Optimizing Pyronaridine Production: A Technical Breakthrough in API Intermediate Synthesis
Optimizing Pyronaridine Production: A Technical Breakthrough in API Intermediate Synthesis
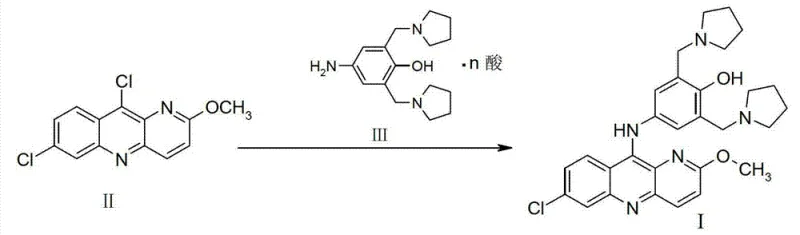
The global demand for effective antimalarial agents continues to drive innovation in pharmaceutical intermediate manufacturing, particularly for complex heterocyclic compounds like pyronaridine. Patent CN103373995A introduces a transformative methodology for preparing high-purity pyronaridine (Formula I) that addresses long-standing challenges in yield and impurity control. This technical insight report analyzes the strategic advantages of this novel condensation route, which utilizes a specific acid salt of 4-amino-2-bis(1-pyrrolemethyl)phenol reacting with 7,10-dichloro-2-methoxypyrido[3,2-b]quinoline. By rigorously controlling the water content of the reaction system to ≤3%, manufacturers can achieve molar yields exceeding 80% and product purity greater than 95%. This represents a significant leap forward from historical benchmarks, offering a robust pathway for the commercial scale-up of complex pharmaceutical intermediates required for next-generation antimalarial therapies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historical synthesis routes for pyronaridine, such as those reported in the late 1970s, suffer from excessive step counts and inefficient overall yields that render them economically unviable for modern large-scale production. The traditional approach typically involves a five-step sequence starting from 2-aminopyridine to generate key intermediates, followed by coupling and cyclization steps that accumulate significant material loss. For instance, earlier documented processes achieved a total yield of merely 6.1%, with the final two steps contributing a meager 40.8% efficiency. Furthermore, subsequent modifications, while attempting to streamline the preparation of the chloro-intermediate (Formula II), failed to address the fundamental bottlenecks in the final coupling stage. These legacy methods often rely on harsh conditions or multiple purification stages that increase operational complexity and waste generation, making them unsuitable for a reliable pyronaridine intermediate supplier aiming for cost-effective manufacturing.
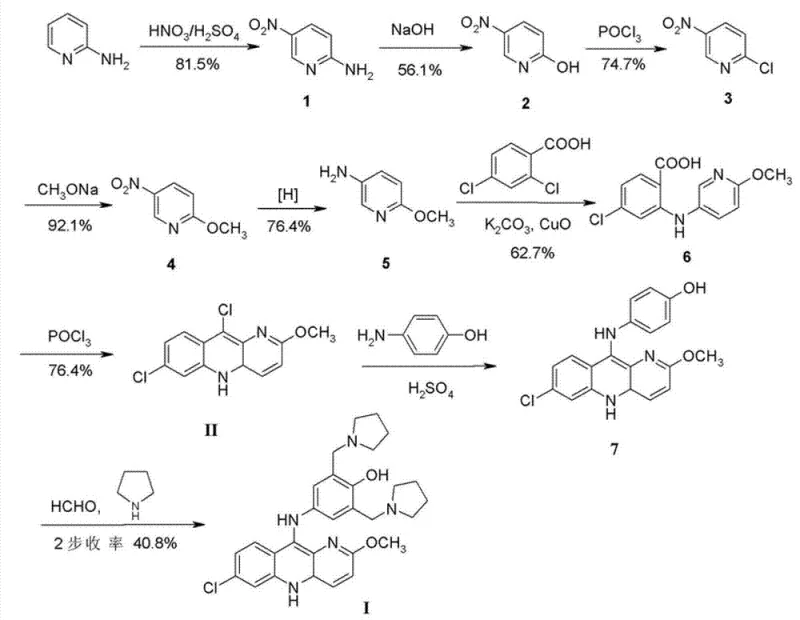
The Novel Approach
The innovative strategy disclosed in the patent fundamentally reengineers the final coupling step by utilizing the acid salt of the amine component rather than the free base. This modification allows the reaction to proceed in an inert solvent, such as methanol or ethanol, under mild thermal conditions ranging from 40°C to 120°C. Crucially, the acid salt serves a dual purpose: it acts as the nucleophile source and provides the necessary acidic environment for catalysis without requiring external acid additives that could introduce moisture. This self-catalytic mechanism simplifies the reaction setup and significantly enhances the kinetics of the nucleophilic aromatic substitution. By bypassing the need for separate acidification steps and minimizing water introduction, this approach drastically reduces the formation of hydrolysis byproducts, thereby facilitating cost reduction in antimalarial API manufacturing through improved material throughput.
Mechanistic Insights into Acid Salt-Mediated Nucleophilic Substitution
The core chemical transformation involves the displacement of a chlorine atom on the electron-deficient pyrido[3,2-b]quinoline ring (Formula II) by the amino group of the phenol derivative (Formula III). In conventional free-base reactions, the nucleophilicity of the amine can be inconsistent, and the addition of strong acids to activate the leaving group often leads to protonation of the amine, rendering it non-nucleophilic. The use of the acid salt in this patented method creates a buffered equilibrium where a sufficient concentration of free amine exists to attack the electrophilic carbon, while the conjugate acid maintains the solubility and stability of the intermediate species. The reaction is highly sensitive to the presence of water, which competes as a nucleophile and promotes the hydrolysis of the chloro-intermediate. Maintaining the system water content at ≤3% is therefore not merely a preference but a mechanistic necessity to ensure the chloride leaving group is displaced exclusively by the amine rather than hydroxide ions.
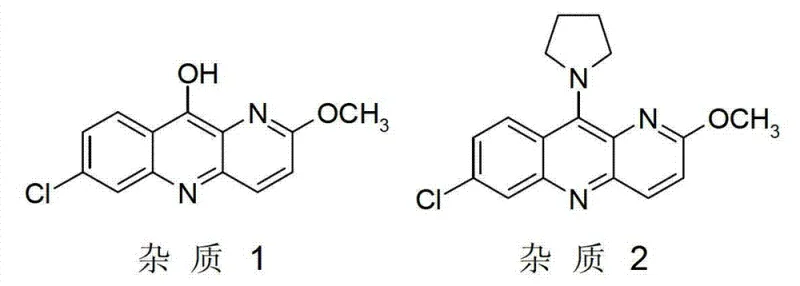
Impurity control is another critical aspect of this mechanism, specifically regarding the suppression of two major degradation products known as Impurity 1 and Impurity 2. Impurity 1 arises from the hydrolysis of the chloro-group to a hydroxy-group, while Impurity 2 results from the substitution of the pyrrolidine ring nitrogen instead of the aniline nitrogen. The strict control of water content directly mitigates the formation of Impurity 1 by limiting the availability of hydroxide ions. Simultaneously, the specific steric and electronic environment provided by the acid salt interaction favors the attack of the primary aniline nitrogen over the secondary pyrrolidine nitrogen, keeping the combined impurity profile below 2%. This level of precision is essential for producing high-purity pyronaridine that meets stringent regulatory standards for pharmaceutical active ingredients.
How to Synthesize Pyronaridine Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for transitioning from laboratory scale to industrial production. The process begins with the preparation of the acid salt of Formula III, ensuring it is dried to a water content of less than 3%, preferably below 1%. This dried salt is then suspended in an anhydrous alcohol solvent, such as absolute ethanol or methanol. Formula II is added to the mixture, and the temperature is carefully ramped to between 45°C and 80°C to initiate the coupling. The reaction progress is monitored to ensure complete consumption of the starting materials, typically within 2 to 5 hours. Following the reaction, the product is isolated through standard workup procedures involving pH adjustment and extraction, followed by recrystallization to achieve the final purity specifications. Detailed standardized synthesis steps are provided in the guide below.
- Prepare the acid salt of 4-amino-2-bis(1-pyrrolemethyl)phenol (Formula III) ensuring water content is strictly controlled below 3%.
- React Formula III acid salt with 7,10-dichloro-2-methoxypyrido[3,2-b]quinoline (Formula II) in an inert C1-C6 alcohol solvent at 40-120°C.
- Maintain the reaction system water content at ≤3% to maximize yield above 80% and minimize specific impurities.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology offers tangible benefits that extend beyond simple chemical yield. The reduction in synthetic steps and the elimination of auxiliary catalysts translate directly into lower raw material consumption and reduced waste disposal costs. By simplifying the process flow, manufacturers can decrease the overall production cycle time, allowing for faster response to market demands and improved inventory turnover. The robustness of the reaction conditions also means that the process is less susceptible to batch-to-batch variability, ensuring a consistent supply of quality intermediates. This reliability is paramount for maintaining uninterrupted production schedules in the pharmaceutical sector, where delays can have significant downstream impacts on drug availability.
- Cost Reduction in Manufacturing: The streamlined nature of this synthesis eliminates the need for expensive transition metal catalysts and complex purification sequences associated with older routes. By utilizing the acid salt as a self-catalyst, the process removes the cost burden of additional reagents and the subsequent neutralization steps. Furthermore, the significant improvement in yield means that less starting material is required to produce the same amount of final product, effectively lowering the cost of goods sold. The ability to use common solvents like ethanol and methanol further contributes to economic efficiency, as these are readily available and inexpensive compared to specialized organic solvents.
- Enhanced Supply Chain Reliability: The intermediates required for this process, such as the chloro-quinoline derivative and the aminophenol salt, are synthesized via robust and scalable pathways that do not rely on scarce or volatile raw materials. This ensures a stable supply chain that is resilient to market fluctuations. The simplified process also reduces the dependency on specialized equipment or extreme operating conditions, making it easier to qualify multiple manufacturing sites. This flexibility allows for diversified sourcing strategies, mitigating the risk of supply disruptions and ensuring continuous availability of critical antimalarial intermediates for global health initiatives.
- Scalability and Environmental Compliance: The mild reaction temperatures and the use of alcohol solvents make this process highly amenable to scale-up in standard stainless steel reactors without requiring exotic metallurgy. The reduction in waste generation, particularly from avoided heavy metal catalysts and reduced solvent usage for purification, aligns with increasingly stringent environmental regulations. This green chemistry profile not only reduces compliance costs but also enhances the corporate sustainability image of the manufacturer. The high purity of the crude product minimizes the need for extensive recrystallization, further reducing solvent waste and energy consumption during the drying phases.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation of this synthesis method. They are derived from the specific experimental data and claims found within the patent documentation, providing clarity on critical process parameters. Understanding these nuances is vital for R&D teams planning to integrate this technology into their existing manufacturing frameworks. The answers highlight the importance of moisture control and the specific benefits of the acid salt formulation in achieving superior product quality.
Q: Why is controlling water content critical in pyronaridine synthesis?
A: According to patent CN103373995A, maintaining water content below 3% in the reaction system significantly increases yield (over 80%) and purity (≥95%), while suppressing the formation of hydroxy and pyrrolidine-substituted impurities.
Q: How does the acid salt method improve upon conventional routes?
A: The acid salt of the amine intermediate acts as a self-catalyst, eliminating the need for additional acid catalysts that often introduce moisture. This shortens the synthetic route and avoids the low yields (e.g., 6.1% total yield) associated with older multi-step methods.
Q: What are the primary impurities controlled in this process?
A: The process specifically targets the reduction of Impurity 1 (hydroxy derivative) and Impurity 2 (pyrrolidine substitution product), keeping their combined content below 2%, which is crucial for meeting pharmaceutical grade specifications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyronaridine Supplier
As the pharmaceutical industry evolves, the need for partners who can deliver both technical excellence and commercial reliability has never been greater. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, leveraging advanced synthetic methodologies like the one described in CN103373995A to produce superior antimalarial intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. We operate stringent purity specifications and maintain rigorous QC labs to guarantee that every batch of pyronaridine intermediate meets the highest international standards, ready for immediate conversion into the final active pharmaceutical ingredient.
We invite you to collaborate with us to optimize your supply chain and reduce your manufacturing costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to innovation can drive value for your organization. Together, we can ensure the reliable delivery of life-saving medications to patients worldwide.
