Advanced Synthesis of Paxlovid Bicyclic Imine Intermediate for Commercial Scale-up
Advanced Synthesis of Paxlovid Bicyclic Imine Intermediate for Commercial Scale-up
The global pharmaceutical landscape has been profoundly reshaped by the urgent demand for effective antiviral therapeutics, particularly small molecule inhibitors targeting the SARS-CoV-2 3CL protease. At the heart of this therapeutic revolution lies Paxlovid, a co-packaged oral treatment whose efficacy relies heavily on the consistent supply of its complex chiral intermediates. Patent CN114644587A introduces a groundbreaking synthetic methodology for producing 3-aza-6,6-dimethylbicyclo[3.1.0]hexane, the critical bicyclic imine scaffold required for the assembly of the PF-07321332 active pharmaceutical ingredient. This technical disclosure represents a significant leap forward in process chemistry, addressing the longstanding bottlenecks of safety, cost, and scalability that have plagued earlier manufacturing routes. By leveraging a concise three-step sequence involving coupling, transition-metal catalyzed cyclization, and selective reduction, this innovation offers a robust pathway for reliable pharmaceutical intermediate supplier networks to secure the global supply chain against disruption.
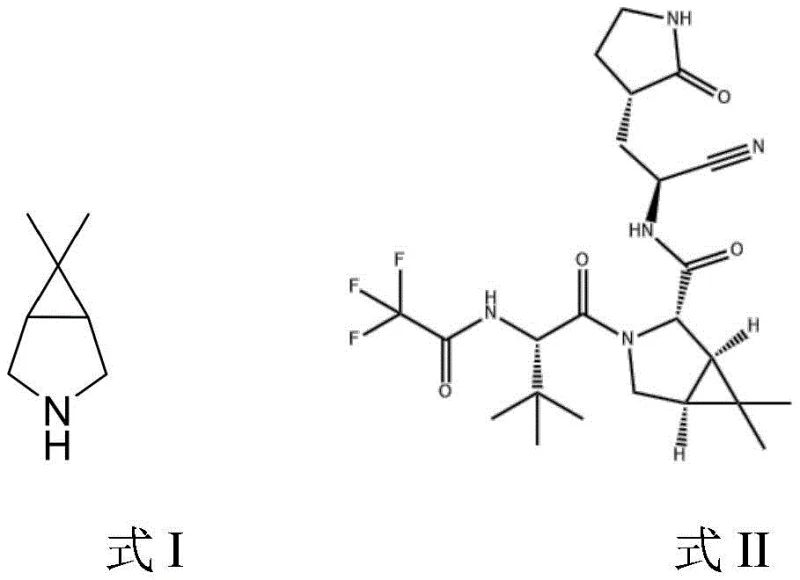
The structural complexity of the target molecule necessitates a synthesis strategy that balances stereochemical precision with operational simplicity. As illustrated in the patent documentation, the target bicyclic imine serves as the foundational core upon which the peptidomimetic structure of the antiviral agent is built. The ability to construct this strained bicyclic system efficiently is paramount for meeting the massive volume requirements dictated by pandemic response efforts. This new process not only streamlines the chemical transformations but also aligns with modern green chemistry principles by minimizing waste generation and eliminating the need for exotic or hazardous reagents that complicate regulatory compliance and facility safety protocols.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this invention, the industrial synthesis of this关键 intermediate was fraught with significant technical and economic challenges that hindered efficient mass production. Existing literature and patent filings, such as those referenced in US20050059684A1 and EP0010799, describe routes that suffer from poor atom economy and excessive step counts, leading to inflated production costs and extended lead times. A particularly problematic aspect of these legacy methods is the reliance on harsh oxidative conditions, specifically the use of potassium permanganate, which generates substantial quantities of manganese dioxide sludge, creating a severe environmental burden and complicating waste disposal logistics. Furthermore, alternative pathways reported in patents like CN114057627 involve the generation and handling of diazo intermediates, which are inherently unstable and pose serious safety risks including potential explosion hazards, making them unsuitable for large-scale operations in standard chemical plants.
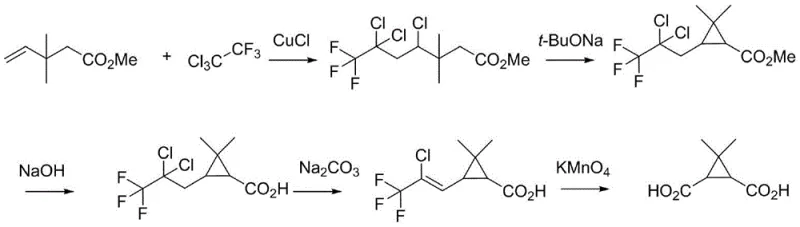
The cumulative effect of these deficiencies is a fragile supply chain vulnerable to shutdowns and quality deviations. The long synthetic sequences increase the probability of yield loss at each stage, while the use of high-risk reagents necessitates specialized equipment and rigorous safety monitoring, driving up capital expenditure. Additionally, the purification of intermediates generated through these convoluted routes often requires energy-intensive chromatography or recrystallization steps, further eroding profit margins. For procurement managers and supply chain heads, these factors translate into higher unit costs and unpredictable delivery schedules, undermining the ability to secure a steady flow of high-purity pharmaceutical intermediates needed for continuous drug manufacturing.
The Novel Approach
In stark contrast to the cumbersome legacy processes, the methodology disclosed in Patent CN114644587A presents a streamlined and economically superior alternative that fundamentally reimagines the construction of the bicyclic core. The new route initiates with a straightforward nucleophilic substitution between dichloroacetamide and an isopentenyl halide, utilizing inexpensive and widely available bases such as potassium carbonate or sodium hydride to forge the initial carbon-nitrogen bond. This is followed by a sophisticated intramolecular cyclization mediated by a cobalt or nickel catalyst in the presence of zinc powder, which effectively constructs the strained cyclopropane ring system in a single operation with high efficiency. The final step involves a chemoselective reduction of the resulting lactam to the corresponding amine using mild reducing agents like Red-Al or borane, completing the synthesis in just three distinct chemical transformations.
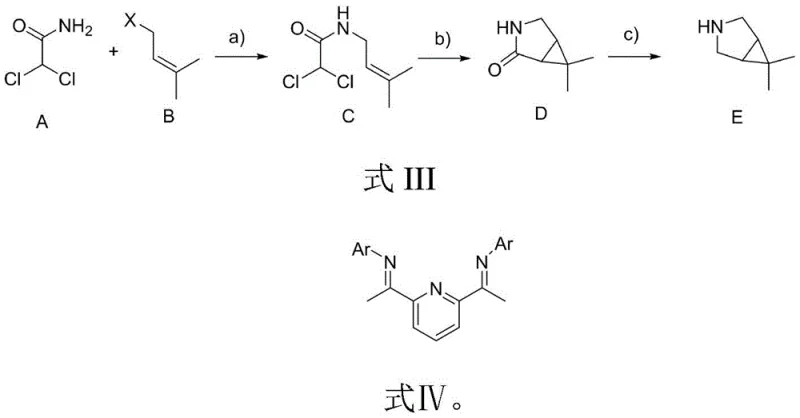
This innovative approach delivers substantial cost reduction in API manufacturing by drastically shortening the production timeline and eliminating the need for hazardous diazo chemistry. The use of earth-abundant transition metals like cobalt and nickel, rather than precious metals, combined with the avoidance of toxic oxidants, significantly lowers the raw material bill and simplifies the downstream purification process. Experimental data from the patent indicates that this optimized sequence can achieve combined molar yields of up to 65%, a figure that underscores the robustness of the chemistry when translated from the laboratory to the pilot plant. For industry stakeholders, this translates to a more resilient supply chain capable of scaling rapidly to meet surging demand without compromising on safety or environmental standards.
Mechanistic Insights into Cobalt-Catalyzed Zinc Carbene Cyclization
The cornerstone of this synthetic breakthrough is the transition metal-catalyzed cyclization step, which orchestrates the formation of the bicyclic framework with remarkable precision. Mechanistically, this transformation likely proceeds through the generation of a metal-carbene species from the dichloro-substituted precursor upon interaction with activated zinc. The cobalt or nickel center, coordinated by specific bidentate nitrogen ligands such as the pyridine-bis(imine) derivatives shown in Formula IV, stabilizes the reactive intermediate and directs the intramolecular insertion into the proximal olefin. This catalytic cycle is finely tuned to favor the formation of the desired cis-fused cyclopropane ring while suppressing competing side reactions such as polymerization or non-selective reduction, ensuring high product fidelity.
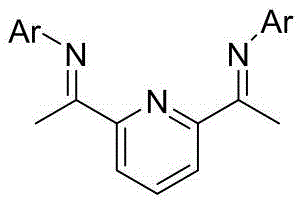
Impurity control is rigorously maintained through the careful selection of ligands and reaction parameters, which govern the stereoelectronic environment of the catalyst. The patent specifies the use of ligands L1 (Ar=2,4-di-tert-butylphenyl) and L2 (Ar=2,4,6-trimethylphenyl), which provide the necessary steric bulk to enforce the correct geometric approach of the substrate during the ring-closing event. By optimizing the ratio of catalyst to substrate and controlling the temperature profile between 30°C and 110°C, the process minimizes the formation of regioisomers and over-reduced byproducts. This level of mechanistic understanding allows process chemists to fine-tune the reaction conditions to maximize yield and purity, ensuring that the final intermediate meets the stringent specifications required for subsequent coupling with the rest of the antiviral molecule.
How to Synthesize 3-aza-6,6-dimethylbicyclo[3.1.0]hexane Efficiently
Implementing this synthesis protocol requires adherence to precise operational parameters to ensure reproducibility and safety across different manufacturing scales. The process begins with the preparation of the amide precursor under controlled basic conditions, followed by the critical cyclization step where moisture and oxygen levels must be carefully managed to preserve catalyst activity. The final reduction is exothermic and requires controlled addition of the reducing agent to prevent thermal runaway, followed by a careful quenching and workup procedure to isolate the free base or its salt form. Detailed standard operating procedures regarding stoichiometry, solvent selection, and purification techniques are essential for successful technology transfer.
- Coupling Reaction: React dichloroacetamide with isopentenyl halide using a base like potassium carbonate or sodium hydride to form the amide intermediate.
- Cyclization: Perform a cobalt or nickel-catalyzed zinc carbene cyclization using specific ligands (L1 or L2) to form the bicyclic lactam structure.
- Reduction: Reduce the lactam carbonyl group using reducing agents such as Red-Al, borane, or lithium aluminum hydride to yield the final bicyclic imine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis route offers compelling strategic benefits that extend beyond simple unit price considerations. By transitioning to a process that utilizes commodity chemicals like dichloroacetamide and isopentenyl halides, manufacturers can decouple their supply chains from the volatility associated with specialized, low-volume reagents. This shift towards readily available starting materials enhances supply security and mitigates the risk of production stoppages due to raw material shortages, a critical factor in maintaining business continuity for high-demand therapeutics. Furthermore, the elimination of high-risk diazo intermediates removes a significant barrier to entry for many contract manufacturing organizations, thereby expanding the pool of qualified suppliers and fostering a more competitive sourcing environment.
- Cost Reduction in Manufacturing: The streamlined three-step sequence inherently reduces manufacturing costs by minimizing the number of unit operations, solvent exchanges, and isolation steps required to reach the final intermediate. The avoidance of expensive oxidants and the use of base metal catalysts instead of precious metals like palladium or rhodium contribute to a lower overall cost of goods sold. Additionally, the improved atom economy and higher yields mean that less raw material is wasted, further driving down the effective cost per kilogram of the produced intermediate without compromising quality standards.
- Enhanced Supply Chain Reliability: The robustness of this chemistry ensures consistent batch-to-batch quality, which is vital for maintaining regulatory compliance and avoiding costly recalls or rejections. By simplifying the synthetic pathway, the potential for process deviations is reduced, leading to more predictable production schedules and shorter lead times for order fulfillment. This reliability allows pharmaceutical companies to optimize their inventory levels and reduce the need for excessive safety stock, freeing up working capital and improving overall supply chain agility in response to market fluctuations.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this process represents a significant improvement over prior art by eliminating the generation of heavy metal waste and hazardous diazo compounds. The reduced waste profile simplifies effluent treatment and lowers the environmental compliance burden, making it easier to obtain necessary permits for large-scale production. The inherent safety of the reagents and conditions also facilitates easier scale-up from pilot to commercial tonnage, enabling rapid capacity expansion to meet global demand without requiring extensive retrofitting of existing manufacturing facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. Understanding these details is crucial for R&D teams evaluating the feasibility of adopting this route and for procurement professionals assessing the long-term viability of the supply source. The answers provided are derived directly from the experimental data and technical disclosures within the patent specification, ensuring accuracy and relevance for decision-making purposes.
Q: What are the safety advantages of this new synthesis route compared to prior art?
A: Unlike previous methods that rely on hazardous diazo intermediates or high-pollution oxidants like potassium permanganate, this process utilizes stable dichloroacetamide and safe transition metal catalysis, significantly reducing explosion risks and environmental waste.
Q: What is the overall yield of the patented synthesis process?
A: According to the experimental examples in Patent CN114644587A, the combined molar yield for the three-step process ranges from 45% to 65%, depending on the specific catalyst and reducing agent combinations used.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is designed for scalability by using readily available raw materials, avoiding cryogenic conditions, and eliminating high-risk steps, making it highly suitable for commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-aza-6,6-dimethylbicyclo[3.1.0]hexane Supplier
As the global demand for antiviral therapeutics continues to evolve, securing a dependable source for critical intermediates like 3-aza-6,6-dimethylbicyclo[3.1.0]hexane is more important than ever. NINGBO INNO PHARMCHEM stands at the forefront of this effort, leveraging our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to deliver high-quality intermediates consistently. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced analytical capabilities to ensure that every batch meets stringent purity specifications, providing our partners with the confidence they need to advance their drug development programs without delay.
We invite you to collaborate with us to explore how this innovative synthesis route can optimize your supply chain and reduce your overall manufacturing costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our expertise can support your mission of delivering life-saving medications to patients worldwide.
