Advanced Asymmetric Birch Reduction for Commercial Production of Chiral 3,4-Dehydroprolines
Advanced Asymmetric Birch Reduction for Commercial Production of Chiral 3,4-Dehydroprolines
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to access rigid, chiral scaffolds that serve as critical building blocks for bioactive molecules. Patent CN1259122A introduces a groundbreaking methodology for the preparation of chiral 3,4-dehydroproline compounds, a structural motif essential for the synthesis of potent thrombin inhibitors and other therapeutic agents. This technology represents a significant departure from legacy synthetic routes by leveraging an asymmetric Birch reduction strategy on pyrrole carboxylic acid derivatives. By reacting these precursors with alkali or alkaline earth metals in liquid ammonia, followed by specific electrophilic trapping, the process achieves high stereoselectivity and yield. For R&D directors and procurement specialists, this patent offers a compelling alternative to toxic and low-yielding historical methods, promising a more sustainable and economically viable supply chain for high-value intermediates.
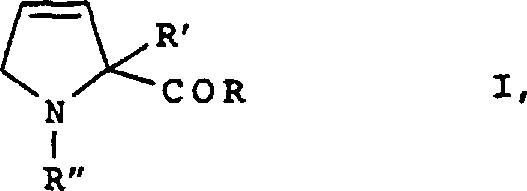
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3,4-dehydroprolines has been plagued by significant safety and efficiency hurdles that impact both operational costs and environmental compliance. The classical Chugaev elimination route, while chemically valid, necessitates the use of extremely hazardous reagents such as dithiocarbonic anhydride, methyl iodide, and thiomethyl alcohol. These substances pose severe handling risks and generate toxic waste streams that require expensive disposal protocols. Furthermore, this method often requires harsh pyrolysis conditions at temperatures ranging from 180°C to 190°C under vacuum, leading to high energy consumption and potential thermal degradation of sensitive intermediates. Alternative approaches involving the resolution of racemic mixtures are equally problematic, as they are theoretically capped at a maximum yield of 50%, effectively wasting half of the starting material and doubling the raw material costs for the desired enantiomer.
The Novel Approach
In stark contrast, the methodology disclosed in CN1259122A circumvents these bottlenecks by utilizing a direct asymmetric synthesis via Birch reduction. This innovative approach eliminates the need for toxic sulfur-based reagents and high-temperature pyrolysis, replacing them with a cleaner reduction using lithium, sodium, or potassium in liquid ammonia. The core breakthrough lies in the use of chiral auxiliaries attached to the pyrrole nitrogen or carboxyl group, which direct the stereochemical outcome of the reduction. This allows for the direct formation of the desired chiral 3,4-dehydroproline with high optical purity, bypassing the inefficient resolution step entirely. By shifting from a resolution-based paradigm to a direct asymmetric synthesis, manufacturers can theoretically double their output from the same amount of starting material, drastically improving the overall process mass intensity and reducing the cost of goods sold.
Mechanistic Insights into Asymmetric Birch Reduction of Pyrroles
The heart of this technology is the controlled reduction of the aromatic pyrrole ring, a transformation that is notoriously difficult to achieve with high regio- and stereoselectivity. In this process, the pyrrole-2-carboxylic acid derivative is dissolved in a mixture of liquid ammonia and an inert solvent like THF at cryogenic temperatures, typically around -30°C to -70°C. The addition of an alkali metal, preferably lithium, generates solvated electrons that attack the electron-deficient pyrrole ring. The presence of the chiral auxiliary—such as a non-aromatic chiral amine or a chiral ester like bornyl ester—creates a steric environment that forces the electron addition to occur from a specific face of the molecule. This facial selectivity is crucial, as it establishes the chiral center at the 4-position of the resulting dihydropyrrole intermediate, which subsequently tautomerizes or is trapped to form the stable 3,4-dehydroproline structure.
Impurity control in this system is managed through the precise selection of the quenching agent and reaction conditions. After the reduction is complete, indicated by the disappearance of the starting pyrrole derivative via HPLC or TLC, the reactive intermediate is treated with an aqueous salt solution or an alkylating agent (R'-X). If protonation is desired, ammonium chloride is used; if alkylation is required, agents like methyl iodide or benzyl bromide can be employed. The patent highlights that despite the electronic variations introduced by different chiral radicals, the reaction maintains high selectivity. This robustness suggests that side reactions, such as over-reduction or polymerization, are minimized by the specific coordination of the metal cation with the chiral auxiliary and the carbonyl oxygen. This level of mechanistic control ensures that the final product profile is clean, reducing the burden on downstream purification steps like chromatography or crystallization.
How to Synthesize Chiral 3,4-Dehydroprolines Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for translating this chemistry from the laboratory bench to pilot plant operations. The procedure involves dissolving the protected pyrrole precursor in a cold ammonia/THF mixture, followed by the controlled addition of lithium wire or granules. The reaction is exothermic and requires careful temperature management to maintain the integrity of the chiral information being transferred. Once the reduction phase is complete, the reaction is quenched, and the product is isolated through standard workup procedures such as extraction with dichloromethane and concentration under reduced pressure. The detailed standardized synthesis steps below outline the specific molar ratios, temperature profiles, and workup sequences required to replicate the high yields reported in the patent examples.
- Dissolve the pyrrole-2-carboxylic acid derivative bearing a chiral auxiliary in a mixture of liquid ammonia and THF at cryogenic temperatures.
- Add alkali metal (Lithium, Sodium, or Potassium) gradually to effect the Birch reduction of the pyrrole ring, generating a chiral enolate intermediate.
- Quench the reaction mixture with an aqueous salt solution or an alkylating agent (R'-X) to form the final chiral 3,4-dehydroproline product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this Birch reduction technology translates into tangible strategic advantages beyond mere chemical elegance. The elimination of highly toxic reagents like dithiocarbonic anhydride significantly reduces the regulatory burden and the costs associated with hazardous waste disposal and worker safety monitoring. Furthermore, by moving away from resolution processes that discard 50% of the material, the effective cost of the starting materials is halved, leading to substantial savings in raw material expenditure. The ability to run the reaction in continuous flow reactors, as suggested by the patent's mention of inline reactor groups, offers additional flexibility for scaling production to meet fluctuating market demands without the need for massive capital investment in new batch infrastructure.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the dramatic improvement in atom economy compared to traditional resolution methods. By synthesizing the chiral center directly rather than separating it from a racemic mixture, the process effectively doubles the yield relative to the input of chiral starting materials. Additionally, the removal of expensive and toxic sulfur reagents lowers the direct material costs, while the milder reaction conditions reduce energy consumption associated with high-temperature pyrolysis. These factors combine to create a significantly lower cost base for the production of these valuable intermediates, allowing for more competitive pricing in the global API market.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of readily available and stable starting materials such as pyrrole-2-carboxylic acid derivatives and common alkali metals. Unlike specialized enzymatic resolution processes that may rely on proprietary biocatalysts with limited shelf-life or supply, the reagents for this Birch reduction are commodity chemicals available from multiple global suppliers. This diversification of the supply base mitigates the risk of single-source bottlenecks. Moreover, the robustness of the reaction across different chiral auxiliaries means that if one specific chiral source becomes scarce, the process can potentially be adapted to use alternative chiral pools without compromising the overall synthetic strategy.
- Scalability and Environmental Compliance: The environmental profile of this process is markedly superior to legacy methods, aligning with the increasingly strict ESG (Environmental, Social, and Governance) mandates of multinational pharmaceutical companies. The absence of heavy metal catalysts and toxic sulfur byproducts simplifies wastewater treatment and reduces the carbon footprint of the manufacturing process. From a scalability perspective, the patent explicitly notes that the reaction can be adapted for continuous processing, which is the gold standard for modern chemical manufacturing. Continuous flow allows for better heat and mass transfer control, ensuring consistent product quality even as production volumes increase from kilograms to multi-ton scales, thereby securing long-term supply continuity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this asymmetric Birch reduction technology. These answers are derived directly from the experimental data and claims presented in patent CN1259122A, providing a factual basis for evaluating the feasibility of this route for your specific project needs. Understanding these nuances is critical for R&D teams planning process validation and for procurement teams assessing vendor capabilities.
Q: What are the primary advantages of this Birch reduction method over traditional Chugaev elimination?
A: Unlike the traditional Chugaev elimination which requires highly toxic reagents like dithiocarbonic anhydride and methyl iodide, this patented method utilizes safer alkali metals in ammonia. Furthermore, it bypasses the theoretical 50% yield ceiling inherent in classical racemic resolution processes, significantly improving atom economy.
Q: How does the process ensure high stereoselectivity without enzymatic resolution?
A: The process employs non-aromatic chiral auxiliaries attached to the pyrrole nitrogen or carboxyl group. These bulky chiral groups direct the facial selectivity of the electron addition during the Birch reduction, effectively transferring chirality directly to the 3,4-dehydroproline core without the need for downstream separation.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the patent explicitly describes that the reaction can be performed in batch reactors with stirring or adapted for continuous flow processing using reaction tubes. The simplicity of the workup, involving standard extraction and concentration, supports robust commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral 3,4-Dehydroproline Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality chiral intermediates play in the development of next-generation therapeutics. Our team of expert chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from patent literature to industrial reality is seamless. We possess the rigorous QC labs and stringent purity specifications necessary to handle sensitive chiral compounds, guaranteeing that every batch of 3,4-dehydroproline meets the exacting standards required for GMP API synthesis. Our commitment to technical excellence means we can navigate the complexities of cryogenic Birch reductions with precision and safety.
We invite you to collaborate with us to leverage this advanced synthetic technology for your pipeline projects. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this Birch reduction route for your specific volume requirements. We encourage you to contact us today to discuss your specific COA data requirements and to receive comprehensive route feasibility assessments tailored to your production timelines and quality targets.
