Advanced Catalytic Synthesis of Pirfenidone: Scaling High-Purity Intermediates for Global Supply
Advanced Catalytic Synthesis of Pirfenidone: Scaling High-Purity Intermediates for Global Supply
The global demand for effective treatments for Idiopathic Pulmonary Fibrosis (IPF) has placed immense pressure on the supply chains of key active pharmaceutical ingredients, specifically pirfenidone. As the industry seeks more robust and economically viable production methods, patent CN101891676A emerges as a critical technological milestone, offering a novel synthetic pathway that fundamentally alters the cost and purity landscape of this vital intermediate. This patent details a sophisticated approach where bromobenzene serves a dual function as both a reactant and the reaction solvent, catalyzed efficiently by cuprous bromide. For R&D directors and procurement strategists, this represents a shift away from traditional, solvent-heavy processes toward a more concentrated, atom-economical methodology that promises enhanced scalability and reduced environmental footprint without compromising the stringent purity specifications required for clinical applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
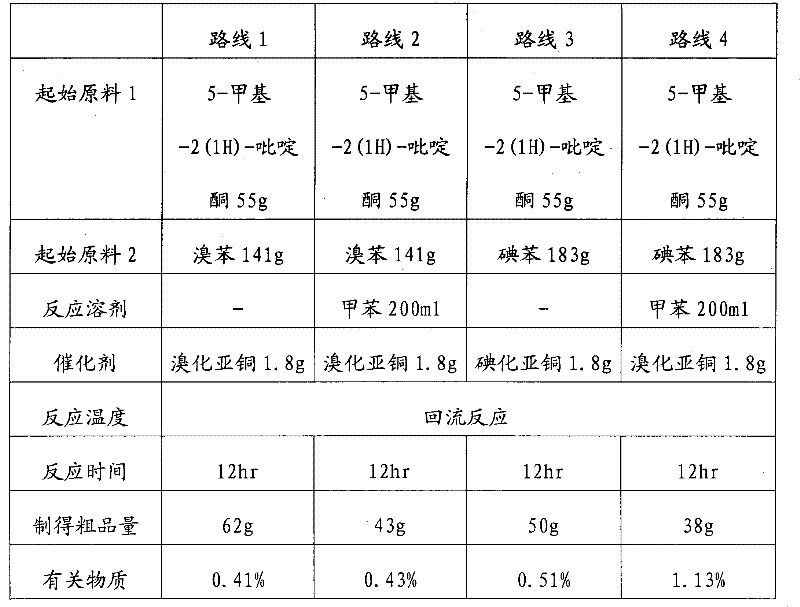
Historically, the synthesis of 5-methyl-1-phenyl-2-(1H)-pyridone has relied heavily on the use of iodobenzene as the arylating agent, often necessitating the addition of auxiliary organic solvents such as toluene to facilitate the reaction kinetics. These conventional routes suffer from inherent economic and operational inefficiencies; iodobenzene is significantly more expensive than its brominated counterpart, driving up raw material costs substantially. Furthermore, the reliance on additional solvents introduces complex downstream processing requirements, including energy-intensive distillation for solvent recovery and increased risks of residual solvent contamination in the final API. The use of alternative catalysts like cuprous iodide in these traditional setups has also been shown to yield lower conversion rates and higher levels of related impurities, complicating the purification process and reducing overall throughput in large-scale manufacturing environments.
The Novel Approach
The innovative methodology described in the patent data revolutionizes this landscape by leveraging bromobenzene not merely as a reagent but as the exclusive reaction medium, thereby creating a high-concentration system that maximizes collision frequency between reactants. By switching the catalyst system to cuprous bromide, the process achieves a remarkable synergy that enhances both yield and selectivity. Experimental data indicates that this specific combination outperforms systems using toluene or iodobenzene, delivering superior crude yields and minimizing the formation of difficult-to-remove byproducts. This approach effectively streamlines the workflow by eliminating the need for external solvent management, allowing for a direct transition from reaction completion to aqueous workup, which is a significant advancement for industrial hygiene and operational simplicity.
Mechanistic Insights into Copper-Catalyzed N-Arylation
At the heart of this synthesis lies a copper-catalyzed N-arylation mechanism, likely proceeding through an oxidative addition-reductive elimination cycle typical of Ullmann-type condensations. The choice of cuprous bromide is pivotal; it appears to offer an optimal balance of solubility and catalytic activity within the bromobenzene medium, facilitating the formation of the carbon-nitrogen bond between the pyridone nitrogen and the phenyl ring. The presence of anhydrous potassium carbonate acts as a base to deprotonate the pyridone, generating the nucleophilic species necessary for the attack on the copper-activated aryl halide complex. This mechanistic pathway is highly sensitive to the halogen identity, where the bromine atom provides a superior leaving group capability in this specific catalytic environment compared to the conditions observed with iodine counterparts in the presence of competing solvents.
Impurity control is another critical aspect where this mechanism excels, particularly regarding the minimization of homocoupling byproducts and unreacted starting materials. The high selectivity observed suggests that the catalytic cycle favors the cross-coupling reaction overwhelmingly, preventing the formation of biphenyl derivatives or other oligomeric impurities that often plague copper-catalyzed reactions. Furthermore, the subsequent purification strategy exploits the physicochemical properties of the product, utilizing water as a crystallization solvent. Since the organic impurities and catalyst residues remain soluble or are removed during the acid-base extraction phases, the final crystallization from water ensures that the resulting solid possesses exceptionally low levels of residual organic solvents, meeting the rigorous standards demanded by regulatory bodies for pharmaceutical intermediates.
How to Synthesize 5-Methyl-1-phenyl-2-(1H)-pyridone Efficiently
Implementing this synthesis requires precise control over reaction parameters to replicate the high yields reported in the patent literature. The process begins with the careful charging of stoichiometric amounts of 5-methyl-2(1H)-pyridone and a significant excess of bromobenzene, which ensures the reaction proceeds to completion while maintaining a fluid reaction mass. The addition of the cuprous bromide catalyst and potassium carbonate base must be managed to prevent localized overheating before the mixture is brought to a sustained reflux. Maintaining the temperature at the boiling point of bromobenzene for a duration of approximately 12 hours is critical to drive the equilibrium toward the product. Following the reaction, the recovery of unreacted bromobenzene under reduced pressure allows for material recycling, enhancing the overall atom economy of the process before the crude product is isolated via aqueous quenching and filtration.
- Charge 5-methyl-2(1H)-pyridone, excess bromobenzene (acting as solvent), cuprous bromide catalyst, and anhydrous potassium carbonate into a reactor.
- Heat the mixture to reflux temperature and maintain stirring for approximately 12 hours to ensure complete conversion.
- Recover excess bromobenzene under reduced pressure, add distilled water for crystallization, filter, and purify via acid-base extraction and recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic route offers compelling strategic advantages that extend far beyond simple laboratory yield improvements. The fundamental shift from iodobenzene to bromobenzene as the primary arylating agent represents a direct and substantial reduction in raw material expenditure, as brominated aromatics are generally more abundant and cost-effective on the global chemical market. Additionally, the elimination of auxiliary solvents like toluene removes an entire layer of logistical complexity, including the costs associated with solvent purchasing, storage, safety compliance, and the energy-intensive recovery processes typically required to meet environmental discharge limits. This simplification of the bill of materials directly translates to a more resilient and cost-efficient supply chain structure.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the dual role of bromobenzene, which negates the need for purchasing separate reaction solvents, thereby reducing the total volume of chemicals required per kilogram of product. By removing the dependency on expensive iodine-based reagents and auxiliary organic solvents, manufacturers can achieve a drastic simplification of the cost structure. The ability to recover and recycle the excess bromobenzene further amplifies these savings, creating a closed-loop system that minimizes waste disposal fees and maximizes resource utilization efficiency throughout the production lifecycle.
- Enhanced Supply Chain Reliability: Sourcing bromobenzene is inherently more stable and less prone to market volatility compared to specialized iodinated compounds, ensuring a consistent flow of raw materials for continuous production campaigns. The robustness of the reaction conditions, which tolerate the high concentrations inherent in a solvent-free design, reduces the risk of batch failures due to solvent quality variations or moisture sensitivity often associated with complex solvent mixtures. This reliability allows supply chain planners to forecast production timelines with greater accuracy, reducing the need for excessive safety stock and enabling a more agile response to fluctuations in market demand for pirfenidone.
- Scalability and Environmental Compliance: From an environmental perspective, the replacement of organic crystallization solvents with water significantly lowers the facility's VOC emissions profile, aligning production with increasingly stringent global environmental regulations. The simplified workup procedure, which relies on filtration and aqueous washing rather than complex chromatographic separations or multi-step extractions, facilitates easier scale-up from pilot plant to commercial tonnage scales. This green chemistry approach not only mitigates regulatory risk but also enhances the corporate sustainability profile, a factor that is becoming increasingly important for partnerships with major multinational pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following technical inquiries address common concerns regarding the implementation and optimization of this specific catalytic system. These answers are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on why this method is superior to legacy technologies. Understanding these nuances is essential for technical teams evaluating the feasibility of technology transfer and for procurement specialists assessing the long-term viability of this supply route.
Q: Why is cuprous bromide preferred over cuprous iodide in this specific synthesis?
A: According to patent data, using cuprous bromide in conjunction with bromobenzene as a solvent yields significantly higher crude product mass (62g vs 50g) and lower related substance impurities (0.41% vs 0.51%) compared to cuprous iodide systems.
Q: How does this method improve environmental compliance compared to traditional routes?
A: The process eliminates the need for additional organic solvents like toluene by using the reactant bromobenzene as the solvent medium. Furthermore, it utilizes water for crystallization and recrystallization, drastically reducing volatile organic compound (VOC) emissions and hazardous waste generation.
Q: What represents the primary cost advantage of this manufacturing route?
A: The primary economic driver is the substitution of expensive iodobenzene with cheaper bromobenzene, coupled with the elimination of solvent recovery costs for auxiliary solvents like toluene, leading to substantial operational expenditure reductions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pirfenidone Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of life-saving therapies like those for IPF depends on a partner who can bridge the gap between innovative patent chemistry and reliable industrial reality. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of the cuprous bromide-catalyzed route are fully realized in a GMP-compliant environment. We maintain stringent purity specifications and operate rigorous QC labs equipped to detect and quantify trace impurities, guaranteeing that every batch of 5-methyl-1-phenyl-2-(1H)-pyridone meets the exacting standards required for downstream API synthesis.
We invite you to engage with our technical procurement team to discuss how this optimized synthetic route can be integrated into your supply chain to drive efficiency and value. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the specific economic impact of switching to this bromobenzene-based methodology. We encourage potential partners to contact us directly to obtain specific COA data and comprehensive route feasibility assessments, allowing you to make informed decisions that secure your supply of high-purity pharmaceutical intermediates for the future.
