Advanced Manufacturing of High-Purity Pyronaridine Intermediates for Global Pharmaceutical Supply Chains
Introduction to Advanced Pyronaridine Manufacturing
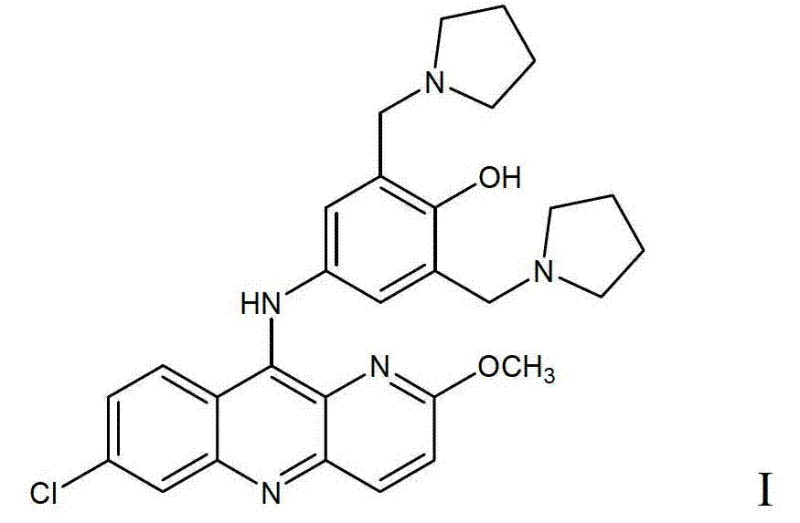
The global demand for effective antimalarial therapies continues to drive innovation in the synthesis of key active pharmaceutical ingredients (APIs). Patent CN103373995A introduces a transformative methodology for the preparation of Pyronaridine, also known as Malaridine, a potent benzonaphthyridine derivative widely used in treating severe malaria including cerebral malaria. This technical disclosure addresses long-standing inefficiencies in legacy manufacturing processes by proposing a streamlined coupling reaction that significantly enhances both yield and purity. By leveraging a novel acid salt intermediate strategy, the invention enables the production of high-purity Pyronaridine (Formula I) with impurity profiles strictly controlled below 2%, meeting the rigorous standards required by international regulatory bodies for essential medicines.
The significance of this technological advancement extends beyond mere academic interest; it represents a pivotal shift towards more sustainable and cost-effective pharmaceutical manufacturing. For R&D directors and procurement specialists, understanding the nuances of this synthesis route is critical for securing a reliable supply chain of high-quality API intermediates. The method specifically targets the reduction of toxic byproducts and the optimization of reaction kinetics, ensuring that the final product not only meets therapeutic efficacy requirements but also adheres to stringent safety protocols regarding residual impurities.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
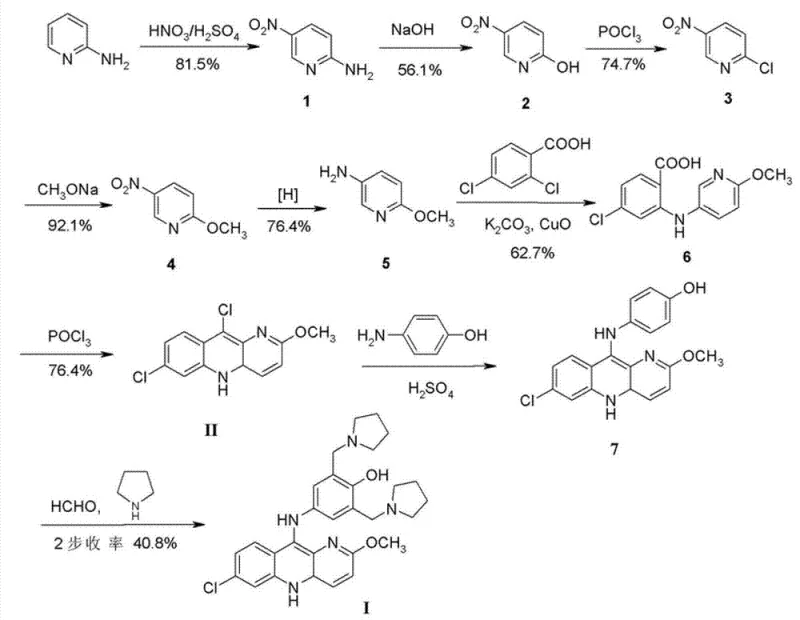
Historically, the industrial synthesis of Pyronaridine has been plagued by cumbersome multi-step sequences that result in prohibitively low overall yields. Early reports from 1979 detailed a route starting from 2-aminopyridine, involving nitration, hydrolysis, chlorination, methoxidation, and catalytic reduction just to reach a key intermediate. This lengthy pathway culminated in a total yield of merely 6.1%, with the final two steps achieving only 40.8% efficiency. Such inefficiency translates directly into excessive waste generation, high solvent consumption, and escalated production costs, rendering the process economically unviable for modern large-scale operations. Furthermore, subsequent modifications, such as those described in KR2009114029, failed to substantially improve the overall yield, which remained stagnant around 41.8% for the final stages, thereby perpetuating the high cost of goods sold.
The Novel Approach
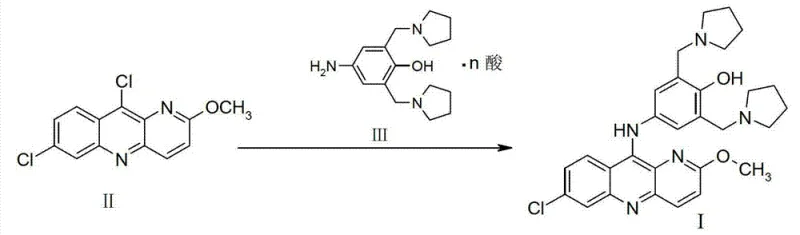
In stark contrast, the methodology disclosed in CN103373995A revolutionizes the final assembly of the Pyronaridine molecule through a direct nucleophilic aromatic substitution. The core innovation lies in the reaction between 7,10-dichloro-2-methoxypyrido[3,2-b]quinoline (Formula II) and the acid salt of 4-amino-2-bis(1-pyrrolemethyl)phenol (Formula III). This convergent strategy drastically shortens the synthetic timeline by eliminating unnecessary protection and deprotection steps inherent in older linear syntheses. By conducting this coupling in an inert solvent under strictly controlled anhydrous conditions, the process achieves a molar yield exceeding 80% based on Formula II. This dramatic improvement in efficiency not only reduces the environmental footprint but also significantly lowers the barrier to entry for cost reduction in antimalarial drug manufacturing, offering a robust alternative for generic API producers.
Mechanistic Insights into Acid Salt Catalyzed Coupling
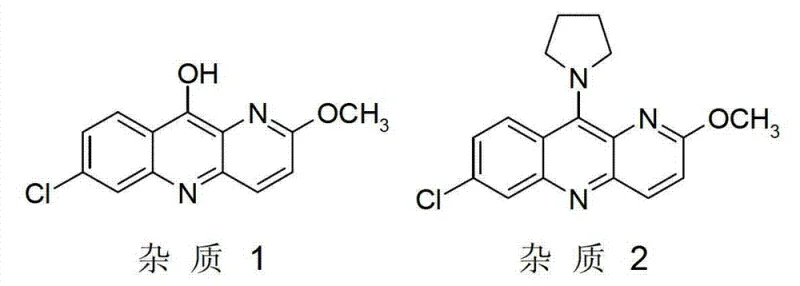
The success of this novel synthesis hinges on a sophisticated understanding of reaction kinetics and moisture sensitivity. The use of the acid salt of Formula III serves a dual purpose: it acts as the nucleophile and simultaneously provides the acidic environment necessary to catalyze the substitution reaction without introducing external aqueous acids. Traditional methods often relied on sulfuric acid catalysis, which inevitably introduced water into the system. This presence of water is detrimental, as it promotes hydrolysis side reactions that lead to the formation of specific degradation products known as Impurity 1 and Impurity 2. The patent explicitly identifies that maintaining the water content of the reaction system at ≤3%, and preferably ≤0.5%, is the critical threshold for suppressing these pathways. This precise control ensures that the nucleophilic attack of the amine on the chloro-substituted quinoline ring proceeds with high selectivity.
Furthermore, the mechanistic advantage extends to the purification profile. By avoiding aqueous workups during the critical coupling phase, the formation of hydroxyl-substituted byproducts is minimized. The structural integrity of the pyrrolidine methyl groups is preserved, preventing the cleavage that typically generates Impurity 1. Similarly, the controlled acidity prevents over-reaction or polymerization that could lead to Impurity 2. This level of impurity control is paramount for R&D teams focused on regulatory filing, as it simplifies the downstream purification process. The ability to consistently achieve a product purity of ≥95% with total impurities below 2% demonstrates the robustness of this acid salt-mediated mechanism, providing a reliable framework for scaling complex pharmaceutical intermediates without compromising on quality specifications.
How to Synthesize Pyronaridine Efficiently
The practical implementation of this synthesis requires strict adherence to anhydrous protocols and precise temperature control to maximize yield and purity. The process begins with the preparation of the dry acid salt of Formula III, which involves a Mannich reaction followed by hydrogenation and subsequent salt formation with acids like HCl or acetic acid. This intermediate must be dried to a water content of less than 3% before being introduced to the reaction vessel containing Formula II. The coupling is then performed in a C1-C6 alcohol solvent, such as anhydrous ethanol or methanol, at temperatures ranging from 40°C to 120°C. Detailed operational parameters, including specific molar ratios and workup procedures, are essential for reproducibility and are outlined in the comprehensive guide below.
- Preparation of Formula III Acid Salt: React p-nitrophenol with formaldehyde and pyrrolidine, followed by acidification and hydrogenation to obtain the dry acid salt of 4-amino-2-bis(1-pyrrolemethyl)phenol with water content ≤3%.
- Coupling Reaction: In an inert anhydrous solvent (e.g., absolute ethanol), react Formula II compound with the Formula III acid salt at 40-120°C for 1-10 hours, ensuring the system water content remains below 3%.
- Purification: Isolate the crude product, adjust pH to neutral, extract, and recrystallize from anhydrous ethanol to achieve purity ≥95% with impurity levels controlled below 2%.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology offers substantial strategic benefits that extend far beyond simple yield metrics. The primary advantage lies in the drastic simplification of the manufacturing workflow, which inherently reduces the operational complexity and the associated overhead costs. By shortening the synthetic route and eliminating low-yielding steps, manufacturers can significantly reduce the consumption of raw materials and solvents. This efficiency gain translates directly into a more competitive pricing structure for the final API, allowing buyers to negotiate better terms and secure a more stable supply of this critical antimalarial medication. The elimination of expensive transition metal catalysts in certain steps further contributes to cost reduction in pharmaceutical intermediate manufacturing.
- Cost Reduction in Manufacturing: The shift from a multi-step linear synthesis with a 6.1% total yield to a convergent route with over 80% yield in the final step represents a massive improvement in material efficiency. This enhancement means that less starting material is required to produce the same amount of finished product, effectively lowering the cost of goods sold. Additionally, the removal of aqueous acid catalysts reduces the burden on wastewater treatment facilities, leading to significant savings in environmental compliance and waste disposal costs. The process operates under mild conditions, reducing energy consumption for heating and cooling, which further optimizes the overall production budget without compromising output quality.
- Enhanced Supply Chain Reliability: The robustness of this synthesis method ensures a more predictable and consistent production schedule. High yields and fewer processing steps mean shorter cycle times, allowing manufacturers to respond more rapidly to fluctuations in market demand. The use of readily available reagents such as formaldehyde, pyrrolidine, and common alcohols minimizes the risk of supply bottlenecks associated with exotic or specialized chemicals. This reliability is crucial for maintaining continuous inventory levels of life-saving antimalarial drugs, ensuring that healthcare providers have uninterrupted access to essential medications during peak malaria seasons or outbreak scenarios.
- Scalability and Environmental Compliance: The process is explicitly designed for industrial scale-up, utilizing standard reactor equipment and common solvents that are easy to recover and recycle. The strict control of water content and the avoidance of heavy metal catalysts simplify the purification process, resulting in a cleaner product that requires less intensive downstream processing. This aligns with modern green chemistry principles, reducing the generation of hazardous waste and lowering the environmental impact of production. For supply chain leaders, this means easier regulatory approval and a lower risk of production halts due to environmental non-compliance, ensuring long-term sustainability of the supply source.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced synthesis route. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing clarity on the feasibility and benefits of adopting this technology for commercial production. Understanding these details is essential for stakeholders evaluating the potential integration of this method into their existing manufacturing portfolios.
Q: How does the new synthesis method improve yield compared to traditional routes?
A: Traditional routes reported yields as low as 6.1% over multiple steps. The novel method described in patent CN103373995A achieves a molar yield exceeding 80% based on Formula II by utilizing a direct coupling reaction with an acid salt intermediate under controlled anhydrous conditions.
Q: What are the critical parameters for controlling impurities in Pyronaridine synthesis?
A: The most critical parameter is the water content of the reaction system, which must be maintained at ≤3% (preferably ≤0.5%). Additionally, using the acid salt of Formula III acts as a self-catalyst, eliminating the need for external aqueous acid catalysts that typically generate Impurity 1 and Impurity 2.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is designed for industrial scalability. It features mild reaction conditions (45-80°C), uses common solvents like methanol or ethanol, and significantly shortens the synthetic route, making it economically viable for commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyronaridine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the global fight against malaria. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated moisture-control techniques required for this synthesis are executed with precision. We are committed to delivering Pyronaridine and its precursors with stringent purity specifications, utilizing our rigorous QC labs to verify that impurity levels remain well below the 2% threshold mandated by modern pharmacopeias. Our facility is equipped to handle the anhydrous conditions and specific acid salt preparations necessary to replicate the high yields described in the patent.
We invite global pharmaceutical partners to collaborate with us to optimize their supply chains for antimalarial medications. By leveraging our expertise in process chemistry, we can offer a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments, ensuring that your next project benefits from the latest advancements in efficient and sustainable API manufacturing.
