages a nucleophilic substitution reaction between amine thiocarbonyl fluoride and malonate esters, catalyzed by inorganic bases under mild thermal conditions. The resulting compounds, characterized by the general structure shown below, exhibit high stability and serve as pivotal intermediates for anti-inflammatory, antibacterial, and anti-tuberculosis agents.
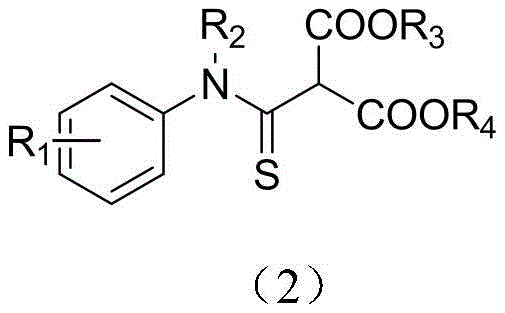
Thioamides are recognized for their superior chemical activity compared to traditional amides, enabling a broader spectrum of downstream conversion reactions. The innovation detailed in this patent provides a reliable pharmaceutical intermediate supplier with a pathway to access these high-value scaffolds through a green, atom-economic process. By utilizing easily accessible starting materials and avoiding complex catalytic systems, this method significantly lowers the barrier for entry into the production of specialized thioamide-based therapeutics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of thioamide derivatives has been plagued by significant operational challenges that hinder cost reduction in API manufacturing. Traditional routes often rely on the use of hazardous hydrogen sulfide gas or toxic thiophosphoryl chloride, posing severe safety risks and requiring specialized containment infrastructure. Furthermore, conventional methods frequently suffer from poor atom economy, generating substantial amounts of waste salts and by-products that complicate purification protocols. Many existing processes require cryogenic temperatures or prolonged reaction times, leading to high energy consumption and reduced throughput in commercial settings. The reliance on expensive transition metal catalysts in some modern variations further exacerbates cost issues, necessitating rigorous metal scavenging steps to meet stringent regulatory limits for residual impurities in final drug products.
The Novel Approach
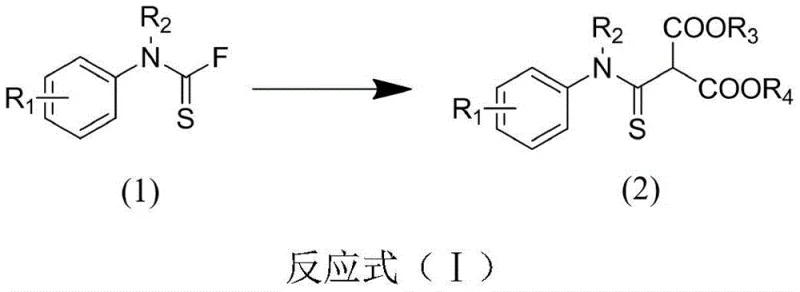
In stark contrast, the novel approach disclosed in CN111056889B utilizes amine thiocarbonyl fluoride as a stable and reactive electrophile, reacting seamlessly with malonate esters in the presence of benign inorganic bases. This method operates at a moderate temperature of 80°C, eliminating the need for energy-intensive cooling or heating cycles. The reaction proceeds with high efficiency, often achieving yields exceeding 80% across a diverse range of substrates, as demonstrated by the specific examples in the patent data. The use of acetonitrile as a preferred solvent ensures excellent solubility for reactants while maintaining a manageable boiling point for easy recovery. This streamlined protocol not only simplifies the operational workflow but also enhances the overall safety profile of the manufacturing process, making it an ideal candidate for the commercial scale-up of complex organic sulfides.
Mechanistic Insights into Base-Promoted Nucleophilic Substitution
The core of this synthetic breakthrough lies in the efficient activation of the methylene group in the malonate ester by the inorganic base. When cesium carbonate or potassium carbonate is introduced into the reaction mixture, it deprotonates the acidic alpha-hydrogen of the malonate, generating a highly nucleophilic enolate species. This enolate then attacks the electrophilic carbon of the thiocarbonyl fluoride moiety in a classic nucleophilic acyl substitution mechanism. The fluoride ion serves as an exceptional leaving group in this context, driven by the formation of strong bonds with the cationic species of the base, thereby driving the equilibrium towards the product formation. The reaction kinetics are optimized at 80°C, providing sufficient thermal energy to overcome the activation barrier without inducing thermal decomposition of the sensitive thioamide functionality.
Impurity control is inherently managed through the choice of reagents and the simplicity of the workup procedure. Since the reaction employs stoichiometric inorganic bases, the primary by-products are inorganic salts which precipitate out of the organic solvent or remain soluble in the aqueous phase during workup. This allows for a simple filtration step to remove the bulk of solid waste before concentration. The absence of transition metals eliminates the risk of metal-catalyzed side reactions such as homocoupling or over-reduction, which are common pitfalls in other thioamide syntheses. Consequently, the crude product obtained after concentration is of high purity, requiring only standard silica gel column chromatography to achieve pharmaceutical-grade specifications, thus ensuring a clean impurity profile for downstream applications.
How to Synthesize N-Alkyl/N-Aryl Thioamide Derivatives Efficiently
To implement this synthesis effectively, operators must adhere to precise molar ratios and reaction conditions to maximize yield and minimize side reactions. The patent specifies a molar ratio of amine thiocarbonyl fluoride to malonate of 1:1.5, ensuring that the electrophile is fully consumed while minimizing excess reagent waste. The base is used in slight excess, typically at a ratio of 1:2.5 relative to the fluoride substrate, to maintain a basic environment throughout the 8 to 12-hour reaction window. Monitoring via Thin Layer Chromatography (TLC) is critical to determine the exact endpoint, preventing over-reaction which could lead to hydrolysis of the ester groups. Following the reaction, the mixture is filtered to remove precipitated salts, and the filtrate is concentrated under reduced pressure to isolate the crude material.
- Charge amine thiocarbonyl fluoride, malonate, and inorganic base (preferably cesium carbonate) into a reaction flask with acetonitrile.
- Heat the reaction system to 80°C and maintain for 8 to 12 hours while monitoring progress via TLC.
- Filter to remove insoluble solids, concentrate the filtrate under reduced pressure, and purify the residue using silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers transformative benefits regarding cost structure and logistical reliability. The shift from hazardous gaseous reagents to stable solid or liquid precursors drastically simplifies raw material handling and storage requirements. This reduction in hazard classification translates directly into lower insurance premiums and reduced compliance costs associated with transporting dangerous goods. Furthermore, the high atom economy of the reaction means that a greater proportion of the input mass is converted into the desired product, reducing the volume of waste disposal needed and lowering the overall environmental footprint of the manufacturing site.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts removes a significant cost driver from the bill of materials. Additionally, the use of commodity inorganic bases like cesium carbonate or potassium carbonate ensures that reagent costs remain low and predictable. The simplified workup procedure, which avoids complex extraction or distillation steps, reduces labor hours and utility consumption per batch. By streamlining the purification process to a single column chromatography step, manufacturers can achieve substantial cost savings in both time and solvent usage compared to multi-step traditional methods.
- Enhanced Supply Chain Reliability: The starting materials, specifically malonate esters and substituted anilines, are widely available from multiple global suppliers, mitigating the risk of single-source dependency. The robustness of the reaction conditions allows for flexibility in sourcing, as the process tolerates minor variations in reagent quality without compromising yield. This resilience ensures consistent production schedules and reduces the likelihood of delays caused by raw material shortages. The ability to produce a wide array of derivatives from a common platform further strengthens supply security by allowing for rapid switching between products based on market demand.
- Scalability and Environmental Compliance: The process is inherently scalable, moving seamlessly from gram-scale laboratory optimization to multi-kilogram pilot runs. The use of acetonitrile, a solvent with well-established recovery and recycling protocols, supports sustainable manufacturing practices. The generation of primarily inorganic salt waste simplifies effluent treatment, ensuring compliance with increasingly strict environmental regulations. This green chemistry profile not only future-proofs the manufacturing asset but also aligns with the corporate sustainability goals of major pharmaceutical clients seeking eco-friendly supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this thioamide synthesis technology. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation. Understanding these details is crucial for R&D teams evaluating the feasibility of integrating this route into their existing pipelines.
Q: What are the primary advantages of this thioamide synthesis method?
A: The method described in CN111056889B offers significant advantages including atom economy, step economy, and environmental friendliness. It utilizes easily prepared raw materials and green inorganic bases to achieve good to excellent yields without requiring harsh conditions or toxic catalysts.
Q: What is the substrate scope for this nucleophilic substitution reaction?
A: The reaction demonstrates a wide substrate application range, accommodating various R1 groups such as hydrogen, alkyl, phenyl, acyl, ester, halogen, and alkoxy. It also tolerates diverse N-alkyl and N-aryl substitutions, making it highly versatile for generating libraries of bioactive compounds.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process is designed for scalability. The use of simple inorganic bases and standard organic solvents like acetonitrile, combined with a straightforward workup involving filtration and column chromatography, facilitates easy transition from laboratory to pilot and commercial scales.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Alkyl/N-Aryl Thioamide Derivatives Supplier
NINGBO INNO PHARMCHEM stands at the forefront of translating innovative patent technologies like CN111056889B into commercial reality. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project moves efficiently from concept to market. Our facilities are equipped with state-of-the-art rigorous QC labs capable of meeting stringent purity specifications required for pharmaceutical intermediates. We understand the critical nature of supply continuity and have established robust protocols to manage raw material inventory and production scheduling effectively.
We invite you to collaborate with us to leverage this advanced synthesis method for your specific therapeutic programs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements and quality targets. Please contact our technical procurement team today to request specific COA data for our thioamide portfolio and discuss route feasibility assessments for your custom synthesis needs. Together, we can accelerate the development of next-generation bioactive sulfur-containing compounds.
