Advanced Catalytic Synthesis Of Paxlovid Bicyclic Imine Intermediate For Commercial Scale Production
Advanced Catalytic Synthesis Of Paxlovid Bicyclic Imine Intermediate For Commercial Scale Production
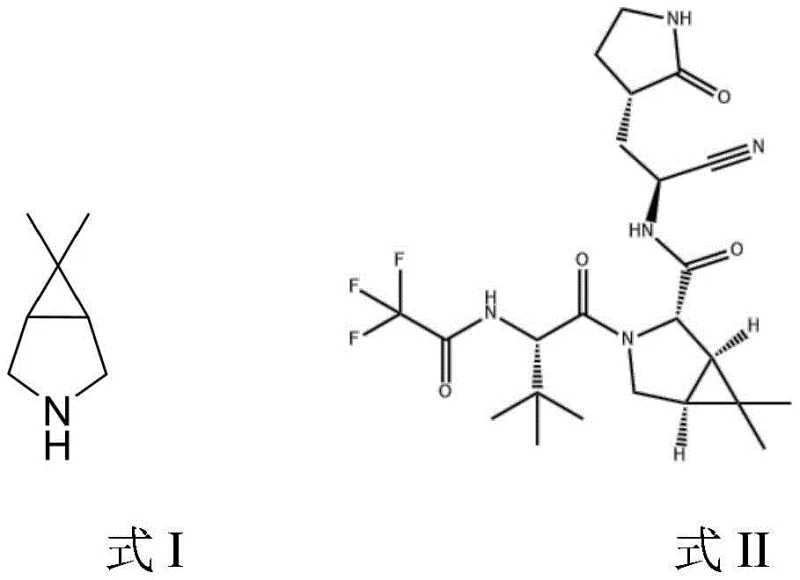
The global pharmaceutical landscape has been profoundly reshaped by the urgent demand for effective antiviral therapeutics, specifically targeting SARS-CoV-2. Central to this effort is the oral protease inhibitor Paxlovid, whose complex molecular architecture relies heavily on the availability of high-purity chiral building blocks. Patent CN114644587A discloses a groundbreaking synthetic methodology for producing the critical bicyclic imine intermediate, chemically defined as 3-aza-6,6-dimethylbicyclo[3.1.0]hexane derivatives. This innovation addresses the severe bottlenecks associated with earlier manufacturing protocols by introducing a streamlined, transition-metal catalyzed approach. The structural relationship between this key intermediate (Formula I) and the final active pharmaceutical ingredient Paxlovid (Formula II) underscores its strategic importance in the supply chain. By leveraging inexpensive starting materials like dichloroacetamide and isopentenyl halides, this technology offers a viable pathway for cost reduction in API manufacturing while maintaining rigorous quality standards required for clinical applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
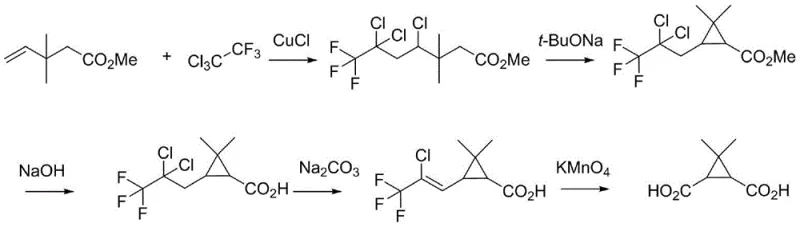
Prior to this technological breakthrough, the industrial synthesis of this specific bicyclic scaffold was plagued by significant operational hazards and environmental inefficiencies. Historical literature, such as the routes described in US20050059684A1, relied heavily on multi-step sequences that suffered from poor atom economy and generated substantial chemical waste. A particularly egregious example involves the use of potassium permanganate for oxidative cleavage, a process that not only creates heavy metal sludge requiring expensive disposal but also introduces purification challenges that compromise overall yield. Furthermore, alternative pathways documented in patents like CN114057627 necessitate the formation and handling of diazo intermediates. These diazo compounds are inherently unstable and pose severe safety risks, including potential explosion hazards during scale-up, making them unsuitable for large-scale commercial production under modern safety regulations. The cumulative effect of these drawbacks is a fragile supply chain characterized by long lead times and unpredictable production costs.
The Novel Approach
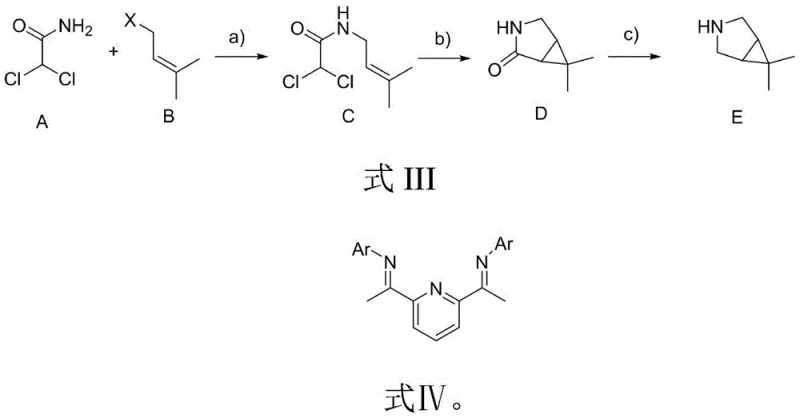
In stark contrast to these legacy methods, the process outlined in CN114644587A introduces a concise three-step strategy that fundamentally reimagines the construction of the bicyclic core. The new route initiates with a straightforward base-mediated coupling reaction, followed by a sophisticated cobalt or nickel-catalyzed zinc-carbene cyclization, and concludes with a selective amide reduction. This methodology completely bypasses the need for hazardous diazo species and aggressive oxidizing agents, thereby drastically simplifying the safety profile of the manufacturing plant. By utilizing readily available commodity chemicals such as dichloroacetamide and isopentenyl chloride, the process ensures a robust and continuous supply of raw materials, mitigating the risk of procurement delays. The elimination of high-risk unit operations not only enhances worker safety but also reduces the regulatory burden associated with handling explosive precursors, facilitating faster technology transfer from laboratory to pilot plant.
Mechanistic Insights into Cobalt-Catalyzed Zinc-Carbene Cyclization
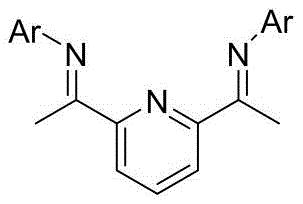
The cornerstone of this synthetic advancement lies in the second step: the transition metal-catalyzed cyclization of the linear dichloro-amide precursor. Mechanistically, this transformation involves the generation of a reactive zinc-carbene species from the gem-dichloro moiety under the influence of a low-valent cobalt or nickel catalyst. The reaction proceeds through a concerted cyclopropanation mechanism where the metal-carbene intermediate inserts into the proximal olefinic bond present in the isopentenyl side chain. Critical to the success of this step is the use of specialized nitrogen-based ligands, specifically the pyridine-bis(imine) derivatives labeled as L1 and L2 in the patent documentation. These ligands modulate the electronic properties of the metal center, stabilizing the active catalytic species and ensuring high regioselectivity for the formation of the strained [3.1.0] bicyclic system over potential polymeric byproducts. The precise control over stereochemistry and ring closure efficiency is what allows this route to achieve superior purity profiles compared to non-catalytic thermal cyclizations.
Following the cyclization, the resulting bicyclic lactam undergoes a rigorous reduction phase to yield the target imine. The patent explores various reducing agents, including sodium borohydride, lithium aluminum hydride, and red-al (sodium bis(2-methoxyethoxy)aluminum hydride), demonstrating flexibility in process optimization. The choice of reducing agent can be tailored based on specific impurity profiles; for instance, using red-al in THF at controlled temperatures allows for the complete conversion of the amide carbonyl to the methylene group without affecting other sensitive functionalities. This step is crucial for establishing the correct oxidation state of the nitrogen atom within the bicyclic framework. Furthermore, the workup procedures described, involving acid quenching and careful pH adjustment during extraction, are designed to minimize the formation of oligomeric impurities, ensuring that the final crude product meets the stringent specifications required for subsequent coupling reactions in the total synthesis of Paxlovid.
How to Synthesize 3-aza-6,6-dimethylbicyclo[3.1.0]hexane Efficiently
Implementing this synthesis requires precise control over reaction parameters to maximize the reported yields of up to 65%. The process begins with the alkylation of dichloroacetamide using a strong base such as potassium carbonate or sodium hydride in polar aprotic solvents like DMF. Once the linear precursor is isolated, it is subjected to the cyclization conditions using zinc dust and a catalytic loading of cobalt bromide alongside the specific ligand L2. The final reduction is typically performed in etheral solvents, followed by a distillation or crystallization to isolate the pure amine. For detailed operational parameters, temperature gradients, and stoichiometric ratios essential for reproducibility, please refer to the standardized protocol below.
- Coupling Reaction: React dichloroacetamide with isopentenyl halide using a base like potassium carbonate or sodium hydride in DMF or THF to form the linear precursor.
- Cyclization: Treat the precursor with zinc powder and a catalytic amount of cobalt or nickel halide with specific ligands (L1 or L2) to induce zinc-carbene cyclization.
- Reduction: Reduce the resulting cyclic amide using reducing agents such as red-al, lithium aluminum hydride, or borane to yield the final bicyclic imine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route translates into tangible strategic benefits beyond mere technical novelty. The primary advantage lies in the drastic simplification of the raw material basket; by shifting away from custom-synthesized, high-cost precursors to bulk commodity chemicals like dichloroacetamide, the exposure to volatile market pricing is significantly minimized. This shift ensures a more stable cost structure for the intermediate, allowing for better long-term budget forecasting. Additionally, the removal of hazardous diazo chemistry eliminates the need for specialized explosion-proof reactor infrastructure, which often represents a significant capital expenditure barrier for contract manufacturing organizations. This accessibility means that a broader range of qualified suppliers can potentially manufacture this intermediate, increasing competition and further driving down costs for the end buyer.
- Cost Reduction in Manufacturing: The economic viability of this process is underpinned by its exceptional atom economy and the avoidance of expensive reagents. Traditional routes often require stoichiometric amounts of heavy metal oxidants or precious metal catalysts that are difficult to recover; in contrast, this method utilizes earth-abundant cobalt or nickel salts in catalytic quantities. The reduction in reagent costs is compounded by the simplified downstream processing; fewer purification steps mean lower solvent consumption and reduced energy usage for distillation and drying. Consequently, the overall cost of goods sold (COGS) for the bicyclic imine is substantially lower, providing a competitive edge in the pricing of the final antiviral medication.
- Enhanced Supply Chain Reliability: Supply continuity is a critical metric for pharmaceutical manufacturers, and this route excels by decoupling production from scarce reagents. The reliance on isopentenyl halides and dichloroacetamide ensures that raw material sourcing is not a bottleneck, as these are produced globally at massive scales for other industries. Furthermore, the robustness of the reaction conditions—operating at moderate temperatures and pressures—reduces the likelihood of batch failures due to equipment sensitivity. This reliability shortens the effective lead time for high-purity pharmaceutical intermediates, enabling manufacturers to respond more agilely to surges in global demand for pandemic-related therapeutics without compromising on quality assurance protocols.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this process represents a significant leap forward in green chemistry principles. The elimination of potassium permanganate waste streams removes a major liability associated with heavy metal disposal and wastewater treatment. The process generates significantly less hazardous waste per kilogram of product, aligning with increasingly strict global environmental regulations. This compliance reduces the administrative and financial burden of waste management, facilitating smoother regulatory approvals for manufacturing sites. The inherent safety of avoiding energetic diazo compounds also lowers insurance premiums and operational risks, making the commercial scale-up of complex pharmaceutical intermediates a more attractive investment for stakeholders.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent literature, offering a transparent view of the process capabilities. Understanding these nuances is essential for R&D teams evaluating technology transfer and procurement officers assessing supplier qualifications. The answers reflect the current state of the art as defined by the intellectual property rights associated with this specific catalytic methodology.
Q: What are the safety advantages of this new synthesis route compared to traditional methods?
A: Unlike conventional routes that utilize hazardous diazo intermediates or high-pollution oxidants like potassium permanganate, this novel process employs stable dichloroacetamide and safe transition metal catalysis, significantly eliminating explosion risks and toxic waste generation.
Q: What is the overall yield efficiency of the patented catalytic process?
A: The optimized three-step sequence demonstrates robust technical feasibility with combined molar yields reaching up to 65% in specific embodiments, proving high atom economy suitable for industrial scaling.
Q: Which catalysts and ligands are critical for the cyclization step?
A: The process relies on cobalt or nickel halides paired with specialized pyridine-bis(imine) ligands (such as L1 or L2) to effectively mediate the zinc-carbene cyclization, ensuring high selectivity for the bicyclic structure.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-aza-6,6-dimethylbicyclo[3.1.0]hexane Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful deployment of advanced synthetic routes like the one described in CN114644587A requires more than just theoretical knowledge; it demands practical expertise in process engineering and quality control. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from benchtop to reactor is seamless and efficient. Our facilities are equipped with state-of-the-art rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of bicyclic imine intermediate meets the exacting standards required for GMP manufacturing of antiviral APIs. We are committed to delivering consistent quality that supports your clinical and commercial timelines.
We invite you to collaborate with us to leverage this innovative technology for your supply chain needs. Our technical team is prepared to conduct a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this catalytic route can optimize your budget. We encourage potential partners to contact our technical procurement team to request specific COA data and route feasibility assessments. By partnering with us, you secure not just a chemical supplier, but a strategic ally dedicated to advancing the availability of life-saving medications through superior chemical manufacturing excellence.
