Advanced Manufacturing of N-Acyl-3,4-Dehydroprolines: A Strategic Upgrade for API Intermediates
Advanced Manufacturing of N-Acyl-3,4-Dehydroprolines: A Strategic Upgrade for API Intermediates
The landscape of pharmaceutical intermediate manufacturing is constantly evolving, driven by the urgent need for safer, more efficient, and environmentally sustainable synthetic routes. A pivotal advancement in this domain is detailed in patent CN1345310A, which discloses a robust method for producing N-acyl-3,4-dehydroprolines and N-acyl-3,4-dehydropiperidines-2-carboxylic derivatives. These compounds serve as critical building blocks for the synthesis of thrombin inhibitors, crop protection agents, and specialized dyes. The technology represents a significant departure from legacy methodologies, offering a pathway that aligns with modern green chemistry principles while addressing the stringent purity requirements demanded by global regulatory bodies. For R&D directors and procurement strategists, understanding the nuances of this patent is essential for optimizing supply chains and reducing the total cost of ownership in API manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3,4-dehydroproline has been fraught with significant technical and safety challenges that hinder efficient commercial production. The traditional Tschugaeff reaction, starting from 4-hydroxyproline, necessitates the use of highly toxic and hazardous reagents such as carbon disulfide, methyl iodide, and methyl mercaptan. Beyond the severe safety risks associated with handling these volatile substances, the process requires extreme thermal conditions, specifically pyrolysis at temperatures ranging from 180°C to 190°C under reduced pressure. Such harsh conditions not only demand complex and expensive engineering controls but also result in relatively low yields, typically around 64%, which is suboptimal for large-scale operations. Furthermore, alternative achiral syntheses involving pyrrole carboxylic acid reduction rely on toxic phosphines and offer a maximum theoretical yield of only 50% after racemate separation, creating a substantial bottleneck for cost reduction in pharmaceutical intermediate manufacturing.
The Novel Approach
In stark contrast to these cumbersome legacy processes, the methodology outlined in CN1345310A introduces a streamlined reaction scheme that operates under remarkably mild conditions. By reacting a pyrroline or dehydropiperidine derivative of general formula (II) with a strong base and a carboxylating agent, the process avoids the need for toxic heavy metals or extreme thermal decomposition. The reaction typically proceeds at temperatures between -20°C and +20°C, significantly lowering energy consumption and simplifying reactor design requirements. This approach not only mitigates the environmental and safety liabilities associated with traditional methods but also enhances the overall process efficiency. The ability to utilize readily available reagents like dimethyl carbonate and alkali metal amides transforms the production of these valuable intermediates into a more predictable and economically viable operation, directly addressing the pain points of supply chain continuity and operational safety.
Mechanistic Insights into Base-Mediated Carboxylation
The core innovation of this technology lies in the precise control of the deprotonation and subsequent carboxylation steps. The reaction initiates with the treatment of the N-protected pyrroline or dehydropiperidine substrate with a strong base, preferably an alkali metal amide such as Lithium Diisopropylamide (LDA). This step generates a reactive anionic species at the 2-position of the heterocyclic ring, which is then intercepted by an electrophilic carboxylating agent, such as a dialkyl carbonate (e.g., dimethyl carbonate or diethyl carbonate). The choice of solvent plays a critical role in stabilizing these intermediates; inert solvents like tetrahydrofuran (THF), hexane, or mixtures containing DMPU are employed to ensure optimal solubility and reaction kinetics without participating in side reactions. The mechanistic elegance of this route ensures high regioselectivity, preventing the formation of unwanted byproducts that often plague less controlled synthetic pathways.
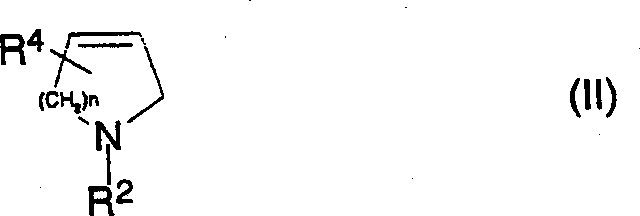
Furthermore, the process demonstrates superior impurity control mechanisms compared to prior art. Previous attempts at carboxylating similar substrates, such as carbomethoxy-3-pyrroline, often suffered from undesired intermolecular reactions that compromised yield and purity. The specific conditions defined in this patent, particularly the controlled addition of the base at low temperatures (often around 0°C or slightly below), effectively suppresses these competing pathways. By maintaining strict thermal regulation during the addition of the lithiating agent, the reaction favors the desired intramolecular or direct carboxylation trajectory. This results in a cleaner crude product profile, which simplifies downstream purification steps such as distillation or crystallization, ultimately delivering a high-purity pharmaceutical intermediate that meets the rigorous specifications required for subsequent coupling reactions in drug synthesis.
How to Synthesize N-Acyl-3,4-Dehydroproline Efficiently
The practical implementation of this synthesis route involves a sequence of well-defined unit operations that are amenable to both batch and continuous processing. The protocol begins with the dissolution of the starting pyrroline derivative and the carbonate reagent in an anhydrous solvent, followed by cooling to sub-ambient temperatures to manage the exotherm upon base addition. The standardized synthetic steps involve the dropwise addition of the LDA solution while monitoring the internal temperature to ensure it remains within the optimal window, typically not exceeding 4°C during the addition phase. Following the reaction period, the mixture is quenched with dilute acid, and the product is isolated through phase separation and extraction. Detailed standardized synthesis steps are provided in the guide below.
- Dissolve the pyrroline or dehydropiperidine derivative (Formula II) and a carboxylating agent such as dimethyl carbonate in an inert solvent like tetrahydrofuran.
- Cool the mixture to a temperature range between -20°C and +20°C, preferably around 0°C to -5°C, to control exothermic reactions.
- Add a strong base, specifically an alkali metal amide like Lithium Diisopropylamide (LDA), dropwise while maintaining strict temperature control, followed by standard aqueous workup and purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented technology offers tangible strategic benefits that extend beyond mere chemical novelty. The elimination of highly toxic reagents like carbon disulfide and phosphines removes the need for specialized containment systems and expensive waste disposal protocols associated with hazardous materials. This shift fundamentally alters the cost structure of production, allowing for significant cost savings in manufacturing overhead and regulatory compliance. Moreover, the milder reaction conditions reduce the energy load on production facilities, contributing to a lower carbon footprint and aligning with corporate sustainability goals. The robustness of the process ensures that supply disruptions caused by safety incidents or environmental violations are minimized, thereby enhancing the reliability of the supply chain for critical API intermediates.
- Cost Reduction in Manufacturing: The transition away from expensive and hazardous reagents towards commodity chemicals like dimethyl carbonate and LDA drives down raw material costs. Additionally, the higher yields achieved (up to 82% in demonstrated examples compared to 50-64% in older methods) mean that less starting material is required to produce the same amount of final product, effectively increasing the throughput of existing assets. The simplified purification process, often requiring only distillation or simple crystallization rather than complex chromatography, further reduces operational expenditures and solvent consumption, leading to substantial cost optimization across the entire production lifecycle.
- Enhanced Supply Chain Reliability: The use of stable, commercially available reagents ensures that the supply chain is not vulnerable to the shortages often associated with specialty toxic chemicals. The process flexibility, which allows for operation in standard stirred reactors or continuous flow setups, provides manufacturers with the agility to scale production up or down based on market demand without significant capital investment. This adaptability is crucial for maintaining continuous supply to downstream pharmaceutical customers, reducing lead times for high-purity intermediates and ensuring that production schedules are met consistently even in volatile market conditions.
- Scalability and Environmental Compliance: The patent explicitly highlights the potential for continuous operation, a key factor for industrial scale-up. Continuous processing offers better heat and mass transfer control, which is vital for maintaining product quality at larger scales. From an environmental perspective, the absence of heavy metals and sulfur-based toxins simplifies effluent treatment, making it easier to meet stringent environmental regulations. This compliance advantage reduces the risk of production shutdowns due to regulatory non-compliance and positions the manufacturer as a responsible partner in the global pharmaceutical value chain, facilitating smoother audits and long-term contracts.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the technical specifications and experimental data provided in the patent documentation, ensuring accuracy and relevance for industry professionals. Understanding these details is vital for assessing the feasibility of integrating this route into existing manufacturing portfolios.
Q: What are the primary safety advantages of this new synthesis method compared to the Tschugaeff reaction?
A: The novel method described in patent CN1345310A eliminates the need for highly toxic reagents such as carbon disulfide, methyl iodide, and methyl mercaptan, which were required in the traditional Tschugaeff reaction. Furthermore, it operates at significantly milder temperatures (-20°C to +20°C) compared to the pyrolysis conditions of 180°C to 190°C required by older methods, drastically reducing thermal hazards and energy consumption.
Q: How does this process improve yield and purity for pharmaceutical intermediates?
A: By utilizing specific alkali metal amides like LDA in conjunction with dialkyl carbonates, the process achieves yields up to 82% in laboratory examples, surpassing the 50% maximum theoretical yield often associated with racemate separation in achiral syntheses. The mild conditions also minimize side reactions, such as the undesired intermolecular reactions observed in previous carboxylation attempts, leading to a cleaner impurity profile suitable for high-purity API manufacturing.
Q: Is this synthesis route scalable for commercial production of thrombin inhibitor intermediates?
A: Yes, the patent explicitly notes that while the process can be run discontinuously in stirred reactors, the simplicity of the reaction scheme allows for adaptation into continuous operations using reactor tubes or series of stirred reactors. This flexibility supports the commercial scale-up of complex pharmaceutical intermediates, ensuring consistent supply chain reliability for downstream drug production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Acyl-3,4-Dehydroproline Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to maintain competitiveness in the global fine chemicals market. Our team of expert chemists has extensively analyzed the potential of the route described in CN1345310A and possesses the technical capability to translate this laboratory-scale innovation into commercial reality. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot plant to full-scale manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of N-acyl-3,4-dehydroproline or dehydropiperidine derivative meets the exacting standards required for pharmaceutical applications.
We invite you to collaborate with us to leverage this technology for your specific project needs. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your volume requirements and quality targets. We encourage potential partners to contact us directly to request specific COA data and route feasibility assessments, allowing you to make informed decisions that drive value and efficiency in your supply chain. Let us be your trusted partner in delivering high-quality intermediates that power the next generation of therapeutic solutions.
