Advanced Catalyst-Free Synthesis of Benzimidazole Derivatives for Scalable Pharmaceutical Manufacturing
Advanced Catalyst-Free Synthesis of Benzimidazole Derivatives for Scalable Pharmaceutical Manufacturing
The pharmaceutical industry constantly seeks robust synthetic routes that balance high purity with operational safety, particularly for complex heterocyclic scaffolds like benzimidazoles. A recent breakthrough detailed in patent CN107400090B introduces a transformative synthesis method for benzimidazole derivatives and their pharmaceutically acceptable salts. This innovation addresses critical bottlenecks in the production of key intermediates, such as 2-methyl-2-[[1-(4-cyanonaphthalen-1-yl)-1H-benzo[d]imidazol-2-yl]mercapto]propionic acid. By replacing hazardous reagents like sodium hydride (NaH) and expensive transition metal catalysts with safer inorganic bases like sodium hydroxide (NaOH) and calcium oxide (CaO), this technology offers a pathway to significantly lower manufacturing costs while enhancing process safety. For global procurement teams and R&D directors, this represents a pivotal shift towards more sustainable and economically viable pharmaceutical intermediate sourcing strategies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
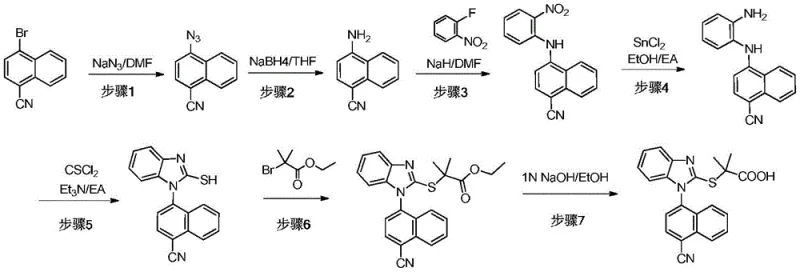
Historically, the synthesis of these critical benzimidazole structures relied heavily on harsh conditions that posed severe risks to both personnel and product quality. Traditional protocols often utilized sodium hydride (NaH) as a strong base to facilitate the coupling of naphthylamine derivatives. However, NaH is pyrophoric and reacts violently with moisture, leading to uncontrollable exotherms that make scale-up extremely dangerous. Furthermore, alternative pathways frequently employed palladium or copper-catalyzed cross-coupling reactions, such as Ullmann or Buchwald-Hartwig couplings. While effective in small batches, these metal-catalyzed routes introduce persistent heavy metal impurities that are difficult and costly to remove to meet stringent regulatory limits. The combination of low yields (often reported around 15% in background art), high safety risks, and complex purification requirements rendered these conventional methods unsuitable for cost-effective commercial scale-up of complex pharmaceutical intermediates.
The Novel Approach
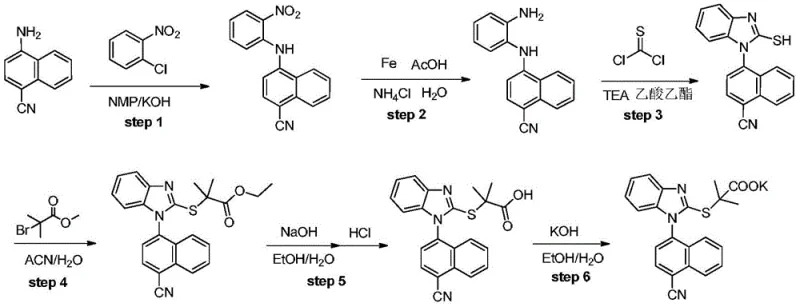
The inventive methodology described in the patent fundamentally re-engineers the synthetic logic by leveraging a mixed inorganic base system. Instead of relying on dangerous NaH, the process employs a combination of NaOH and CaO, or alternatively KOH, to drive the nucleophilic aromatic substitution. This modification not only mitigates the risk of fire and explosion but also effectively manages the reaction thermodynamics, preventing the runaway heat release associated with the old methods. Crucially, this approach eliminates the need for transition metal catalysts entirely. By utilizing a direct substitution mechanism activated by the ortho-nitro group, the process avoids heavy metal contamination at the source. This results in a cleaner crude product profile, simplifying downstream purification and drastically reducing the overall production timeline. This novel route stands as a superior solution for cost reduction in pharmaceutical intermediate manufacturing, delivering higher purity with fewer processing steps.
Mechanistic Insights into Base-Promoted Nucleophilic Substitution
The core chemical innovation lies in the synergistic effect of the mixed base system during the initial coupling step. In this mechanism, the presence of an electron-withdrawing nitro group at the ortho-position of the aniline ring significantly enhances the electrophilicity of the adjacent halogen atom on the naphthalene ring. The inorganic base, specifically the hydroxide ion from NaOH or KOH, deprotonates the amino group of the naphthylamine or aniline derivative, generating a highly nucleophilic amide anion. Simultaneously, the calcium oxide (CaO) acts as a potent water scavenger within the reaction medium. This is critical because water, often present in hygroscopic solvents like DMF or DMAc, can hydrolyze the sensitive nitrile groups or deactivate the base. By sequestering water, CaO maintains the anhydrous conditions necessary for high conversion rates without requiring the extreme dryness needed for NaH. This delicate balance ensures that the substitution proceeds smoothly to form the diphenylamine intermediate with minimal side reactions.
Following the coupling, the subsequent cyclization and functionalization steps are designed to maximize atom economy and minimize waste. The reduction of the nitro group to an amine is achieved using environmentally friendlier reagents like iron powder in acetic acid or hydrazine hydrate, avoiding high-pressure hydrogenation equipment. The cyclization to form the benzimidazole ring utilizes thiophosgene or N,N'-thiocarbonyldiimidazole (TCDI), which efficiently closes the ring to create the mercapto-benzimidazole core. Finally, the alkylation with ethyl 2-bromoisobutyrate and subsequent hydrolysis installs the propionic acid side chain. Throughout this cascade, the absence of metal catalysts means there is no need for expensive scavengers like silica-thiol or activated carbon treatments specifically for metal removal, further streamlining the high-purity pharmaceutical intermediate production workflow.
How to Synthesize Benzimidazole Derivatives Efficiently
The implementation of this synthesis route requires precise control over reaction parameters to replicate the high yields reported in the patent data. The process begins with the careful addition of the halogenated naphthalene to the base mixture at controlled temperatures to manage exotherms. Following the coupling, the workup involves simple acidification and filtration, bypassing complex chromatographic separations. The subsequent reduction and cyclization steps utilize common organic solvents like ethanol and acetonitrile, which are easily recovered and recycled. This operational simplicity makes the technology highly attractive for reliable pharmaceutical intermediate suppliers aiming to optimize their production lines. The detailed standardized synthesis steps for replicating this high-efficiency route are outlined below.
- Perform nucleophilic substitution using 4-bromo-1-naphthonitrile and 2-nitroaniline with NaOH/CaO mixed base in DMAc solvent.
- Reduce the nitro group to an amine using iron powder/acetic acid or hydrazine hydrate systems.
- Cyclize the diamine intermediate using thiophosgene or TCDI to form the benzimidazole core, followed by alkylation and hydrolysis.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route translates directly into tangible bottom-line improvements and risk mitigation. The shift away from hazardous reagents and precious metal catalysts addresses two of the most volatile cost drivers in fine chemical manufacturing. By eliminating the need for palladium or copper catalysts, the process removes the substantial expense associated with purchasing these metals and the subsequent validation required to prove their removal from the final API. Moreover, the replacement of NaH with stable inorganic bases like NaOH and CaO significantly lowers the cost of goods sold (COGS) by reducing safety infrastructure requirements and insurance premiums associated with handling pyrophoric materials. Patent data explicitly highlights a dramatic cost reduction, noting a decrease from over 150,000 RMB/kg in conventional methods to approximately 50,000 RMB/kg with this new technology, representing a massive opportunity for cost reduction in pharmaceutical intermediate manufacturing.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the elimination of expensive catalytic systems and the simplification of purification workflows. Traditional methods often require multiple recrystallizations or column chromatography to remove metal residues and side products generated by harsh bases. In contrast, the new method produces a cleaner crude product that can often be purified via simple recrystallization or filtration. This reduction in unit operations lowers energy consumption, solvent usage, and labor hours. Furthermore, the use of commodity chemicals like NaOH, CaO, and iron powder ensures that raw material costs remain stable and predictable, shielding the supply chain from the price volatility often seen with specialized organometallic reagents.
- Enhanced Supply Chain Reliability: Supply continuity is paramount for pharmaceutical production, and this method strengthens reliability by relying on widely available, non-controlled reagents. Sodium hydride and certain palladium catalysts can be subject to strict transportation regulations and supply shortages. By substituting these with common inorganic bases and iron powder, the manufacturing process becomes more resilient to logistical disruptions. The improved safety profile also means that production facilities face fewer regulatory hurdles and inspection delays, ensuring a consistent flow of materials. This stability allows for better inventory planning and reducing lead time for high-purity pharmaceutical intermediates, enabling faster response to market demands.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies safety and environmental challenges, but this invention is inherently designed for industrial expansion. The controlled exotherm achieved by the NaOH/CaO system prevents the thermal runaways that limit batch sizes in traditional NaH processes. Additionally, the absence of heavy metals simplifies wastewater treatment and waste disposal, aligning with increasingly strict environmental regulations. The process generates less hazardous waste, reducing disposal costs and environmental liability. This green chemistry approach not only facilitates commercial scale-up of complex pharmaceutical intermediates from kilogram to tonne scales but also enhances the corporate sustainability profile of the manufacturing partner.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this benzimidazole synthesis technology. These answers are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on how this method outperforms legacy techniques in terms of safety, yield, and purity. Understanding these distinctions is crucial for technical teams evaluating the feasibility of adopting this route for their specific supply chain needs.
Q: Why is the new NaOH/CaO base system superior to the traditional NaH method?
A: The traditional NaH method poses significant safety risks due to pyrophoric nature and violent exotherms, often yielding only 15%. The new NaOH/CaO system eliminates fire hazards, controls heat release effectively, and boosts yields to over 80%.
Q: Does this process involve heavy metal catalysts like Palladium or Copper?
A: No. Unlike Ullmann or Buchwald-Hartwig couplings that require expensive Pd or Cu catalysts and complex removal steps, this invention uses a catalyst-free nucleophilic substitution, ensuring the final product is free from heavy metal residues.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes. The process utilizes readily available inorganic reagents and avoids hazardous conditions, making it highly scalable. The controlled exotherm and simplified purification steps significantly reduce production costs and lead times.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzimidazole Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic technologies to maintain competitiveness in the global pharmaceutical market. Our team of expert chemists has thoroughly analyzed the route described in patent CN107400090B and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We are equipped with rigorous QC labs and state-of-the-art manufacturing facilities capable of meeting stringent purity specifications required for GMP environments. Our commitment to technical excellence ensures that we can deliver high-quality benzimidazole intermediates that adhere to the highest standards of safety and efficacy, providing our partners with a secure and reliable supply foundation.
We invite potential partners to engage with our technical procurement team to discuss how this innovative synthesis method can be tailored to your specific project requirements. By leveraging our expertise, you can access a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this catalyst-free process for your specific volume needs. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our capabilities can drive efficiency and value in your supply chain.
