Optimizing Liraglutide Manufacturing via Advanced Two-Fragment Solid-Phase Synthesis Strategies
Optimizing Liraglutide Manufacturing via Advanced Two-Fragment Solid-Phase Synthesis Strategies
The pharmaceutical industry continuously seeks robust manufacturing routes for complex peptide therapeutics like Liraglutide, a glucagon-like peptide-1 (GLP-1) receptor agonist critical for diabetes management. Patent CN110894227A introduces a refined solid-phase synthesis methodology that strategically divides the 31-amino acid sequence into two manageable fragments to overcome historical bottlenecks in yield and purity. This technical disclosure represents a significant evolution from traditional stepwise elongation, addressing the cumulative risk of deletion sequences and racemization that plagues long-chain peptide assembly. By implementing a convergent strategy where a resin-bound N-terminal fragment is coupled with a solution-phase C-terminal fragment, the process minimizes the formation of difficult-to-remove impurities. For R&D directors and process chemists, this approach offers a viable pathway to scale production while maintaining stringent quality control standards required for regulatory approval in major markets.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Liraglutide has relied heavily on either linear stepwise solid-phase synthesis or solution-phase fragment condensation, both of which present distinct scalability challenges. In stepwise synthesis, as the peptide chain extends beyond twenty residues, the efficiency of each coupling step diminishes, leading to a statistical accumulation of deletion peptides that are structurally similar to the target molecule. These deletion impurities are notoriously difficult to separate via chromatography due to their minimal polarity differences, often requiring multiple purification cycles that drastically reduce overall yield. Furthermore, traditional fragment condensation methods often involve isolating intermediate peptide segments that exist as viscous oils rather than crystalline solids. These oily intermediates complicate weighing, transfer, and reaction monitoring, introducing significant variability in stoichiometry and increasing the risk of batch failure during the critical final ligation step.
The Novel Approach
The methodology outlined in the patent data circumvents these issues by employing a hybrid solid-phase strategy that leverages the advantages of both resin-bound stability and solution-phase flexibility. Instead of attempting to synthesize the entire sequence on a single resin or condensing fully deprotected fragments in solution, the process synthesizes the 1-20 residue fragment on Wang resin while keeping the Lysine-12 side chain protected with an Alloc group. Simultaneously, the 21-31 residue fragment is synthesized on a separate 2-CTC resin and cleaved under mild acidic conditions that preserve side-chain protecting groups. This ensures that the final coupling occurs between a solid-supported amine and a protected carboxylic acid fragment in solution, effectively preventing the aggregation and solubility issues associated with free peptide fragments. This strategic division of labor allows for parallel processing, significantly compressing the overall production timeline and enhancing the physical handling properties of the intermediates.
Mechanistic Insights into Fragment Condensation and Orthogonal Protection
The core chemical innovation lies in the orthogonal protection strategy employed at the Lysine-12 position, which serves as the anchor point for the fatty acid modification essential for Liraglutide's pharmacokinetic profile. The use of the Allyloxycarbonyl (Alloc) group is critical because it can be removed selectively using palladium catalysis in the presence of phenylsilane, a condition that leaves the acid-labile tBu and Trt protecting groups on other residues intact. This orthogonality allows the palmitoyl-glutamic acid moiety to be attached directly to the resin-bound peptide before the final fragment coupling, ensuring that the hydrophobic tail is incorporated early in a controlled environment. By attaching the fatty acid chain while the peptide is still anchored to the solid support, the method avoids the solubility crashes that often occur when modifying long, hydrophobic peptides in homogeneous solution. This on-resin modification step is pivotal for maintaining the homogeneity of the reaction mixture and ensuring consistent acylation across the entire batch.
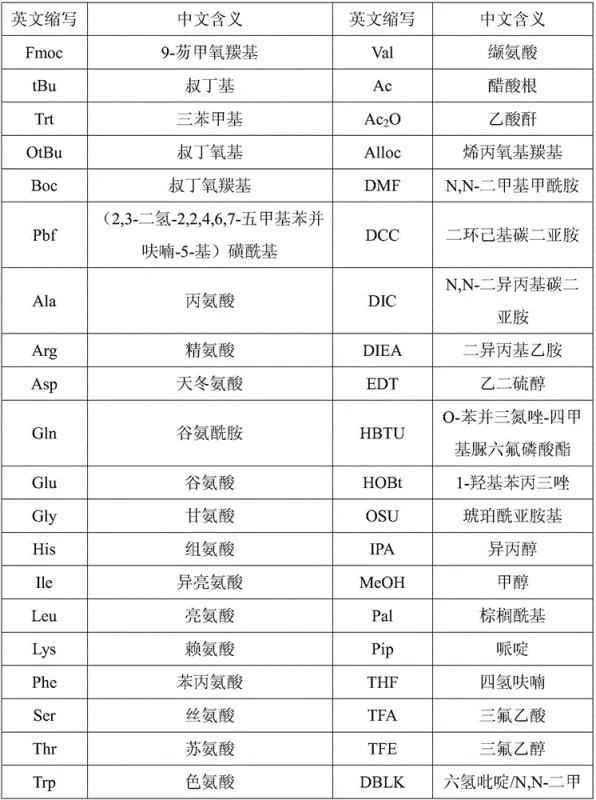
Furthermore, the choice of cleavage reagents for the second fragment demonstrates a nuanced understanding of peptide stability. The patent specifies the use of trifluoroethanol (TFE) in dichloromethane or low-concentration trifluoroacetic acid (TFA) to cleave the C-terminal fragment from the 2-CTC resin. Unlike standard high-acid cleavage cocktails that remove all protecting groups, this mild cleavage protocol releases the peptide fragment while retaining the side-chain protections. This is mechanistically significant because it prevents the exposure of reactive functional groups (such as the imidazole of Histidine or the carboxyl of Aspartic acid) during the storage and handling of the second fragment. When this protected fragment is subsequently coupled to the N-terminal resin-bound segment, the reaction proceeds with high fidelity, minimizing side reactions such as aspartimide formation or racemization at the coupling junction. The result is a crude product profile that is significantly cleaner, with the maximum single impurity controlled below 0.2%, facilitating a more efficient final purification step.
How to Synthesize Liraglutide Efficiently
The implementation of this synthesis route requires precise control over resin substitution degrees and coupling activation energies to ensure high fidelity at each step. The process begins with the loading of the first amino acid onto Wang resin, followed by iterative cycles of Fmoc deprotection and amino acid coupling using activating agents like DIC and HOBt. Critical attention must be paid to the deprotection of the Alloc group on Lysine-12, which requires strictly anhydrous conditions and scavengers like phenylsilane to prevent allyl group migration. Following the attachment of the fatty acid chain, the second fragment is prepared separately and coupled to the resin-bound species using potent coupling reagents such as PyBOP or HBTU to drive the sterically hindered amide bond formation to completion.
- Synthesize the first polypeptide fragment (residues 1-20) on Wang resin, utilizing Alloc protection for Lys12 to allow subsequent fatty acid coupling.
- Prepare the second polypeptide fragment (residues 21-31) on 2-CTC resin and perform mild cleavage to obtain the protected fragment in solution.
- Couple the two fragments on the solid support, followed by global deprotection and purification to yield high-purity Liraglutide acetate.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain and procurement perspective, this optimized synthesis route offers substantial advantages by mitigating the risks associated with raw material volatility and processing bottlenecks. The ability to synthesize the two fragments in parallel effectively decouples the production timeline, allowing manufacturing facilities to stockpile validated intermediates and respond rapidly to fluctuations in market demand without restarting the entire synthesis from scratch. This modularity enhances supply continuity, as a failure in one fragment synthesis does not necessarily compromise the inventory of the other, providing a buffer against batch losses that is absent in linear synthesis models. Moreover, the elimination of oily intermediates simplifies the logistical handling of materials, reducing the need for specialized equipment for viscous fluid transfer and minimizing material loss during weighing and charging operations.
- Cost Reduction in Manufacturing: The process achieves cost efficiency primarily through the reduction of downstream purification burdens and the optimization of raw material utilization. By preventing the formation of complex deletion peptide impurities that co-elute with the product, the method significantly reduces the consumption of expensive preparative HPLC columns and solvents, which are typically the largest variable costs in peptide manufacturing. Additionally, the high crude purity means that fewer recycling loops are required, leading to a higher effective throughput per reactor volume. The avoidance of transition metal catalysts in the main coupling steps further eliminates the need for costly heavy metal scavenging resins and the associated validation testing for residual metals, streamlining the overall cost structure.
- Enhanced Supply Chain Reliability: The reliance on standard Fmoc-protected amino acids and widely available coupling reagents ensures that the supply chain is not dependent on exotic or single-source proprietary building blocks. The use of robust solid-phase supports like Wang and 2-CTC resins, which are commodity items in the peptide industry, guarantees consistent availability and pricing stability. Furthermore, the improved physical state of the intermediates—avoiding the formation of intractable oils—reduces the risk of processing delays caused by clogging filters or pumping failures, thereby ensuring more predictable batch cycle times and reliable delivery schedules for downstream formulators.
- Scalability and Environmental Compliance: This methodology is inherently designed for scale-up, as solid-phase synthesis allows for straightforward expansion by simply increasing reactor bed volumes without altering the fundamental reaction kinetics. The reduction in solvent usage per gram of product, driven by higher yields and fewer purification passes, aligns with modern green chemistry principles and reduces the environmental footprint of the manufacturing site. The waste stream is also more manageable, as the absence of complex oily byproducts simplifies effluent treatment processes, helping manufacturers maintain compliance with increasingly stringent environmental regulations regarding organic solvent discharge and hazardous waste disposal.
Frequently Asked Questions (FAQ)
The following technical inquiries address common concerns regarding the implementation and validation of this specific solid-phase synthesis protocol for Liraglutide. These answers are derived directly from the experimental data and comparative examples provided in the patent documentation, focusing on the practical implications for process development and quality assurance teams. Understanding these nuances is essential for evaluating the feasibility of technology transfer and the potential for successful regulatory filing.
Q: How does this method address the issue of oily intermediates in Liraglutide synthesis?
A: By synthesizing the fragments separately and keeping the key coupling on-resin, this method avoids the isolation of difficult-to-handle oily short-chain fragments common in traditional solution-phase fragment condensation, thereby simplifying purification.
Q: What is the advantage of using Alloc protection for Lysine at position 12?
A: The Alloc group is orthogonal to the standard Fmoc/tBu protecting group strategy. This allows for the selective removal of the Lys12 side-chain protection to attach the palmitoyl-glutamic acid moiety without affecting the rest of the peptide chain or the resin linkage.
Q: What purity levels can be achieved with this specific solid-phase protocol?
A: The patent data indicates that the resulting Liraglutide acetate can achieve a purity of greater than 99.6%, with maximum single impurities controlled below 0.2%, significantly reducing the burden on downstream purification processes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Liraglutide Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires deep technical expertise and rigorous process control. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of advanced synthesis routes like the one described in CN110894227A are fully realized in a GMP environment. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify that every batch meets the exacting standards required for global pharmaceutical markets. Our commitment to quality ensures that the complex challenges of peptide coupling and impurity control are managed proactively, delivering a product that is ready for formulation.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis strategy can be tailored to your specific supply needs. By requesting a Customized Cost-Saving Analysis, you can gain visibility into how our process efficiencies translate into tangible value for your organization. We encourage you to contact us to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions based on verified performance metrics rather than theoretical projections. Let us collaborate to secure a stable, high-quality supply of Liraglutide that supports your long-term commercial goals.
